A Comprehensive Guide to Cytosine Base Editor (CBE) Protocols in Plants: From Design to Validation
This article provides a detailed, step-by-step guide for researchers and scientists on implementing cytosine base editing (CBE) technology in plant systems.
A Comprehensive Guide to Cytosine Base Editor (CBE) Protocols in Plants: From Design to Validation
Abstract
This article provides a detailed, step-by-step guide for researchers and scientists on implementing cytosine base editing (CBE) technology in plant systems. It covers foundational principles of CBE architecture and mechanism, comprehensive methodological protocols for design, delivery, and regeneration, common troubleshooting and optimization strategies to enhance efficiency and specificity, and rigorous validation and comparative analysis techniques. The content is tailored to empower professionals in plant genomics and biotechnology to successfully apply this precise genome editing tool for functional genomics and crop improvement.
Understanding Cytosine Base Editors: Core Principles and Plant-Specific Considerations
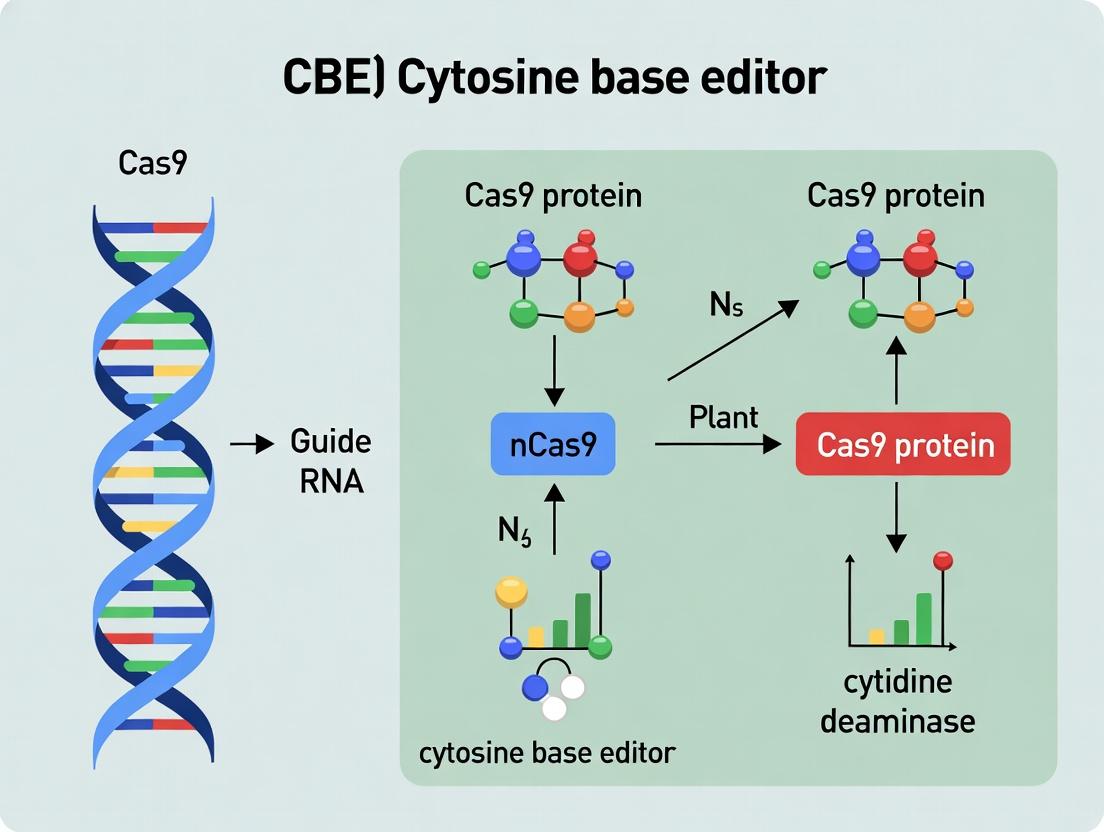
Within the context of plant research, Cytosine Base Editors (CBEs) enable precise, programmable C•G to T•A conversions without requiring double-stranded DNA breaks or donor templates. This technology is revolutionizing plant functional genomics and crop improvement by facilitating the creation of single-nucleotide polymorphisms (SNPs) and gene knockouts. The editing efficiency, precision, and product purity are critically dependent on the molecular architecture of the CBE, which integrates three core components: a cytidine deaminase, a Cas9 variant (nCas9 or nickase Cas9), and a connecting linker.
- Deaminase Domain: Catalyzes the hydrolytic deamination of cytidine to uridine. Common variants include APOBEC1 (from rat), CDA1 (from sea lamprey), and evolved versions like evoFERNY or evoAPOBEC1, which offer improved efficiency and reduced off-target activity.
- Cas9 Variant: A catalytically impaired Streptococcus pyogenes Cas9 (D10A mutation), nCas9, generates a single-strand nick in the non-edited DNA strand. This nick biases DNA repair to favor the U-containing strand, thereby increasing editing efficiency.
- Linker Design: The peptide linker connecting the deaminase and nCas9 influences spatial orientation, editing window width, and overall stability. Rigid (e.g., (EAAAK)n) or flexible (e.g., (GGGGS)n, XTEN) linkers yield different performance profiles.
This protocol details methods for assembling, testing, and quantifying the performance of CBE architectures in plant systems.
Experimental Protocols
Protocol 1: Modular Assembly of CBE Constructs for Plant Transformation
Objective: To assemble a plant-expression CBE vector using Golden Gate or Gateway cloning. Materials:
- Plant codon-optimized gene fragments: Deaminase (e.g., rAPOBEC1), Linker, nCas9 (D10A), and nuclear localization signals (NLS).
- Destination vector: A plant binary vector (e.g., pCAMBIA1300 derivative) with a plant promoter (e.g., 35S, UBI10) and terminator.
- Restriction enzymes/Cloning kits (Golden Gate: BsaI; Gateway: BP/LR Clonase).
- E. coli DH5α competent cells.
Procedure:
- Design: Assemble the construct in the order: Promoter - NLS - Deaminase - Linker - nCas9 - NLS - Terminator. Include a plant selectable marker (e.g., hptII for hygromycin).
- Golden Gate Assembly:
- Mix 50 ng of destination vector, equimolar amounts of each modular fragment (with compatible BsaI sites), 1.5 µL BsaI-HFv2, 1.5 µL T4 DNA Ligase, 2 µL 10x T4 Ligase Buffer, and H2O to 20 µL.
- Cycle: 25 cycles of (37°C for 3 min, 16°C for 4 min), then 50°C for 5 min, 80°C for 10 min.
- Transformation: Transform 5 µL of the reaction into E. coli DH5α, plate on appropriate antibiotics, and incubate overnight at 37°C.
- Screening: Pick colonies, perform colony PCR, and validate the final plasmid by Sanger sequencing.
Protocol 2: Agrobacterium-mediated Transformation inArabidopsis thaliana(Floral Dip)
Objective: To generate CBE-edited Arabidopsis plants. Materials:
- Construct from Protocol 1.
- Agrobacterium tumefaciens strain GV3101.
- Arabidopsis plants (e.g., Col-0) at early bolting stage.
- Silwet L-77, Sucrose, Murashige and Skoog (MS) salts.
Procedure:
- Agrobacterium Preparation: Transform the CBE binary vector into GV3101. Inoculate a single colony in 50 mL LB with appropriate antibiotics. Pellet cells at OD600 ~1.5 and resuspend in infiltration medium (5% sucrose, 0.05% Silwet L-77, 1/2x MS salts).
- Floral Dip: Submerge the aerial parts of flowering Arabidopsis plants in the Agrobacterium suspension for 30 seconds. Place plants sideways in a dark tray for 24h, then return to normal growth conditions.
- Seed Selection: Harvest seeds (T1). Surface sterilize and plate on MS agar plates containing the appropriate selection agent (e.g., hygromycin). Resistant green seedlings (T1) are potential transgenic edit events.
Protocol 3: Analysis of Editing Efficiency and Purity by Targeted Sequencing
Objective: To quantify C-to-T editing efficiency and identify byproducts. Materials:
- Plant genomic DNA extraction kit.
- PCR primers flanking the target site.
- High-fidelity PCR mix, gel extraction kit.
- Sanger sequencing or Next-Generation Sequencing (NGS) platform.
Procedure:
- DNA Extraction: Extract genomic DNA from leaf tissue of T1 plants.
- Target Amplification: Amplify the target region (~250-350 bp) using high-fidelity PCR.
- Sequencing & Analysis:
- For Sanger Sequencing: Purify PCR product and submit for sequencing. Analyze chromatograms for overlapping peaks using tools like EditR or Synthego ICE.
- For NGS: Barcode and pool amplicons from multiple plants. Perform paired-end sequencing (150 bp). Process data using a pipeline (e.g., CRISPResso2) with parameters for CBE analysis to calculate: % C-to-T conversion (efficiency), % of reads with intended edit (product purity), and indel frequency (an indicator of DSB formation).
Table 1: Performance of Common Deaminase Domains in Plants (Model Systems)
| Deaminase Variant | Origin | Avg. Editing Efficiency* (%) | Editing Window (Positions from PAM) | Notable Features |
|---|---|---|---|---|
| rAPOBEC1 | Rat | 15-40 | 3-9 | Standard, may have higher off-target activity |
| CDA1 | Sea Lamprey | 10-30 | 3-10 | Wider window, lower sequence context preference |
| evoAPOBEC1 | Evolved (rAPOBEC1) | 25-55 | 4-8 | High efficiency, reduced off-target deamination |
| evoFERNY | Evolved (C. elegans) | 20-50 | 3-9 | High efficiency, good performance in plants |
Efficiency varies by target site and species. Ranges are indicative for *Arabidopsis.
Table 2: Impact of Linker Composition on CBE Editing Profile
| Linker Type | Example Sequence | Key Characteristics | Effect on Editing Window (vs. Standard) |
|---|---|---|---|
| Flexible | (GGGGS)₃ | High conformational freedom | Can broaden or shift window |
| Rigid | (EAAAK)₃ | α-helical, reduces flexibility | Often narrows window, may increase efficiency at specific positions |
| XTEN | Long, disordered | Proteolytically stable, soluble | Can improve expression and overall activity |
Visualization: CBE Mechanism and Workflow
Title: CBE Molecular Mechanism in a Plant Cell
Title: CBE Plant Editing Experimental Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for CBE Research in Plants
| Item | Function in Protocol | Example/Supplier Consideration |
|---|---|---|
| Plant Codon-Optimized CBE Parts | Ensures high expression in plant cells. Key for efficiency. | Gene fragments from Twist Bioscience or IDT, codon-optimized for Arabidopsis or tobacco. |
| Plant Binary Vector | Agrobacterium-compatible T-DNA vector for plant transformation. | pCAMBIA1300, pGreenII, or pHEE401E (for CRISPR). |
| Golden Gate Assembly Kit | For seamless, modular assembly of CBE constructs. | NEB Golden Gate Assembly Kit (BsaI-HFv2) or similar. |
| Agrobacterium Strain | Mediates DNA transfer into plant genome. | GV3101 (pMP90) or EHA105 for Arabidopsis and many crops. |
| Silwet L-77 | Surfactant that enables Agrobacterium infiltration during floral dip. | Critical for high transformation efficiency. |
| Selection Antibiotics (Plant) | Selects for transformed tissue. | Hygromycin B, Kanamycin, or Glufosinate (Basta), depending on vector. |
| High-Fidelity PCR Mix | Accurate amplification of target genomic locus for sequencing analysis. | KAPA HiFi or Q5 Hot Start from NEB. |
| NGS Amplicon-EQ Library Kit | Prepares target amplicons for high-throughput sequencing to quantify editing. | Illumina DNA Prep or Swift Biosciences Accel-NGS 2S Plus. |
| CRISPResso2 Software | Computationally quantifies base editing efficiency and outcomes from NGS data. | Open-source tool for precise analysis. |
Within the broader thesis on developing and applying Cytosine Base Editor (CBE) protocols in plant research, understanding the precise biochemical mechanism is paramount. This application note details how CBEs achieve precise point mutations—specifically catalyzing the conversion of a C•G base pair to a T•A base pair—while avoiding the generation of deleterious double-strand breaks (DSBs). This DSB-free characteristic is a critical advantage over traditional CRISPR-Cas9 nuclease-based editing, as it minimizes unintended genomic rearrangements and is essential for applications in plant biology where karyotype stability is crucial.
Core Mechanism of Action: A Stepwise Biochemical Process
The canonical CBE system is a fusion protein comprising a catalytically impaired Cas9 nuclease (Cas9 nickase or dead Cas9) and a cytidine deaminase enzyme (e.g., APOBEC1). The mechanism proceeds via an orchestrated, multi-step pathway without DSB formation.
Detailed Stepwise Mechanism:
Targeted DNA Binding: The Cas9 component is guided by a single-guide RNA (sgRNA) to a specific genomic locus, where it unwinds the DNA duplex and binds to the target strand (the strand complementary to the sgRNA). The catalytically impaired Cas9 does not generate a DSB.
Deamination of Cytosine: Once the DNA is exposed in a transient, single-stranded state within the Cas9-sgRNA R-loop complex, the tethered cytidine deaminase acts on a defined window of cytosines (typically positions 3-9 within the protospacer, counting from the distal end). It catalyzes the hydrolytic deamination of cytosine (C) to form uracil (U) exclusively on the exposed single-stranded DNA. This converts a C•G pair to a U•G mismatch within the genome.
Cellular DNA Repair: The U•G mismatch is then recognized and processed by the cell's endogenous DNA repair machinery:
- Uracil N-Glycosylase Inhibition: Some advanced CBE architectures include a uracil glycosylase inhibitor (UGI) domain. UGI blocks the action of uracil DNA glycosylase (UDG), which would otherwise remove the uracil base, initiating error-prone repair pathways that could lead to undesired outcomes.
- DNA Replication or Mismatch Repair: During subsequent rounds of DNA replication, the U in the U•G pair is templated as thymine (T), leading to a T•A pair in one daughter strand. Alternatively, the mismatch repair (MMR) system may favor excision and replacement of the G (in the U•G mismatch) with an A, ultimately resulting in a permanent, precise C•G to T•A base pair substitution.
Table 1: Key Performance Metrics of Common CBEs in Plant Systems
| CBE Variant | Deaminase Domain | Efficiency Range* (%) | Typical Product Purity† (%) | Target Window (Positions) | Common Plants Tested |
|---|---|---|---|---|---|
| BE3 | rAPOBEC1 | 1-20 | ~50 | 4-8 | Arabidopsis, Rice |
| HF-BE3 | rAPOBEC1 (High-Fidelity Cas9) | 5-25 | ~60 | 4-8 | Rice, Wheat |
| A3A-PBE | A3A (Petromyzon marinus) | 10-40 | ~75 | 3-9 | Tomato, Potato |
| evoFERNY-CBE | evoFERNY | 15-50 | >90 | 3-10 | Rice, Maize |
| Target-AID | PmCDA1 | 5-30 | ~70 | 2-6 | Arabidopsis, Tobacco |
*Efficiency: Percentage of successfully edited alleles in transformed cells. †Purity: Percentage of edited products that are the desired C-to-T change without indels.
Table 2: Comparison of Editing Outcomes: CBE vs. CRISPR-Cas9 Nuclease
| Feature | Cytosine Base Editor (CBE) | CRISPR-Cas9 Nuclease (DSB-Dependent) |
|---|---|---|
| Primary Product | Precise C•G to T•A point mutation | Double-strand break (DSB) |
| DSB Formation | No | Yes |
| Major Repair Pathway Utilized | DNA replication / MMR | NHEJ / HDR |
| Indel Frequency | Very Low (<1% typically) | High (variable, often >10%) |
| Editing Window | Narrow (~5-7 nucleotides) | Single cleavage site |
| Theoretical Outcome Control | High (predictable base change) | Low (stochastic indels) |
Experimental Protocols
Protocol 1: In Vitro Deamination Assay to Validate CBE Activity
Purpose: To biochemically confirm the deaminase activity and target window of a purified CBE protein. Materials: Purified CBE protein, synthetic target DNA duplex with protospacer, appropriate sgRNA, reaction buffer (Tris-HCl, EDTA, DTT), UDG enzyme (negative control), UGI protein. Procedure:
- Assemble a 50 µL reaction containing 100 nM target DNA, 200 nM CBE protein, 400 nM sgRNA, and 1x reaction buffer.
- Incubate at 37°C for 60 minutes.
- Stop the reaction by adding Proteinase K and incubating at 55°C for 15 min.
- Purify the DNA product using a spin column.
- Treat half of the product with UDG (10 U, 37°C, 1 hr) to cleave any deaminated uracil-containing DNA.
- Analyze both (-UDG and +UDG) samples via denaturing gel electrophoresis or Sanger sequencing. Cleavage in the +UDG sample indicates successful C-to-U deamination.
Protocol 2: Plant Protoplast Transfection for CBE Efficiency Analysis
Purpose: To measure CBE editing efficiency and product purity in a plant cell system. Materials: Plant protoplasts (e.g., from Arabidopsis or rice callus), PEG-Ca²⁺ transformation solution, CBE expression plasmid (driven by plant promoter, e.g., 35S or Ubi), sgRNA expression construct, DNA extraction kit, PCR reagents, sequencing primers. Procedure:
- Isolate healthy protoplasts and adjust density to 2x10⁵ cells/mL.
- Mix 10 µg of CBE plasmid and 5 µg of sgRNA plasmid with 100 µL of protoplasts.
- Add 110 µL of 40% PEG-Ca²⁺ solution, mix gently, and incubate at room temp for 15-20 min.
- Dilute slowly with W5 solution, pellet cells, and resuspend in culture medium.
- Incubate in the dark for 48-72 hours.
- Harvest cells, extract genomic DNA.
- PCR-amplify the target locus and analyze by Sanger sequencing (trace decomposition) or Next-Generation Sequencing (NGS) for precise quantification of editing efficiency and byproduct profile.
Signaling Pathway & Workflow Diagrams
Diagram 1: CBE Catalytic Pathway from Binding to Edit
Diagram 2: Plant CBE Editing Experimental Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for CBE Plant Research
| Item | Function in CBE Experiments | Example/Note |
|---|---|---|
| CBE Expression Vector | Delivers the fusion gene (dCas9-Deaminase-UGI) into plant cells. | pZB-evoFERNY-CBE driven by ZmUbi promoter for monocots. |
| sgRNA Expression Cassette | Provides target specificity; often cloned into the same or a separate T-DNA. | Polymerized tRNA-gRNA units under AtU6 or OsU6 promoter. |
| Plant Delivery Agent | Facilitates entry of genetic material into plant cells. | Agrobacterium tumefaciens strain EHA105, or PEG for protoplasts. |
| Selection Agent | Selects for transformed plant tissues/cells. | Hygromycin B, Kanamycin, or herbicide (e.g., Basta/PPT). |
| Uracil DNA Glycosylase (UDG) | Used in in vitro assays to detect uracil formation (deamination activity). | Commercial enzyme for biochemical validation. |
| High-Fidelity PCR Kit | To accurately amplify the target genomic locus from edited plants without introducing errors. | Essential for pre-sequencing amplification. |
| Sanger Sequencing Primers | To sequence PCR products for initial confirmation of editing. | Flanking primers ~300-500bp from target site. |
| NGS Library Prep Kit | For deep sequencing to quantify editing efficiency, purity, and detect off-target effects. | Amplicon-seq or whole-genome sequencing kits. |
| Plant DNA Extraction Kit | To obtain high-quality, PCR-amplifiable genomic DNA from plant tissues. | Must handle polysaccharide and phenolic compounds. |
| Cellulase & Macerozyme | Enzymes for generating protoplasts from plant tissues for transient assays. | Critical for protoplast-based transformation and testing. |
Key Advantages of Base Editing Over Traditional CRISPR-Cas9 for Plant Research
Application Notes
Base editing, and specifically Cytosine Base Editors (CBEs), represent a transformative advancement in plant genome engineering. While traditional CRISPR-Cas9 relies on creating double-strand breaks (DSBs) and the subsequent error-prone repair pathways (NHEJ or HDR), CBEs directly and irreversibly convert one DNA base pair to another (C•G to T•A) without inducing DSBs. This technical distinction underpins critical advantages for plant research and development.
The primary advantages are summarized quantitatively below:
Table 1: Comparative Analysis of Key Editing Outcomes in Plants
| Parameter | Traditional CRISPR-Cas9 (DSB-dependent) | Cytosine Base Editing (DSB-independent) |
|---|---|---|
| Primary Product | Indels (insertions/deletions) | Precise point mutations (C-to-T) |
| Efficiency of Desired Point Mutation | Typically low (<10%) due to inefficient HDR in plants | Can be very high (often 10-50%, up to 80% reported in protoplasts) |
| Frequency of Unwanted Indels | High (primary outcome) | Very low to negligible (<1% in optimized systems) |
| Complexity of Donor Template | HDR requires co-delivery of a repair DNA template | No donor template required |
| Purity of Edited Line (Transgene-free) | Challenging due to random integration of donor template | Simplified; editor is transiently expressed, then segregated away |
| Multiplexing Capability | Possible but can cause chromosomal rearrangements | Highly suitable for multiplexed C-to-T changes |
| Applications | Gene knock-outs, large deletions, HDR-mediated knock-ins | Gene knock-outs (introduce premature stop codons), precise amino acid changes, correction of point mutations, directed evolution of traits |
Detailed Protocol: Application of CBE for Creating Herbicide Tolerance inArabidopsis
Objective: To introduce a specific C-to-T point mutation in the acetolactate synthase (ALS) gene, conferring resistance to chlorsulfuron herbicide, using a plant-optimized CBE (e.g., rAPOBEC1-nCas9-UGI).
I. Research Reagent Solutions Toolkit
Table 2: Essential Materials for CBE Plant Transformation
| Item | Function | Example/Supplier |
|---|---|---|
| Plant-Optimized CBE Expression Vector | All-in-one binary vector containing CBE components (e.g., cytidine deaminase, nCas9, UGI) under plant-specific promoters (e.g., AtU6, CaMV 35S). | pGTR-BE (Addgene), or pRCS11-35S::BE3 |
| sgRNA Expression Cassette | Targets the CBE complex to the specific cytidine within the ALS gene protospacer. | Cloned into the CBE vector's sgRNA scaffold region. |
| Agrobacterium tumefaciens Strain | Mediates stable plant transformation via floral dip. | GV3101 (pMP90) |
| Plant Material | Healthy, flowering Arabidopsis thaliana plants (e.g., Col-0 ecotype). | Grown under standard conditions. |
| Selection Antibiotics (Plant) | For selecting transformed seeds (T1). | e.g., Glufosinate (BASTA) if vector carries bar gene. |
| Selection Herbicide | For identifying successful ALS base edits. | Chlorsulfuron (e.g., 100 nM in 1/2 MS media). |
| PCR & Sanger Sequencing Primers | To amplify and sequence the target locus from putative edited plants. | Designed to flank the target window (15-25 bp around the target C). |
| High-Fidelity DNA Polymerase | For accurate amplification of the target locus for sequencing analysis. | Q5 High-Fidelity DNA Polymerase (NEB). |
| Tracking of Indels by Decomposition (TIDE) or BE-Analyzer | Online software tools for quantifying base editing efficiency from Sanger sequencing chromatograms. | https://tide.nki.nl/, http://www.rgenome.net/be-analyzer/ |
II. Step-by-Step Protocol
1. Target Selection and Vector Construction
- Identify the target adenine (complementary to the target cytosine) within the ALS gene coding sequence that, when changed to guanine (C-to-T on the opposite strand), will produce the desired amino acid change (e.g., Pro197Ser).
- Design a 20-nt sgRNA spacer sequence 5' of the Protospacer Adjacent Motif (PAM, NGG for SpCas9) such that the target cytosine is within positions 4-10 (deamination window) of the protospacer on the non-target strand.
- Clone the sgRNA expression cassette (AtU6 promoter::sgRNA scaffold) into the plant CBE binary vector using Golden Gate or restriction-ligation.
2. Plant Transformation
- Transform the assembled vector into Agrobacterium strain GV3101.
- Grow a 50 mL culture of the transformed Agrobacterium to OD600 ~1.5. Pellet cells and resuspend in 5% sucrose + 0.05% Silwet L-77 infiltration medium.
- Perform the floral dip method on primary inflorescences of 4-6 week-old Arabidopsis plants. Cover plants for 24h, then return to normal growth.
3. Selection and Screening of T1 Plants
- Harvest T1 seeds. Surface sterilize and sow on selective media (e.g., containing BASTA).
- After 7-10 days, transfer resistant (transgenic) seedlings to soil. Collect a small leaf sample for genotyping.
4. Genotyping and Efficiency Analysis
- Extract genomic DNA from leaf samples.
- Perform PCR using primers flanking the target site.
- Sanger sequence the PCR products.
- Analysis: Submit the Sanger sequencing traces (from edited plants and a wild-type control) to the BE-Analyzer web tool. The software will deconvolute the trace to calculate the percentage of C-to-T conversion at each position within the editing window.
5. Identification of Transgene-Free, Edited Plants (T2 Generation)
- Grow T1 plants to maturity and harvest T2 seeds individually.
- Plate T2 seeds on both BASTA (to select for the transgene) and chlorsulfuron (to select for the ALS edit). Identify lines where the herbicide resistance (edited trait) segregates independently from the BASTA resistance (transgene).
- In such lines, select healthy, chlorsulfuron-resistant plants that are BASTA-sensitive. These are transgene-free, base-edited plants.
- Confirm by PCR for the absence of the T-DNA and by sequencing for the homozygous presence of the intended point mutation.
Visualization
Diagram 1: CRISPR-Cas9 vs CBE Mechanism
Diagram 2: CBE Plant Experiment Workflow
Cytosine Base Editors (CBEs) enable precise, programmable C•G to T•A conversion without requiring double-stranded DNA breaks or donor DNA templates. Developed from CRISPR-Cas systems, CBEs consist of a catalytically impaired Cas protein (e.g., dCas9 or nickase Cas9, nCas9) fused to a cytidine deaminase enzyme. This Application Note details the evolutionary trajectory of CBEs, from initial proof-of-concept systems to sophisticated, high-fidelity variants, within the context of plant genome engineering research.
CBE Version Evolution and Key Characteristics
The evolution of CBEs has been driven by goals to improve editing efficiency, product purity (reduce indel formation), minimize off-target edits (both DNA and RNA), and expand targeting scope.
Table 1: Evolution and Quantitative Performance Summary of Major CBE Variants
| CBE Variant | Core Components (Fusion) | Key Innovation | Typical C•G to T•A Efficiency* | Indel Formation* | Primary Limitations |
|---|---|---|---|---|---|
| BE1 | dCas9 + rAPOBEC1 | Proof-of-concept; deaminase activity only | Very Low (<1%) | Very Low | No cellular repair engagement; inefficient. |
| BE2 | dCas9 + rAPOBEC1 + UGI | Addition of uracil glycosylase inhibitor (UGI) | Moderate (5-20%) | Low | Relies on endogenous repair; lower efficiency than BE3. |
| BE3 | nCas9 (D10A) + rAPOBEC1 + UGI | Nickase Cas9 creates nick in non-edited strand to bias repair. | High (10-50%) | Moderate (1-10%) | Significant off-target DNA/RNA editing; bystander edits. |
| BE4 | nCas9 + rAPOBEC1 + 2xUGI | Two UGI molecules to further inhibit UDG. | High (30-60%) | Reduced vs. BE3 | Improved product purity; RNA off-targets remain. |
| BE4max | nCas9 + rAPOBEC1 + 2xUGI + Optimized linkers & NLS | Codon, linker, and nuclear localization signal (NLS) optimization. | Very High (up to 1.8x BE4) | Low | Enhanced efficiency in mammalian cells; standard for plants. |
| HF-CBE (BE4h) | High-Fidelity nCas9 (e.g., SpCas9-HF1) + rAPOBEC1 + 2xUGI | HF-Cas9 reduces DNA off-target binding. | High (similar to BE4) | Low | Reduced DNA off-target; retains rAPOBEC1 RNA off-target. |
| SECURE-CBE (BE4-SECURE) | nCas9 + rAPOBEC1 mutants (e.g., R33A/K34A) + 2xUGI | Engineered deaminase with reduced RNA binding. | High (similar to BE4) | Low | Dramatically reduced RNA off-target editing. |
| eA3A-CBE (eA3A-BE4max) | nCas9 + engineered A3A (eA3A) + 2xUGI | Narrower editing window (positions 2-5 in protospacer); reduced bystander edits. | High (context-dependent) | Low | Tighter editing window; reduced RNA off-target vs. rAPOBEC1. |
| Target-AID | nCas9 (D10A) + PmCDA1 + UGI | Alternative deaminase (sea lamprey); often used in plants. | Moderate to High | Moderate | Different sequence context preference. |
| yCBE | nCas9 + hAPOBEC3A + UGI | Human A3A deaminase; broadened sequence context (TC motifs). | High | Moderate | Different sequence preference; potential for higher RNA off-target. |
*Efficiency and indel rates are highly dependent on target locus, cell type, delivery method, and organism. Values represent generalized trends from mammalian and plant literature.
Detailed Experimental Protocols for Plant Research
Protocol 1: Agrobacterium-Mediated Delivery of CBEs for Stable Transformation inArabidopsis
Objective: Generate stable, heritable C•G to T•A mutations in Arabidopsis thaliana.
Materials (Research Reagent Solutions):
- Binary Vector: e.g., pBEE4max or pBEE4-SECURE (Addgene) containing the CBE expression cassette (UBQ10 promoter-driven nCas9-2xUGI-rAPOBEC1, NOS terminator) and sgRNA (AtU6 promoter-driven).
- Agrobacterium tumefaciens Strain: GV3101 (pMP90).
- Plant Material: Arabidopsis seeds (ecotype Col-0).
- Infiltration Media: 5% (w/v) sucrose, 0.05% (v/v) Silwet L-77.
- Selection Antibiotics: For plants: Basta (glufosinate ammonium) or hygromycin, depending on vector marker.
Methodology:
- Vector Construction: Clone a 20-nt target spacer sequence into the sgRNA scaffold of your chosen CBE binary vector via Golden Gate or restriction-ligation.
- Agrobacterium Transformation: Introduce the assembled vector into A. tumefaciens GV3101 via electroporation.
- Plant Transformation (Floral Dip): a. Grow donor plants to the stage of numerous unopened floral buds. b. Culture Agrobacterium harboring the CBE vector to late-log phase (OD600 ~1.5). Pellet and resuspend in infiltration media to OD600 ~0.8. c. Immerse aerial parts of the plant in the suspension for 30 seconds with gentle agitation. d. Cover plants for 24h, then grow normally until seed set (T1 generation).
- Selection and Genotyping: a. Sow T1 seeds on soil or medium containing the appropriate antibiotic. b. Extract genomic DNA from resistant T1 plantlets. c. PCR-amplify the target region and submit for Sanger sequencing. Use decomposition tools (e.g., BEAT, EditR) or sequencing trace decomposition to calculate editing efficiency. d. Identify T1 plants with desired edits and screen for Cas9-free segregants in the T2 generation.
Protocol 2: Protoplast Transfection for Rapid CBE Efficiency Testing in Plants
Objective: Rapidly quantify CBE editing efficiency and profile in plant cells within 2-3 days.
Materials (Research Reagent Solutions):
- Expression Plasmids: 1. CBE expression vector (e.g., 35S::BE4max). 2. sgRNA expression vector (e.g., AtU6::sgRNA).
- Plant Material: Leaf tissue from 3-4 week old Nicotiana benthamiana or Arabidopsis.
- Enzyme Solution: 1.5% Cellulase R10, 0.4% Macerozyme R10, 0.4M mannitol, 20mM KCl, 20mM MES (pH 5.7), 10mM CaCl2, 0.1% BSA.
- PEG Solution: 40% PEG-4000, 0.2M mannitol, 0.1M CaCl2.
- W5 Solution: 154mM NaCl, 125mM CaCl2, 5mM KCl, 2mM MES (pH 5.7).
Methodology:
- Protoplast Isolation: a. Slice young leaves into 0.5-1mm strips and immerse in enzyme solution. b. Digest in the dark with gentle shaking (30-50 rpm) for 3-6 hours. c. Filter the digest through a 70μm nylon mesh. Pellet protoplasts by centrifugation at 100xg for 5 min. d. Wash pellet gently with W5 solution. Resuspend in W5 and incubate on ice for 30 min.
- Transfection: a. Pellet protoplasts (100xg, 5 min) and resuspend in MMg solution (0.4M mannitol, 15mM MgCl2, 4mM MES pH 5.7) at a density of 2x10^5 cells/mL. b. For each sample, mix 10μg total plasmid DNA (CBE:sgRNA at a 1:1 molar ratio) with 100μL protoplast suspension. c. Add 110μL of PEG solution, mix gently, and incubate for 15 min at room temperature. d. Dilute slowly with 0.5mL W5, then 1mL W5. Pellet cells (100xg, 5 min) and resuspend in 1mL culture medium (e.g., 0.4M mannitol, 4mM MES, 4mM KCl).
- Analysis: a. Incubate transfected protoplasts in the dark at 22-25°C for 48-72 hours. b. Harvest cells, extract genomic DNA. c. PCR amplify the target locus and perform next-generation sequencing (amplicon-seq) for precise quantification of C•G to T•A conversion and indel rates.
Visualizations
Diagram 1: Evolutionary Pathways of Key CBE Variants
Diagram 2: CBE4 Mechanism: Deamination, Nick, and Repair
Application Notes Cytosine base editor (CBE) application in plants confronts unique biological barriers absent in mammalian systems. Success hinges on overcoming the plant cell wall, achieving efficient delivery to meristematic or regenerable cells, and ensuring edited cells can regenerate into whole plants. Current research focuses on delivery methods like Agrobacterium-mediated transformation, biolistics, and novel nanomaterials, alongside regeneration-optimized tissue culture protocols. The editing window and bystander edits are critical quantitative parameters that must be characterized for each new plant system. The following data, derived from recent studies (2023-2024), highlights key performance metrics across delivery methods in model and crop plants.
Table 1: Quantitative Performance of CBE Delivery Methods in Plants
| Plant Species | Delivery Method | Target Tissue | Editing Efficiency (Range %) | Regeneration Efficiency (% of explants) | Key Citation/DOI |
|---|---|---|---|---|---|
| Nicotiana benthamiana | Agrobacterium (Leaf disc) | Leaf mesophyll | 15.2 - 58.7 | 85-95 | Zhang et al., 2023, 10.1038/s41477-023-01412-1 |
| Rice (Oryza sativa) | Agrobacterium (Callus) | Embryogenic callus | 2.1 - 41.5 | 60-75 | Liu et al., 2023, 10.1038/s41587-023-01710-1 |
| Maize (Zea mays) | Biolistics (Particle Gun) | Immature embryos | 1.8 - 23.4 | 30-50 | Wang et al., 2023, 10.1111/tpj.16145 |
| Wheat (Triticum aestivum) | Agrobacterium (Callus) | Embryogenic callus | 0.7 - 19.8 | 20-40 | Li et al., 2024, 10.1093/plcell/koad293 |
| Potato (Solanum tuberosum) | Nanoparticle (Citrate-coated) | Leaf protoplasts | 5.6 - 12.3 | N/A (Protoplasts) | Chen et al., 2024, 10.1038/s41565-023-01585-y |
| Arabidopsis thaliana | Floral Dip | Female gametophyte | 0.5 - 5.2 | N/A (Seed set) | Arora et al., 2023, 10.1111/tpj.16421 |
Protocol 1: Agrobacterium-Mediated CBE Delivery in Rice Embryogenic Callus Objective: Achieve heritable CBE edits in regenerated rice plants. Materials: Japonica rice seeds, CBE expression vector (pRGEB32-CBE), A. tumefaciens strain EHA105, N6D, N6-1, N6-2, and selection media. Procedure:
- Callus Induction: Surface-sterilize mature seeds. Culture on N6D medium (2,4-D 2 mg/L) for 4 weeks at 28°C in dark.
- Agrobacterium Preparation: Transform EHA105 with pRGEB32-CBE. Grow a single colony in YEP + antibiotics to OD₆₀₀ = 0.8. Pellet and resuspend in AAM medium + 100 µM acetosyringone.
- Co-cultivation: Immerse calli in Agrobacterium suspension for 20 min. Blot dry and co-culture on N6-1 medium for 3 days at 22°C in dark.
- Selection & Regeneration: Transfer calli to N6D selection medium (Hygromycin 50 mg/L) for 4 weeks, subculturing every 2 weeks. Move resistant calli to Regeneration medium (N6-3, kinetin 2 mg/L) for shoot induction (2-4 weeks), then to root medium.
- Molecular Analysis: Extract genomic DNA from regenerated plantlets. PCR-amplify target locus and perform Sanger sequencing. Analyze editing efficiency via BE-Analyzer or EditR.
Protocol 2: Protoplast Isolation & CBE RNP Transfection for Rapid Validation Objective: Quickly test CBE activity and specificity in a plant system pre-regeneration. Materials: Young leaves, Cellulase R10, Macerozyme R10, Mannitol, PEG4000, CBE ribonucleoprotein (RNP) complex. Procedure:
- Protoplast Isolation: Slice 1g of leaf tissue into thin strips. Digest in enzyme solution (1.5% Cellulase, 0.4% Macerozyme, 0.4M Mannitol) for 6-16 hours in dark with gentle shaking.
- Purification: Filter digest through 75µm mesh. Wash protoplasts 3x with W5 solution via centrifugation (100xg, 2 min). Resuspend in MMg solution, count, and adjust to 2x10⁵ cells/mL.
- RNP Transfection: Pre-assemble CBE protein with sgRNA (molar ratio 1:3) for 10 min at 25°C. Mix 10µL protoplasts with 10µL RNP complex. Add 20µL PEG4000 (40% w/v), mix gently, incubate 15 min.
- Post-Transfection: Dilute with 1mL W5, pellet cells. Resuspend in culture medium. Incubate 48-72 hours.
- Analysis: Harvest protoplasts, extract DNA. Use targeted deep sequencing (e.g., Illumina MiSeq) to quantify editing efficiency and byproduct profile.
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in Plant CBE Work | Example Product/Catalog # |
|---|---|---|
| CBE Plasmid System | Expresses base editor and sgRNA in plant cells. | pRGEB32-CBE (Addgene #157163) |
| Cellulase R10 | Degrades cellulose in cell wall for protoplast isolation. | Yakult Pharmaceutical, C8001 |
| Macerozyme R10 | Degrades pectin in cell wall for protoplast isolation. | Yakult Pharmaceutical, M8002 |
| Acetosyringone | Phenolic compound inducing Agrobacterium vir genes. | Sigma-Aldrich, D134406 |
| Hygromycin B | Selective agent for transformed plant tissues. | Thermo Fisher, 10687010 |
| PEG4000 | Polyethylene glycol mediating protoplast transfection. | Sigma-Aldrich, 81240 |
| BE-Analyzer Software | Computational tool for deconvoluting Sanger sequencing to quantify base editing. | https://github.com/maximanilow/BE-Analyzer |
Diagram 1: CBE Plant Workflow & Barriers
Diagram 2: CBE Mechanism in Plant Cell
Step-by-Step CBE Protocol for Plants: Design, Delivery, and Regeneration
Within the broader thesis on developing and applying Cytosine Base Editor (CBE) systems in plants, the design of the single guide RNA (sgRNA) is the most critical determinant of experimental success. CBEs, typically fusing a cytidine deaminase (e.g., APOBEC1) to a Cas9 nickase (nCas9), enable programmable C-to-T (or G-to-A) conversion without inducing double-strand breaks. In plant systems, where precise point mutations can confer herbicide resistance, improved yield, or altered metabolic pathways, accurate prediction of the editing outcome is paramount. This protocol details the integrated bioinformatic and experimental workflow for selecting target sites, assessing PAM compatibility for common Cas9 variants, and predicting the editing window to maximize efficiency and minimize off-target effects in plant genomes.
Core Principles and Quantitative Data
The editing window of a CBE is constrained by the steric accessibility of the DNA strand to the deaminase domain relative to the bound Cas9. For the widely used BE3/BE4 architecture in plants, the optimal deamination window is typically positions 4-8 (1-based indexing) within the protospacer, measuring from the distal end of the PAM. Editing efficiency drops sharply outside this window.
Table 1: Key CBE Variants and Their PAM Requirements in Plant Systems
| CBE System (Common Name) | Cas9 Variant | PAM Sequence (5' → 3') | Optimal Editing Window* (Protospacer Position) | Typical Efficiency Range in Plants (C-to-T) |
|---|---|---|---|---|
| BE3, BE4, BE4max | Streptococcus pyogenes Cas9n (SpCas9) | NGG (canonical) | 4-10 (peak 4-8) | 10-50% (stable lines) |
| HF-BE, YE1-BE | High-Fidelity SpCas9 (SpCas9-HF1) | NGG | 4-8 | 5-30% |
| SaBE | Staphylococcus aureus Cas9n (SaCas9) | NNGRRT (or NNGRRN) | 3-9 | 5-25% |
| CBE-ABE (using Cas12a) | Francisella novicida Cas12a (FnCas12a) | TTTV (T-rich PAM) | 12-17† | 1-15% (less common) |
*Positions are 1-based, where position 1 is the first base upstream of the PAM (NGG PAM = positions 21-23 for a 20-nt spacer). †Editing window for Cas12a-based editors is measured differently due to distant PAM.
Table 2: Quantitative Factors Influencing sgRNA Efficiency for CBE in Plants
| Factor | Optimal Characteristic | Rationale & Impact |
|---|---|---|
| GC Content | 40-60% | Very low (<20%) or high (>80%) GC can impair sgRNA expression (Pol III promoter) and stability. |
| Target Site Context | Avoid sequences 5'-TC-3' or 5'-CC-3' immediately 5' of target C | Deaminase preference: RC>YC (R=A/G; Y=C/T). TC and CC are preferred motifs. |
| Poly-T Stretch | Avoid ≥4 consecutive T's | Acts as a termination signal for U6/U3 Pol III promoters in sgRNA expression vectors. |
| Seed Region (PAM-proximal) | Low tolerance for mismatches (bases 1-12) | Critical for specific target DNA binding. Mismatches here drastically reduce on-target activity. |
| Off-Target Potential | Maximize unique sequence in genome | Use BLAST against the specific plant genome (e.g., TAIR for Arabidopsis, EnsemblPlants for crops). |
| Genomic Feature | Consider nucleosome positioning data if available | Accessible chromatin (e.g., DNase I hypersensitive sites) enhances editing efficiency. |
Integrated Protocol for sgRNA Design and Validation
Protocol 3.1: In Silico Target Site Selection and sgRNA Design
Objective: To computationally identify and rank optimal sgRNA targets for CBE application in a plant gene of interest.
Materials & Reagents:
- Plant Genomic Sequence: FASTA file of the target gene locus or whole chromosome.
- Bioinformatics Tools: Local command-line tools (e.g., CRISPResso2, BEDTools) or web servers (CRISPR-P 2.0, CROPSR, Cas-Designer).
- Computer: With internet access and standard specs for sequence analysis.
Procedure:
- Define Target Region: Identify the exonic region(s) containing the cytidine(s) to be edited. A 200-300 bp window around the codon of interest is typical.
- Scan for PAM Sequences: a. For SpCas9-based CBE, scan the both strands of the target sequence for all instances of the 5'-NGG-3' PAM. b. Extract the 20-nt protospacer sequence immediately upstream (5') of each identified NGG PAM on the same strand.
- Filter and Rank sgRNAs: a. Filter 1 (Essential): Discard any sgRNA whose protospacer contains a 5'-TTTT-3' (poly-T) sequence or lacks a cytidine within positions 4-10 relative to the PAM. b. Filter 2 (Specificity): Perform a genome-wide BLASTN search of the remaining 20-nt spacer sequences against the reference genome of your plant species. Discard sgRNAs with perfect or near-perfect (≤2 mismatches) matches at multiple genomic loci. c. Ranking: Prioritize sgRNAs based on: i. Presence of multiple target Cs within positions 4-8. ii. Target C within a 5'-TC-3' or 5'-CC-3' context. iii. Moderate GC content (40-60%). iv. Low predicted off-target score (use scores from tools like CRISPR-P 2.0).
- Select 3-5 Final Candidates: Choose top-ranked sgRNAs for synthesis. Always design sgRNAs targeting both the sense and antisense strands of the genomic DNA to empirically determine the most efficient orientation.
Protocol 3.2: Experimental Validation of Editing Window via Amplicon Sequencing
Objective: To empirically determine the base editing efficiency and window profile of a designed sgRNA in planta.
Materials & Reagents (The Scientist's Toolkit):
Table 3: Essential Research Reagent Solutions for CBE sgRNA Validation in Plants
| Item | Function | Example/Supplier |
|---|---|---|
| Plant-Specific CBE Expression Vector | Delivers nCas9-deaminase fusion and sgRNA expression cassettes. | pBE3/pBEE4 series (Addgene), pRCS系列 for monocots. |
| sgRNA Cloning Kit | Facilitates insertion of oligonucleotides into the sgRNA scaffold vector. | Golden Gate (BsaI) or Type IIS assembly kits. |
| Agrobacterium tumefaciens Strain | For stable plant transformation (dicots) or transient expression. | GV3101, EHA105, LBA4404. |
| Plant Tissue Culture Media | For selection and regeneration of transformed tissue. | MS basal medium with appropriate hormones (e.g., 2,4-D for callus). |
| High-Fidelity PCR Master Mix | For specific amplification of the target genomic locus from edited tissue. | Q5 (NEB), KAPA HiFi (Roche). |
| PCR Purification Kit | Cleans up amplicons for sequencing library prep. | QIAquick (Qiagen), AMPure XP beads (Beckman). |
| Next-Generation Sequencing (NGS) Library Prep Kit | Prepares barcoded amplicon libraries for multiplexed sequencing. | NEBNext Ultra II DNA Library Prep. |
| Bioinformatics Analysis Pipeline | Quantifies base editing frequencies from NGS data. | CRISPResso2, BATCH-GE, BE-Analyzer. |
Procedure:
- Construct Assembly: Clone the oligonucleotide duplex encoding the selected 20-nt protospacer sequence into the sgRNA expression vector. Subsequently, assemble the final T-DNA vector containing the CBE and sgRNA expression cassettes.
- Plant Transformation: Introduce the assembled vector into your plant system (e.g., Agrobacterium-mediated transformation of Arabidopsis, rice callus, or leaf disc inoculation for Nicotiana).
- Sample Harvest: For stable transformation, collect leaf tissue from primary transformants (T0 or T1). For transient assays (e.g., protoplast or agroinfiltration), harvest tissue 2-5 days post-transfection.
- Genomic DNA Extraction: Use a standard CTAB or commercial kit method to extract high-quality gDNA.
- Target Region Amplification: Design primers ~150-250 bp flanking the target site. Perform PCR using a high-fidelity polymerase.
- Amplicon Sequencing (NGS): Purify PCR products, prepare barcoded NGS libraries, and sequence on an Illumina MiSeq or similar platform (aim for >10,000x read depth per sample).
- Data Analysis: Use CRISPResso2 with the following command to quantify editing:
Visual Workflows and Diagrams
Title: CBE sgRNA Design and Validation Workflow
Title: CBE Optimal Deamination Window on Protospacer
In plant cytosine base editor (CBE) research, precise vector construction is paramount for achieving efficient and reliable genome editing. The editor's components—a Cas9 nickase fused to a cytidine deaminase and a uracil glycosylase inhibitor (UGI)—must be expressed at optimal levels and ratios to maximize on-target conversion while minimizing off-target effects and cellular toxicity. This document provides application notes and protocols for selecting and deploying regulatory elements, specifically promoters and terminators, to fine-tune CBE expression in plants, directly supporting thesis research focused on developing optimized CBE protocols for crop improvement.
Promoter Selection: Quantitative Comparison
The choice of promoter dictates the spatiotemporal expression pattern and strength of the CBE machinery. Recent data (2023-2024) from studies in Nicotiana benthamiana, Arabidopsis, and rice provide comparative insights.
Table 1: Comparison of Key Promoters for CBE Expression in Plants
| Promoter | Organism Origin | Expression Profile | Relative Strength (Normalized to 35S) | Best Use Case in CBE Research | Key Considerations |
|---|---|---|---|---|---|
| CaMV 35S | Cauliflower Mosaic Virus | Constitutive, strong in most tissues. | 1.0 (Reference) | Rapid transient assays; strong, ubiquitous editing. | Can be too strong, potentially increasing off-targets; silencing in monocots. |
| ZmUbi (Maize Ubiquitin) | Maize (Zea mays) | Constitutive, very strong in monocots. | 1.2 - 1.5 (in monocots) | Stable transformation of cereals (rice, wheat, maize). | Less effective in dicots; large size (~2 kb). |
| AtUbi10 (Arabidopsis Ubiquitin) | Arabidopsis thaliana | Constitutive, strong in dicots. | 0.9 - 1.1 (in dicots) | Stable Arabidopsis transformation; reliable constitutive expression. | Well-characterized in model dicots. |
| OsAct1 (Rice Actin) | Rice (Oryza sativa) | Constitutive, strong in monocots. | 1.1 - 1.3 (in monocots) | Alternative to ZmUbi for rice and other grasses. | Intron required for full activity. |
| EC1 | Arabidopsis thaliana | Egg cell-specific, early embryo. | Varies by tissue | Heritable edits: drives expression in the germline to increase editing transmission. | Minimal expression in somatic tissues. |
| RPS5a | Arabidopsis thaliana | Meristematic and dividing cells. | Moderate | Enriching edits in meristems for potential germline transmission. | Tissue-specific, not universal. |
Terminator Selection and Impact
Terminators ensure proper mRNA 3' end formation, influencing mRNA stability and translation efficiency. Using mismatched or weak terminators can dramatically reduce expression.
Table 2: Common Terminators and Their Performance
| Terminator | Origin | Function | Recommended Pairing |
|---|---|---|---|
| 35S terminator | Cauliflower Mosaic Virus | Standard terminator, reliable. | With 35S or other viral/dicot promoters. |
| Nos terminator | Agrobacterium tumefaciens | Widely used, moderate efficiency. | Versatile, but may be weaker than specialized terminators. |
| AtUbi10 terminator | Arabidopsis thaliana | Native terminator for AtUbi10 promoter. | Optimal with AtUbi10 promoter for synthetic gene cassette fidelity. |
| ZmUbi terminator | Maize | Native terminator for ZmUbi promoter. | Critical for maximal ZmUbi-driven expression in monocots. |
| rbcS E9 terminator | Pea (Pisum sativum) | Enhances mRNA stability; often yields high expression. | Can be used as a strong universal terminator in many dicots. |
Core Protocol: Assembling a CBE Expression Vector for Plants
Protocol 1: Modular Golden Gate Assembly for Promoter/Terminator Testing
Objective: Rapidly assemble vectors with different promoter-CBE-terminator combinations to test expression and editing efficiency. Reagents: See "The Scientist's Toolkit" below.
Method:
- Design: Select your CBE cassette (e.g., nCas9-PmCDA1-UGI). Flank it with BsaI recognition sites, removing internal BsaI sites via silent mutagenesis.
- Prepare Modules: Clone your candidate promoters (35S, ZmUbi, AtUbi10, EC1) and terminators (Nos, E9, native) into separate entry vectors with compatible BsaI overhangs (e.g., Level 0 MoClo parts).
- Golden Gate Reaction:
- Set up a 20 µL Golden Gate assembly mix:
- 50 ng of BsaI-digested destination vector (Level 1 or 2 binary vector).
- 30 fmol of each module (Promoter, CBE CDS, Terminator).
- 1.5 µL T4 DNA Ligase Buffer (10X).
- 1 µL BsaI-HFv2 (NEB).
- 1 µL T4 DNA Ligase (HC, NEB).
- Nuclease-free water to 20 µL.
- Run thermocycler program: (37°C for 2 min, 16°C for 5 min) x 25 cycles, 50°C for 5 min, 80°C for 10 min.
- Set up a 20 µL Golden Gate assembly mix:
- Transformation & Validation: Transform 2 µL into E. coli DH5α, screen colonies by colony PCR and Sanger sequencing to confirm assembly.
- Plant Transformation: Use confirmed binary vectors for Agrobacterium-mediated transformation (stable or transient) of your target plant species.
Protocol 2: Transient Expression Assay inN. benthamianafor Quick Validation
Objective: Quickly compare the expression strength and nuclear localization of different CBE constructs before stable transformation.
Method:
- Agrobacterium Preparation:
- Transform each assembled binary vector into Agrobacterium tumefaciens strain GV3101.
- Inoculate a single colony in 5 mL LB with appropriate antibiotics (rifampicin, gentamicin, spectinomycin). Grow at 28°C, 220 rpm for 24h.
- Induction:
- Pellet cells at 4000 rpm for 10 min. Resuspend in infiltration buffer (10 mM MES, 10 mM MgCl2, 150 µM acetosyringone, pH 5.6) to an OD600 of 0.5.
- Incubate at room temperature for 2-4 hours in the dark.
- Infiltration:
- Use a 1 mL needleless syringe to infiltrate the Agrobacterium suspension into the abaxial side of 3-4 week-old N. benthamiana leaves.
- Infiltrate multiple spots per leaf, labeling constructs clearly.
- Analysis (3 days post-infiltration):
- Fluorescence: If using a fluorescent protein tag, visualize under a confocal microscope.
- Protein Extraction: Grind leaf discs in extraction buffer, perform Western blot with anti-Cas9 or anti-HA antibodies for quantification.
- DNA Extraction: Extract genomic DNA to assess preliminary on-target editing via PCR/restriction enzyme (RE) assay or sequencing.
Visualizing Construct Design and Workflow
Diagram 1: CBE Expression Cassette Optimization Workflow
Diagram 2: Basic CBE Gene Cassette Structure
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Vector Construction and CBE Testing
| Item | Supplier Examples | Function in Protocol |
|---|---|---|
| BsaI-HFv2 Restriction Enzyme | New England Biolabs (NEB) | Creates specific overhangs for Golden Gate assembly. |
| T4 DNA Ligase (High Concentration) | NEB, Thermo Fisher | Joins DNA fragments with compatible overhangs during Golden Gate. |
| MoClo-Compatible Plant Parts (Level 0) | Addgene, in-house libraries | Source of standardized promoter, CDS, and terminator modules. |
| pICH47732 (Level 1 Binary Vector) | Addgene (#48000) | Common destination vector for assembling transcriptional units. |
| Agrobacterium tumefaciens GV3101 | Various strain collections | Standard strain for plant transformation (transient and stable). |
| Acetosyringone | Sigma-Aldrich | Phenolic inducer of Agrobacterium vir genes for T-DNA transfer. |
| Anti-Cas9 Monoclonal Antibody | Diagenode, Cell Signaling | Detects Cas9 fusion protein expression in Western blots. |
| Guide-It Genomic Cleavage Detection Kit | Takara Bio | Detects on-target editing efficiency via mismatch cleavage assay. |
| Plant DNA Isolation Kit (CTAB method) | - | Reliable DNA extraction for PCR-based editing analysis. |
| Confocal Microscope (e.g., LSM 900) | Zeiss | Visualizes subcellular localization of fluorescently tagged CBE. |
Within the context of advancing plant genome editing using Cytosine Base Editors (CBEs), the selection of an appropriate delivery method is paramount. This article details three principal delivery strategies—Agrobacterium-mediated transformation, protoplast transfection, and delivery via pre-assembled Ribonucleoprotein (RNP) complexes. Each method presents unique advantages in terms of efficiency, species range, regulatory status (e.g., transgene-free editing), and applicability to different plant tissues. The protocols herein are framed for CBE delivery, where the precise, programmable deamination of cytosine to thymine is the goal, enabling targeted point mutations without double-strand DNA breaks.
Application Notes & Comparative Analysis
Agrobacterium-Mediated Transformation
- Principle: Utilizes the natural DNA transfer capability of Agrobacterium tumefaciens to deliver T-DNA containing CBE expression cassettes (e.g., nCas9-DDD-APOBEC1 and UGI) into the plant genome.
- Key Applications: Stable transformation and regeneration of edited plants; creation of a segregating population to obtain transgene-free edited lines; broad host range, especially in dicots.
- Advantages: Produces stable, heritable edits; allows for selection and regeneration of whole plants; well-established for many species.
- Disadvantages: Lengthy process; involves genomic integration of T-DNA (requiring segregation); lower efficiency in some monocots; potential for somaclonal variation.
Protoplast Transfection
- Principle: Isolation of plant cells without cell walls (protoplasts) followed by polyethylene glycol (PEG)-mediated or electroporation-driven delivery of CBE-encoding plasmid DNA or mRNA.
- Key Applications: Rapid assessment of CBE efficiency and specificity in a transient system; editing in species recalcitrant to Agrobacterium; high-throughput screening.
- Advantages: High transient transformation efficiency; applicable to a wide range of species; enables quantitative analysis at the cellular level; no T-DNA integration.
- Disadvantages: Regeneration of whole plants from protoplasts is difficult and genotype-dependent; edits are not heritable unless plants are regenerated.
Ribonucleoprotein (RNP) Complex Delivery
- Principle: Direct delivery of in vitro pre-assembled complexes of purified Cas9 nickase protein fused to cytidine deaminase and guide RNA (gRNA) into plant cells or tissues, typically via PEG-mediated transfection of protoplasts or biolistics.
- Key Applications: Generation of transgene-free edited plants; rapid, transient activity minimizing off-target effects; ideal for species with strict GMO regulations.
- Advantages: No foreign DNA integration; rapid degradation reduces off-target potential; immediate activity upon delivery.
- Disadvantages: Technically challenging protein production/purification; delivery efficiency can be lower than DNA methods; limited to tissues amenable to transfection or bombardment.
Quantitative Data Comparison
Table 1: Comparison of Key Parameters for CBE Delivery Methods
| Parameter | Agrobacterium-Mediated | Protoplast Transfection (DNA) | RNP Delivery (Protoplast) |
|---|---|---|---|
| Typical Editing Efficiency* | 1-30% (in regenerated plants) | 10-40% (transient, cells) | 5-20% (transient, cells) |
| Time to Result (Initial) | Weeks to months (regeneration) | 2-7 days (molecular analysis) | 2-3 days (molecular analysis) |
| Transgene Integration | Yes (requires segregation) | Possible (if DNA integrates) | No |
| Plant Regeneration Feasibility | High (inherent to method) | Low to very low | Low to very low |
| Species Range | Broad (dicots > monocots) | Very broad | Very broad |
| Primary Use Case | Stable line generation | Rapid in vitro screening | Transgene-free editing |
| Technical Complexity | Moderate | High (protoplast isolation) | Very High (RNP production) |
*Efficiency is highly target- and species-dependent. Data represents common ranges reported in recent literature.
Detailed Protocols
Protocol 1: Agrobacterium-Mediated CBE Delivery inNicotiana benthamianaLeaves (Transient Assay)
- Objective: To rapidly assess CBE function and efficiency in planta via transient expression.
- Materials: See Scientist's Toolkit.
- Method:
- Vector Construction: Clone your target gRNA into a binary vector containing your CBE (e.g., BE3, rAPOBEC1-nCas9-UGI) under a plant promoter (e.g., CaMV 35S).
- Agrobacterium Preparation: Transform the binary vector into A. tumefaciens strain GV3101. Inoculate a single colony in 5 mL LB with appropriate antibiotics. Grow overnight at 28°C, 200 rpm.
- Induction: Pellet cells at 4000 rpm for 10 min. Resuspend in infiltration medium (10 mM MES, 10 mM MgCl₂, 150 µM Acetosyringone, pH 5.6) to an OD₆₀₀ of ~0.5. Incubate at room temperature for 2-4 hours.
- Infiltration: Using a needleless syringe, press the tip against the abaxial side of a young N. benthamiana leaf and slowly infiltrate the bacterial suspension.
- Sampling & Analysis: Harvest leaf discs 3-5 days post-infiltration. Extract genomic DNA and assess editing efficiency by targeted PCR followed by Sanger sequencing and decomposition analysis (e.g., using ICE or EditR) or deep sequencing.
Protocol 2: PEG-Mediated CBE RNP Delivery into Arabidopsis Protoplasts
- Objective: To achieve DNA-free cytosine base editing in plant cells.
- Materials: See Scientist's Toolkit.
- Method:
- Protoplast Isolation: a. Harvest leaves from 3-4 week old Arabidopsis plants. b. Slice leaves into thin strips in enzyme solution (1.5% Cellulase R10, 0.4% Macerozyme R10, 0.4 M Mannitol, 20 mM KCl, 20 mM MES pH 5.7, 10 mM CaCl₂, 0.1% BSA). c. Vacuum-infiltrate for 30 min, then digest in the dark for 3-4 hours with gentle shaking. d. Filter through a 70 µm nylon mesh. Pellet protoplasts at 100 x g for 3 min. e. Wash twice with W5 solution (154 mM NaCl, 125 mM CaCl₂, 5 mM KCl, 2 mM MES pH 5.7). f. Resuspend in MMg solution (0.4 M Mannitol, 15 mM MgCl₂, 4 mM MES pH 5.7) and count cells.
- RNP Complex Assembly: Pre-assemble purified CBE protein (e.g., purified recombinant BE protein) with chemically synthesized or in vitro transcribed sgRNA at a molar ratio of 1:2-1:5 in nuclease-free buffer. Incubate at 25°C for 10 min.
- PEG Transfection: Aliquot 2 x 10⁴ protoplasts in 20 µL MMg. Add 5 µL of assembled RNP complexes. Add 45 µL of freshly prepared PEG solution (40% PEG4000, 0.2 M Mannitol, 0.1 M CaCl₂). Mix gently and incubate at room temperature for 15 min.
- Dilution & Recovery: Slowly add 200 µL of W5 solution, mix, and then add 1 mL more W5. Pellet cells at 100 x g for 3 min. Resuspend in 1 mL of culture medium. Incubate in the dark for 48-72 hours.
- Analysis: Harvest protoplasts, extract genomic DNA, and analyze editing at the target site via PCR and deep sequencing.
Visualization: CBE Delivery Workflow & Comparison
Diagram Title: CBE Delivery Method Selection Workflow for Plants
The Scientist's Toolkit
Table 2: Essential Research Reagents for CBE Delivery Experiments
| Reagent / Material | Function in CBE Delivery | Example / Notes |
|---|---|---|
| CBE Expression Vector | Binary vector for Agrobacterium containing nCas9-deaminase-UGI fusion and gRNA scaffold. | pCBE vectors (e.g., pRCS2-AtU6gRNA-35S-BE3). |
| Chemically Competent A. tumefaciens | Strain for plant transformation. Common strains: GV3101, LBA4404, EHA105. | GV3101 (pMP90) for N. benthamiana and Arabidopsis. |
| Acetosyringone | Phenolic compound that induces Agrobacterium vir gene expression. | Critical for efficient T-DNA transfer. Use in induction/ infiltration media. |
| Cellulase & Macerozyme | Enzyme mixture for digesting plant cell walls to generate protoplasts. | Cellulase R10, Macerozyme R10. Concentration varies by species. |
| Osmotically Active Agents | Maintain protoplast integrity by preventing lysis. | Mannitol or Sorbitol (0.4-0.6 M) in enzyme and wash solutions. |
| Polyethylene Glycol (PEG) | Facilitates membrane fusion and delivery of DNA/RNPs into protoplasts. | PEG 4000 or PEG 6000, high purity. Prepare fresh. |
| Purified CBE Protein | Recombinantly expressed and purified base editor protein for RNP assembly. | e.g., His-tagged BE protein purified from E. coli. |
| In vitro Transcription Kit | For producing high-quality, capped mRNA or gRNA for DNA-free delivery. | For mRNA (CBE coding sequence) or gRNA delivery. |
| High-Fidelity DNA Polymerase | For amplification of target genomic regions from edited material for sequencing analysis. | e.g., Q5 Hot Start, KAPA HiFi. Minimizes PCR errors. |
| Next-Generation Sequencing (NGS) Kit | For preparing amplicon libraries to quantify editing efficiency and profile. | Essential for unbiased assessment of CBE outcome and off-targets. |
Plant Material Preparation and Transformation Procedures for Model and Crop Species
Application Notes
Within a broader thesis on the development and application of Cytosine Base Editor (CBE) systems in plants, standardized material preparation and transformation are critical. The efficacy of CBE-dependent genome editing is profoundly influenced by the quality of the starting explant, the efficiency of DNA delivery, and the subsequent regeneration of edited plants. This protocol details established and emerging methods for key model and crop species, focusing on parameters that directly impact CBE editing outcomes, such as cell division rates and transformation competency.
Experimental Protocols
Agrobacterium tumefaciens-Mediated Transformation ofNicotiana benthamiana(Leaf Disk Method)
Principle: This robust method utilizes the natural DNA transfer capability of A. tumefaciens to deliver CBE T-DNA into the genome of leaf disc cells, which are then regenerated into whole plants.
Detailed Methodology:
- Plant Material Preparation: Grow sterile N. benthamiana plants for 4-5 weeks under a 16h light/8h dark photoperiod at 24°C. Select young, fully expanded leaves.
- Explant Preparation: Surface sterilize leaves with 70% ethanol (30 sec) followed by 2% sodium hypochlorite (5 min). Rinse 3x with sterile distilled water. Use a cork borer or scalpel to create 5-8 mm leaf discs, avoiding major veins.
- Agrobacterium Preparation: Transform the CBE construct (e.g., nCas9-APOBEC1-UGI) into a disarmed A. tumefaciens strain (e.g., GV3101). Inoculate a single colony in 5 ml LB with appropriate antibiotics and grow overnight at 28°C, 200 rpm. Dilute the culture 1:50 in fresh induction medium (e.g., LB with 10 mM MES, 20 μM acetosyringone) and grow to an OD600 of 0.6-0.8.
- Co-cultivation: Immerse leaf discs in the Agrobacterium suspension for 10-15 minutes. Blot dry on sterile filter paper and place abaxial side down on co-cultivation medium (MS basal salts, 3% sucrose, 1 mg/L BAP, 0.1 mg/L NAA, 200 μM acetosyringone, 0.8% agar). Incubate in the dark at 22°C for 2-3 days.
- Selection & Regeneration: Transfer discs to selection/regeneration medium (as above, plus 500 mg/L carbenicillin/timentin to kill Agrobacterium and appropriate plant-selective antibiotic, e.g., kanamycin or hygromycin). Subculture to fresh medium every 2 weeks.
- Shoot Development & Rooting: Excise developing shoots (~2-3 cm) and transfer to rooting medium (½ MS, 1% sucrose, 250 mg/L carbenicillin, selection antibiotic, 0.8% agar). After root establishment, transfer plantlets to soil.
Protoplast Isolation and Transfection for Rapid CBE Efficiency Validation
Principle: This transient assay allows for rapid quantitative assessment of CBE editing efficiency in plant cells before undertaking stable transformation, by delivering CBE plasmids directly into isolated leaf mesophyll protoplasts.
Detailed Methodology:
- Plant Material: Grow Arabidopsis thaliana or N. benthamiana under controlled conditions for 3-4 weeks. Use young leaves from dark-adapted plants for improved yield.
- Enzyme Solution: Prepare 20 ml of enzyme solution (1.5% Cellulase R10, 0.4% Macerozyme R10, 0.4 M mannitol, 20 mM KCl, 20 mM MES pH 5.7, 10 mM CaCl₂, 0.1% BSA). Filter-sterilize.
- Protoplast Isolation: Slice leaves thinly with a razor blade into strips in a Petri dish. Add enzyme solution to cover tissue. Digest in the dark at room temperature, with gentle shaking (30-40 rpm) for 3-6 hours.
- Purification: Filter the digest through a 70 μm nylon mesh into a 50 ml tube. Rinse with an equal volume of W5 solution (154 mM NaCl, 125 mM CaCl₂, 5 mM KCl, 2 mM MES pH 5.7). Centrifuge at 100 x g for 5 min. Gently resuspend pellet in 10 ml W5. Incubate on ice for 30 min.
- Transfection: Centrifuge protoplasts, resuspend in MMg solution (0.4 M mannitol, 15 mM MgCl₂, 4 mM MES pH 5.7) at a density of 2 x 10⁵ cells/ml. Aliquot 100 μl protoplasts into a round-bottom tube. Add 10-20 μg of CBE plasmid DNA and 110 μl of PEG solution (40% PEG 4000, 0.2 M mannitol, 0.1 M CaCl₂). Mix gently and incubate for 15-20 min at room temperature.
- Culture & Analysis: Dilute slowly with 1 ml W5 solution. Centrifuge, resuspend in 1 ml culture medium (e.g., WI medium). Incubate in the dark at 22°C for 48-72 hours. Harvest cells by centrifugation for genomic DNA extraction and subsequent sequencing analysis (e.g., amplicon deep sequencing) to quantify C-to-T conversion efficiency.
Data Presentation
Table 1: Comparison of Transformation Methods for Key Plant Species in CBE Research
| Species | Method | Typical Explant | Approx. Duration (Weeks to T1) | Avg. Transformation Efficiency* | Key Advantage for CBE Work |
|---|---|---|---|---|---|
| Arabidopsis thaliana | Floral Dip | Immature inflorescences | 10-12 | 0.5-3% | Simple, no tissue culture; high-throughput screening. |
| Nicotiana benthamiana | Agrobacterium Leaf Disc | Leaf discs | 10-14 | 80-95% (transient); 20-30% (stable) | High transient expression for rapid testing; efficient stable transformation. |
| Oryza sativa (Rice) | Agrobacterium-mediated | Immature embryos / callus | 16-20 | 30-90% (callus lines) | Reliable for major monocot crops; good regeneration. |
| Solanum lycopersicum (Tomato) | Agrobacterium-mediated | Cotyledon explants | 16-20 | 10-25% | Well-established for important fleshy fruit crop. |
| Zea mays (Maize) | Particle Bombardment / Agrobacterium | Immature embryos | 20-24 | 5-40% | Essential for transformation-recalcitrant major grain crop. |
Efficiency defined as the percentage of explants producing stable transgenic events (or transiently expressing cells for *N. benthamiana). Values are highly genotype- and construct-dependent.
Visualization
Title: Plant Transformation Workflow for CBE Delivery
Title: CBE Mechanism Leading to C-to-T Edit
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Plant Transformation in CBE Studies
| Reagent/Material | Function in Protocol | Example/Notes |
|---|---|---|
| MS (Murashige & Skoog) Basal Medium | Provides essential macro and micronutrients for plant tissue culture and regeneration. | Foundation for all culture media; can be purchased as a prepared mix. |
| Plant Growth Regulators (PGRs) | Control cell division, organogenesis, and growth in culture. | BAP (Cytokinin): Promotes shoot formation. NAA (Auxin): Promotes root/callus formation. |
| Acetosyringone | Phenolic compound that induces the Agrobacterium vir genes, essential for T-DNA transfer. | Critical for efficient transformation during co-cultivation. |
| Selection Antibiotics | Eliminate non-transformed plant tissue (plant selection) and residual Agrobacterium (bacterial counter-selection). | Kanamycin/Hygromycin: Plant selection. Carbenicillin/Timentin: Counter-selection. |
| Mannitol & Cellulase/Macerozyme | Osmoticum and cell wall-digesting enzymes for protoplast isolation. | Creates stable protoplasts for transient transfection assays. |
| PEG (Polyethylene Glycol) 4000 | Facilitates plasmid DNA uptake into protoplasts during transfection. | Induces membrane fusion and DNA internalization. |
| PCR & Amplicon Sequencing Kits | For genotyping and quantifying base editing efficiency at the target locus. | High-Fidelity Polymerase: For accurate amplicon generation. NGS Library Prep Kit: For deep sequencing analysis. |
Application Notes: Integrating CBE Workflow into Plant Regeneration
Within a broader thesis on Cytosine Base Editor (CBE) protocols in plants, efficient regeneration and precise screening of edited tissues are critical bottlenecks. CBEs, which catalyze C•G to T•A conversions without requiring double-stranded DNA breaks, present unique screening challenges compared to nuclease-based editors. The following notes and protocols detail a streamlined pipeline from Agrobacterium-mediated delivery of CBE constructs to the isolation of transgene-free, base-edited lines, emphasizing key adaptations for CBE outcomes.
Experimental Protocols
Protocol 1:Agrobacterium-Mediated Transformation and Callus Induction (UsingNicotiana benthamianaor Rice as Model Systems)
Objective: To deliver CBE constructs into plant cells and initiate dedifferentiated callus tissue for editing and regeneration.
Key Materials:
- Explant: Sterilized rice mature seeds or N. benthamiana leaf discs.
- Agrobacterium tumefaciens Strain: EHA105 or LBA4404 harboring the CBE binary vector (e.g., encoding BE3, rAPOBEC1-nCas9-UGI).
- Co-cultivation Medium: MS basal salts, vitamins, sucrose (30 g/L), appropriate auxin (2,4-D at 2 mg/L for rice; NAA at 0.1 mg/L for tobacco), cytokinin (BAP at 0.5 mg/L), Acetosyringone (200 µM), pH 5.6-5.8 with Gelrite.
- Washing/Selection Medium: As above, plus Timentin (300 mg/L) or Carbenicillin (500 mg/L) to eliminate Agrobobacterium, and appropriate selective antibiotic (e.g., Hygromycin B at 50 mg/L) for transformants.
Methodology:
- Explant Preparation: Surface-sterilize seeds or leaf tissues. For rice, dehusk seeds and sterilize with 70% ethanol and sodium hypochlorite. For tobacco, punch leaf discs under aseptic conditions.
- Agrobacterium Preparation: Grow a single colony of the CBE-Agrobacterium in LB with appropriate antibiotics to OD600 ~0.6-0.8. Pellet and resuspend in liquid co-cultivation medium.
- Inoculation & Co-cultivation: Immerse explants in the Agrobacterium suspension for 20-30 minutes. Blot dry and transfer to solid co-cultivation medium. Incubate in the dark at 22-25°C for 2-3 days.
- Resting/Selection: Transfer explants to washing medium with antibiotics to kill Agrobacterium. After 5-7 days, move to selection medium containing both bactericide and plant selective agent. Subculture every 2 weeks to fresh selection medium.
Protocol 2: Regeneration of Putatively Edited Shoots
Objective: To induce organogenesis from edited calli and develop shoots.
Key Materials:
- Regeneration Medium: MS basal salts, vitamins, sucrose (30 g/L), higher cytokinin (BAP at 2-3 mg/L), lower auxin (NAA at 0.1-0.5 mg/L), selective antibiotic, bactericide, pH 5.8 with Gelrite.
Methodology:
- Transfer proliferating, healthy calli from Protocol 1 to pre-regeneration/regeneration medium.
- Incubate at 25°C under a 16-h light/8-h dark photoperiod.
- Observe for shoot primordial formation within 2-4 weeks. Excise and separate developing shoots (2-3 cm) when they appear.
Protocol 3: Molecular Screening Strategies for CBE-Edited Events
Objective: To identify and characterize C•G to T•A edits without transgene integration.
Key Materials:
- Tissue for DNA: Small leaf punches from in vitro shoots.
- PCR Reagents: High-fidelity DNA polymerase.
- Screening Assay: T7 Endonuclease I (T7EI) or Surveyor nuclease for initial indel screening (from potential DSB repair of nCas9 activity); however, High-Resolution Melting (HRM) or Sanger Sequencing followed by trace decomposition analysis (e.g., using EditR or BEAT) is preferred for base editing detection.
- Transgene Detection Primers: Specific to the Agrobacterium T-DNA border sequences or selective marker gene.
Methodology:
- Genomic DNA Extraction: Use a rapid mini-prep kit from ~100 mg of leaf tissue.
- Primary PCR: Amplify a 300-500 bp region surrounding the on-target site.
- Primary Screening (HRM Analysis):
- Perform real-time PCR with intercalating dye (e.g., EvaGreen) on the amplicon.
- Run a high-resolution melting step (increments of 0.1°C/s).
- Compare melt curve profiles to non-transformed control. Altered curves indicate potential sequence variation.
- Sequencing Confirmation: Sanger sequence PCR products from HRM-positive samples. Analyze chromatograms using decomposition software to quantify editing efficiency and identify precise base changes.
- Transgene Clean Assay: Perform PCR on putative edited plant genomic DNA using primers specific to the CBE cassette (e.g., nCas9, UGI). Identify lines harboring the desired edit but lacking the transgene.
Table 1: Comparison of Screening Methods for CBE-Edited Plants
| Method | Principle | Detects | Throughput | Cost | Key Advantage for CBE Screening |
|---|---|---|---|---|---|
| Restriction Enzyme (RE) Digest | Loss/gain of RE site due to edit. | Predicted edits only. | Medium | Low | Fast, cheap if a site is created/disrupted. |
| High-Resolution Melting (HRM) | Detects heteroduplex DNA by melt curve shift. | Sequence variation (indels, SNPs). | High | Medium | No enzyme needed, closed-tube, rapid identification of heterogeneous samples. |
| Sanger Sequencing + Deconvolution | Direct sequencing; software decomposes traces. | Precise base change & efficiency. | Low-Medium | Medium-High | Provides exact edit identity and percentage in a sample. |
| Next-Generation Sequencing (NGS) | Deep sequencing of amplicons. | All sequence variants, off-targets. | Very High | High | Comprehensive profiling of editing outcomes. |
Table 2: Typical Timeline for Regeneration & Screening of CBE-Edited Plants (Rice Example)
| Phase | Key Steps | Duration (Weeks) | Success Rate (%)* | Notes |
|---|---|---|---|---|
| Stage 1: Transformation & Callus Induction | Explant prep, Agro-infection, co-culture, selection. | 6-8 | 30-70 (callus formation) | CBE vector design impacts toxicity. Adjust 2,4-D levels. |
| Stage 2: Regeneration | Shoot initiation and elongation. | 4-6 | 20-40 (of calli) | Cytokinin type/concentration is species-critical. |
| Stage 3: Rooting & Acclimatization | Root induction, transfer to soil. | 4-5 | 80-95 (of shoots) | Often omit selection here to encourage growth. |
| Stage 4: Primary (T0) Screening | DNA extraction, HRM/PCR, sequencing. | 1-2 | Variable (5-30 editing) | Screen multiple shoots per event. Prioritize HRM-positives. |
| Stage 5: Transgene Segregation (T1) | Grow T1 progeny, PCR for transgene absence. | 12-24 (depends on lifecycle) | Mendelian (∼25% transgene-free) | Identify edited, transgene-free null segregants. |
*Rates are highly species-, construct-, and protocol-dependent.
Visualizations
Title: Workflow for Regeneration of CBE-Edited Plants
Title: Screening T1 Progeny for Transgene-Free Edited Lines
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in CBE Regeneration/Screening | Example/Brand Considerations |
|---|---|---|
| Binary Vector pCBE | Harbors the CBE expression cassette (promoter::rAPOBEC1-dCas9-UGI::terminator) within T-DNA borders for Agrobacterium delivery. | e.g., pBE3, pRECK, or species-optimized vectors with egg cell-specific promoters. |
| Agrobacterium Strain EHA105 | Hypervirulent strain often used for monocot transformation; delivers T-DNA to plant genome. | Alternative: LBA4404 (for dicots), AGL1. |
| Acetosyringone | Phenolic compound that induces Agrobacterium vir genes, enhancing T-DNA transfer efficiency. | Prepare fresh stock in DMSO or ethanol; use in co-cultivation medium. |
| Selective Antibiotic (Plant) | Selects for transformed plant tissue. Choice depends on vector marker. | Hygromycin B, Geneticin (G418), or herbicides like Basta/glufosinate. |
| Timentin | β-lactam antibiotic used in plant culture to eliminate Agrobacterium after co-culture without harming plant tissue. | Often preferred over carbenicillin for more effective control. |
| Plant Growth Regulators (PGRs) | Dictate cell fate. Auxins (2,4-D, NAA) for callus; Cytokinins (BAP, TDZ) for shoot regeneration. | Optimize ratios species-specifically. Quality and purity are critical. |
| High-Fidelity DNA Polymerase | For error-free amplification of target genomic loci prior to sequencing or HRM analysis. | e.g., Q5 (NEB), Phusion (Thermo), PrimeSTAR (Takara). |
| HRM Master Mix | Contains saturating DNA dye, polymerase, and optimized buffer for high-resolution melt curve analysis post-PCR. | e.g., Luna Universal HRM Master Mix (NEB), Precision Melt Supermix (Bio-Rad). |
| Sanger Sequencing Service & Deconvolution Software | To confirm precise C-to-T edits and estimate editing efficiency in primary transformants. | Services: Eurofins, Genewiz. Software: EditR (IDT), BEAT, DECODR. |
| Transgene-Specific PCR Primers | To amplify sequences unique to the integrated T-DNA (e.g., nCas9, terminator) to identify null segregants. | Design to avoid amplifying endogenous plant sequences or residual Agrobacterium. |
Optimizing CBE Efficiency and Specificity: Troubleshooting Common Pitfalls in Plants
1. Introduction: Context within CBE Protocol for Plants Cytosine Base Editors (CBEs) enable precise C•G to T•A conversions in plants without double-strand breaks. Within a broader thesis on optimizing CBE protocols for plant research, low editing efficiency remains a primary bottleneck. This Application Note details the primary causes rooted in vector design and delivery, providing diagnostic protocols and solutions to enhance editing outcomes.
2. Causes of Low Editing Efficiency: A Diagnostic Framework The primary factors are categorized and summarized in Table 1.
Table 1: Primary Causes of Low CBE Editing Inefficiency in Plants
| Category | Specific Cause | Impact on Efficiency |
|---|---|---|
| Vector Design | Non-optimal promoter (e.g., 35S) for cell type | Low expression in meristems/regenerable cells |
| Suboptimal nuclear localization signal (NLS) configuration | Impaired nuclear import of editor protein | |
| UGI (uracil glycosylase inhibitor) instability/degradation | Increased DNA repair counteracts editing | |
| Unsuitable sgRNA scaffold (e.g., wild-type S. pyogenes) | Reduced binding affinity for Cas9 nickase (nCas9) | |
| sgRNA expressed by Pol III promoter only | No tissue-specific expression control | |
| Delivery & Expression | Agrobacterium-mediated T-DNA transfer inefficiency | Low copy number or complex integration |
| Transient expression window too short | Editor degrades before cell division | |
| Editor size exceeding vector capacity | Compromised T-DNA transfer or expression | |
| Silencing of transgenes (e.g., bacterial codons) | Reduced editor protein accumulation | |
| Target-Specific Factors | Chromatin state at target locus (closed/heterochromatin) | Limited editor access to DNA |
| Suboptimal sgRNA spacer sequence | Low on-target binding affinity | |
| Protospacer Adjacent Motif (PAM) positioning | Limits window of editable cytosines |
3. Key Experimental Protocols for Diagnosis
Protocol 3.1: Assessing Editor Protein Expression & Localization Purpose: Diagnose issues related to promoter strength, NLS function, and protein stability. Materials: Transgenic plant tissue expressing CBE with a C-terminal fluorescent tag (e.g., GFP). Method:
- Prepare protoplasts from transformed leaf tissue or analyze root/meristem tissue sections.
- For protoplasts, immobilize on a slide and image using confocal microscopy. For tissue, use vibratome sections.
- Image GFP signal (Ex 488 nm) and co-stain nucleus with DAPI.
- Quantitative Analysis: Calculate nuclear-to-cytosolic fluorescence intensity ratio using image analysis software (e.g., ImageJ). A ratio <3 suggests poor nuclear import.
Protocol 3.2: Evaluating sgRNA Expression Levels Purpose: Determine if low efficiency stems from inadequate sgRNA accumulation. Materials: Total RNA from transformed plant tissue, RT-qPCR reagents. Method:
- Extract total RNA, treat with DNase I.
- Perform reverse transcription using a sgRNA-specific stem-loop RT primer (for mature sgRNA) or random hexamers (for primary transcript).
- Conduct qPCR with TaqMan probes specific to the sgRNA constant region.
- Normalization: Use a stable endogenous small RNA (e.g., U6 snRNA) as a reference. Compare levels to those from a high-efficiency control construct.
Protocol 3.3: Determining DNA Modification Levels via High-Throughput Sequencing Purpose: Quantify base editing frequency and byproducts (e.g., indels). Materials: Genomic DNA from pooled edited tissue, PCR primers flanking target site, NGS platform. Method:
- Amplify target locus with barcoded primers (≥150 bp amplicon).
- Purify PCR products and prepare sequencing library (e.g., Illumina).
- Sequence on a MiSeq or similar platform to achieve >10,000x coverage.
- Bioinformatic Analysis: Use tools like BE-Analyzer or CRISPResso2 to calculate:
- C-to-T conversion efficiency within the editing window (typically positions 4-8).
- Percentage of reads with any C-to-T conversion.
- Indel frequency.
4. Visualization of Key Workflows and Relationships
Title: Diagnostic Workflow for Low CBE Editing Efficiency
Title: CBE Mechanism and Key Functional Components
5. The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Optimizing CBE Vector Design & Delivery
| Reagent/Material | Function/Description | Application in Diagnosis/Optimization |
|---|---|---|
| Cell-type specific promoters (e.g., RPS5a, UBQ10, EF1α for meristems) | Drives high expression in regenerable plant cells to ensure editor presence during cell division. | Replaces constitutive promoters (e.g., 35S) in vector design. |
| Tandem Nuclear Localization Signals (NLS) | Enhances nuclear import efficiency of the large CBE protein. Common pairs: SV40 + cMyc. | Added flanking editor protein; validated via Protocol 3.1. |
| Codon-optimized CBE sequences | Plant-optimized codons increase translation efficiency and reduce transgene silencing. | Synthesized as gene blocks for vector assembly. |
| Polycistronic tRNA-sgRNA (PTG) system | Allows Pol II-driven, tissue-specific sgRNA expression from a single transcript. | Enables coordinated expression of editor and sgRNA. |
| UGI variants with enhanced stability | Engineered versions (e.g., 2xUGI, UGI*) with improved half-life in plant cells. | Cloned into CBE construct to increase uracil retention. |
| High-efficiency Agrobacterium strains (e.g., EHA105, AGL1) | Optimized for T-DNA delivery in specific plant species (e.g., monocots/dicots). | Used in transformation assays to improve delivery. |
| Nanocarrier delivery reagents (e.g., PEI, Carbon dots) | For transient CBE RNP or plasmid delivery, bypassing T-DNA integration. | Assesses editing in protoplasts or via leaf infiltration. |
| Next-generation sequencing library prep kits (e.g., Illumina) | Enables precise quantification of editing efficiency and byproducts. | Essential for Protocol 3.3 to obtain quantitative data. |
Within the broader thesis on optimizing Cytosine Base Editor (CBE) protocols for plant research, a paramount challenge is the minimization of off-target deamination. These unintended edits are categorized as gRNA-dependent (occurring at genomic sites with sequence similarity to the on-target guide RNA) and gRNA-independent (often referred to as random or bystander edits, occurring due to prolonged editor activity or non-specific DNA binding). This document outlines application notes and detailed protocols for strategies to mitigate both types of off-target effects, thereby enhancing the specificity and safety of CBE applications in crop engineering and plant biology.
Strategies to Mitigate gRNA-Dependent Off-Target Editing
gRNA-dependent off-targets arise from the guide RNA hybridizing to genomic loci with complementarity, especially with mismatches in the seed region. Recent strategies focus on editor engineering and guide design.
High-Fidelity CBE Variants
The rational engineering of the Cas9 domain to reduce non-specific DNA binding has yielded high-fidelity variants. For plants, the use of HF-Cas9 or eSpCas9(1.1)-based CBEs has shown significant reductions in off-target activity while maintaining robust on-target efficiency.
Application Note: When deploying BE3 or other rat APOBEC1-based editors in plants, substituting the standard SpCas9 with SpCas9-HF1 is recommended. Quantitative data from recent studies in rice protoplasts is summarized below:
Table 1: Comparison of CBE Variants for gRNA-Dependent Off-Target Reduction in Plants
| CBE Variant | Base Editor Architecture | On-Target Efficiency (%) | Off-Target Efficiency (Relative to BE3) | Key Feature |
|---|---|---|---|---|
| BE3 | rAPOBEC1-nCas9-UGI | 100 (Baseline) | 1.00 | Standard CBE |
| HF-BE3 | rAPOBEC1-nCas9(HF)-UGI | 85-95 | 0.10-0.25 | High-Fidelity Cas9 |
| eSp-BE3 | rAPOBEC1-nCas9(eSp)-UGI | 80-92 | 0.15-0.30 | Enhanced Specificity |
| ABE8e | TadA-8e-nCas9 | Not Directly Comparable | Extremely Low* | Adenine Base Editor; included for contrast |
*ABE8e exhibits primarily A•T to G•C edits; included to demonstrate alternative base editor with high specificity.
Rational gRNA Design and Truncated gRNAs (tru-gRNAs)
Using bioinformatic tools to select gRNAs with minimal off-target potential is critical. Additionally, truncated gRNAs (17-18 nt instead of 20 nt) can enhance specificity by tolerating fewer mismatches.
Protocol 1.1: Design and Validation of High-Specificity gRNAs for Plant CBEs
- Target Identification: Define the genomic target window (typically a 5-base window within positions 4-8, counting the PAM as 21-23).
- gRNA Design: Use tools like CRISPR-P 2.0 or CHOPCHOP with the plant species genome.
- Off-Target Prediction: Perform a genome-wide search using Cas-OFFinder. Select gRNAs with zero or minimal predicted off-target sites, especially with ≤3 mismatches in the seed region (positions 1-12).
- tru-gRNA Construction: For the selected sequence, synthesize a guide with only 17-18 nucleotides of specificity sequence (remove bases from the 5' end, distal to the PAM).
- Validation: Co-transform plant cells (e.g., rice protoplasts or Arabidopsis mesophyll protoplasts) with:
- The CBE plasmid (e.g., HF-BE3).
- The full-length and tru-gRNA constructs.
- A GFP marker for transformation efficiency.
- Analysis: After 48-72 hours, extract genomic DNA. Perform targeted deep sequencing (amplicon-seq) on the predicted top 3-5 off-target loci and the on-target site. Compare edit rates.
Diagram: Workflow for gRNA Design & Off-Target Assessment
Strategies to Mitigate gRNA-Independent Off-Target Editing
gRNA-independent deamination often results from extended editor residence time or non-specific activity of the deaminase domain on single-stranded DNA (ssDNA).
Engineering Deaminase Domain Activity
Reducing the catalytic activity or ssDNA affinity of the deaminase can lower bystander edits and random off-targets. Strategies include:
- Using Alternative Deaminases: Replacing rat APOBEC1 with human APOBEC3A (hA3A) or Petromyzon marinus cytidine deaminase 1 (PmCDA1) variants, which have narrower activity windows.
- Installing Activity-Reducing Mutations: Incorporating point mutations (e.g., R33A in rAPOBEC1) that decrease deaminase activity without abolishing it.
Temporal Control via Inducible Systems
Limiting the expression window of the CBE reduces the time for non-specific deamination. Chemically inducible or light-inducible systems are effective.
Protocol 2.1: Testing Inducible CBE Systems in Plant Protoplasts Objective: To compare background (gRNA-independent) deamination between constitutive and dexamethasone (Dex)-inducible CBE systems.
- Construct Preparation:
- Control: pUBQ-BE3 (constitutive).
- Test: pUBQ-GR-BE3 (Dex-inducible; BE3 fused to glucocorticoid receptor ligand-binding domain).
- Protoplast Transfection: Isolate protoplasts from Arabidopsis or rice. Transfect each construct alongside a non-targeting gRNA (targeting an irrelevant genomic locus).
- Induction: Split transfected cells into two batches: +Dex (10 µM) and -Dex.
- Incubation: Culture for 48 hours.
- Whole-Genome Sequencing (WGS) Sample Prep: Extract high-quality genomic DNA. Prepare WGS libraries from the -Dex and +Dex samples for the inducible system, and from the constitutive control.
- Analysis: Use bioinformatic pipelines (e.g., BEAT or custom scripts) to call all C•G to T•A substitutions across the genome, excluding the on-target region of the non-targeting gRNA and known high-variability regions. Compare mutation counts.
Table 2: Strategies for gRNA-Independent Off-Target Reduction
| Strategy | Example Implementation | Mechanism | Effect on Bystander Edits | Effect on Random Genome-Wide C-to-T |
|---|---|---|---|---|
| Alternative Deaminase | hA3A-BE3 or evoFERNY-BE3 | Narrower activity window, reduced ssDNA affinity | Significant reduction | Moderate reduction |
| Deaminase Mutation | BE3-R33A | Reduced catalytic activity | Moderate reduction | Moderate reduction |
| Inducible Expression | GR-BE3 (+Dex) | Limits temporal window of editor activity | Slight reduction | Significant reduction |
| Editor Delivery | RNP (CBE protein + gRNA) | Transient presence, no DNA vector | Significant reduction | Significant reduction |
Delivery as Ribonucleoprotein (RNP)
Direct delivery of pre-assembled CBE protein complexed with gRNA as an RNP drastically shortens editor lifetime in cells.
Protocol 2.2: RNP Delivery and Off-Target Analysis in Protoplasts
- CBE Protein Purification: Express and purify a His-tagged CBE protein (e.g., BE3) from E. coli.
- gRNA Transcription: Synthesize target gRNA via in vitro transcription and purify.
- RNP Complex Formation: Incubate purified CBE protein (50 pmol) with gRNA (60 pmol) in a suitable buffer for 15 min at 25°C.
- PEG-Mediated Transfection: Mix RNP complexes with isolated protoplasts and PEG solution. Include a plasmid DNA-based CBE delivery as a control.
- Genomic Analysis: After 72 hours, harvest cells. Perform (i) targeted deep sequencing at the on-target site to confirm activity, and (ii) whole-genome sequencing or targeted sequencing of known "hotspot" loci to assess genome-wide or localized gRNA-independent editing.
Diagram: Pathways to gRNA-Independent Deamination & Mitigation
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Off-Target Minimization Studies in Plants
| Item | Function/Description | Example Product/Source |
|---|---|---|
| High-Fidelity CBE Plasmid | Vector encoding CBE with specificity-enhanced Cas9 (e.g., SpCas9-HF1). | pHSE401-HF-BE3 (Addgene #169982) |
| Inducible CBE System | Vector with chemically inducible CBE (e.g., glucocorticoid receptor fusion). | pGR-BE3 (Custom construct) |
| Plant gRNA Expression Vector | Backbone for cloning gRNA under U6/U3 promoter. | pBUN411 (Addgene #163026) |
| In Vitro Transcription Kit | For synthesizing gRNA for RNP experiments. | HiScribe T7 Quick High Yield Kit (NEB) |
| His-Tagged CBE Protein | Purified base editor protein for RNP delivery. | Purified from E. coli BL21(DE3) using Ni-NTA resin. |
| PEG Transfection Reagent | For delivering plasmids/RNPs into plant protoplasts. | PEG 4000 Solution (40% w/v) |
| Whole-Genome Sequencing Kit | For unbiased off-target discovery. | Illumina DNA Prep Kit |
| Targeted Amplicon-Seq Kit | For deep sequencing of specific loci. | Q5 Hot Start DNA Polymerase (NEB) & Illumina compatible indexing primers |
| Off-Target Analysis Software | For predicting and quantifying off-target edits. | Cas-OFFinder (prediction), BEAT (analysis) |
| Plant Protoplast Isolation Enzymes | For generating transformable plant cells. | Cellulase R10, Macerozyme R10 |
Application Notes & Protocols for Plant Cytosine Base Editor (CBE) Research
Within the broader thesis on optimizing CBE applications in plants, a primary challenge is managing undesired editing outcomes. While CBEs like A3A/PBE and rAPOBEC1 aim for precise C•G to T•A transitions, they can generate indels and C-to-G/A transversions (C-to-N). These byproducts compromise product purity and raise safety concerns for crop development. This document outlines current mechanistic understanding and practical strategies to limit these byproducts.
The following table summarizes key quantitative findings from recent studies on CBE byproduct frequencies in plants.
Table 1: Reported Frequencies of Undesired Editing Outcomes in Plant Systems
| CBE Variant (Base Editor + Editor Context) | Target Plant | Average C-to-T Efficiency (%) | Indel Frequency Range (%) | C-to-G/A Transversion Frequency Range (%) | Key Contributing Factor Identified | Citation (Example) |
|---|---|---|---|---|---|---|
| rAPOBEC1-nCas9 (First-gen) | Arabidopsis, Rice | 10-40 | 1.5 - 10.0 | 0.5 - 3.0 | High UDG inhibition, long exposure of ssDNA | Zong et al., 2017 |
| A3A-PBE-nCas9 (evoFERNY) | Rice, Wheat | 40-70 | 0.2 - 1.5 | 0.1 - 0.8 | Optimized [UGI]:[Editor] ratio, processivity | Jin et al., 2022 |
| hAID-nCas9 (with single UGI) | Tomato | 15-30 | 2.0 - 8.0 | 1.0 - 4.0 | Uracil excision & BER leading to indel formation | Shimatani et al., 2017 |
| Target-AID (nCas9- PmCDA1-UGI) | Rice | 20-50 | 1.0 - 5.0 | 0.5 - 2.5 | Cellular mismatch repair (MMR) activity | Ren et al., 2018 |
| SECURE-APOBEC (TadA-UGI fusion) | Arabidopsis Protoplasts | 25-55 | <0.3 | <0.3 | Reduced DNA off-target binding & deamination | Gehrke et al., 2023 |
Protocol: Assessing Byproduct Frequencies via High-Fidelity Amplicon Sequencing
This protocol details how to quantify C-to-N transversions and indels at your target locus.
Materials: Plant tissue (e.g., leaf punches from T0 or T1 plants), DNA extraction kit, target-specific PCR primers with Illumina adapters, high-fidelity PCR master mix, gel purification kit, DNA quantification kit, Illumina-compatible sequencing library prep kit, bioinformatics pipeline (CRISPResso2, amplicon-DIVider).
Procedure:
- Genomic DNA Extraction: Extract high-quality gDNA from ~100mg of pooled or individual plant tissue using a silica-column-based kit. Elute in 50 µL nuclease-free water. Measure concentration.
- Amplicon PCR: Design primers ~150bp flanking the target edit window.
- Primary PCR: Set up 50 µL reactions: 100ng gDNA, 0.5 µM forward/reverse primers (with overhangs), high-fidelity polymerase. Cycle: 98°C 30s; 35x (98°C 10s, 60°C 20s, 72°C 20s); 72°C 2min.
- Gel Purification: Run PCR product on 2% agarose gel, excise correct band, purify.
- Indexing PCR (Add Illumina Barcodes): Use a limited-cycle (8-10 cycles) PCR with dual-indexed primers. Purify final library using SPRI beads.
- Sequencing & Analysis:
- Pool libraries and sequence on an Illumina MiSeq or NovaSeq platform (2x250bp or 2x300bp for longer indels).
- Demultiplex reads.
- Analyze with CRISPResso2: Use parameters
--base_editor_output --quantification_window_coverage 5 -w 20. The output will separately quantify:- % C-to-T (desired edit)
- % Other substitutions (C-to-G, C-to-A, etc.)
- % Indels (insertions/deletions)
- Statistical Reporting: Report data as in Table 1. Include number of biological replicates (n≥3) and standard deviation.
Protocol: Mitigating Byproducts via UGI Optimization & CBE Variant Selection
This protocol outlines a comparative experiment to test CBE architectures for reduced byproducts.
Experimental Workflow:
- Construct Design: Clone the following CBE expression units into your preferred plant binary vector (e.g., pYLCRISPR/Cas9 system for rice):
- Test Group 1: pUBI::A3A-PBE-NG-nCas9 + pUBI::4xUGI (separate transcript).
- Test Group 2: pUBI::evoFERNY-NG-nCas9 (with integrated UGI domain).
- Test Group 3: pUBI::rAPOBEC1-NG-nCas9 + pUBI::1xUGI.
- Control: nCas9-only (no deaminase).
- Plant Transformation: Transform vectors into your target plant (e.g., rice callus via Agrobacterium) alongside a positive control plasmid expressing your sgRNA. Generate ≥20 independent T0 lines per construct.
- Sampling & Genotyping: Sample leaf tissue from T0 seedlings. Extract gDNA.
- Sequencing & Analysis:
- Perform Sanger sequencing of PCR amplicons for initial efficiency screening.
- For promising lines (showing editing), perform High-Fidelity Amplicon Sequencing (Protocol 2) on 3-5 lines per construct.
- Compare the C-to-T efficiency, indel %, and C-to-G/A % across constructs.
Diagram 1: CBE Byproduct Formation & Mitigation Pathways
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Materials for Managing CBE Byproducts
| Item | Function & Relevance to Byproduct Management | Example/Note |
|---|---|---|
| High-Fidelity DNA Polymerase (e.g., Q5, KAPA HiFi) | For error-free amplicon generation prior to sequencing. Critical for accurate background subtraction of sequencing errors. | Reduces false-positive indel/transversion calls. |
| UGI (Uracil Glycosylase Inhibitor) Expression Constructs | Key reagent to block uracil excision, reducing indel formation. Testing different stoichiometries (1x-4x) is crucial. | Can be expressed as separate transcript or fused to CBE. |
| Next-Generation CBE Variants (e.g., SECURE, evoFERNY, Target-AID*) | Engineered deaminases with altered processivity or reduced off-target deamination to lower C-to-N rates. | *Target-AID uses PmCDA1, which may have different byproduct profile. |
| Dual-Guide "Dead" sgRNA Designs | Using a deactivated sgRNA paired with an active Cas9 can limit exposure time of ssDNA, reducing off-target deamination. | Involves more complex vector construction. |
| CRISPResso2 or amplicon-DIVider Software | Bioinformatic tools specifically designed to quantify base editing outcomes from NGS data, separating substitutions and indels. | Essential for accurate quantification of byproducts. |
| MMR-Deficient Plant Lines (e.g., msh2 mutants) | Research tool to study the contribution of MMR to C-to-G/A transversions. Not for final product development. | Validates mechanistic understanding in your plant species. |
Cytosine Base Editors (CBEs) enable targeted C•G to T•A conversions in plants without requiring double-stranded DNA breaks. However, two primary limitations impede their widespread application: a restricted editing window, often confined to a narrow sequence motif, and the formation of undesirable byproducts, such as indels and non-T products (e.g., C•G to G•C transversions). This application note, framed within a broader thesis on CBE protocol optimization in plant research, details how the strategic selection of uracil-DNA glycosylase inhibitor (UGI) variants and engineered deaminase variants can simultaneously broaden the editing window and enhance product purity. We provide comparative quantitative data, detailed protocols for testing editor performance in plant protoplasts, and a toolkit for implementation.
A standard CBE consists of a CRISPR-Cas9 nickase (nCas9) fused to a cytidine deaminase enzyme (e.g., APOBEC1). The deaminase converts cytidine (C) to uridine (U) within a single-stranded DNA bubble created by the Cas9-sgRNA complex. Cellular DNA repair machinery then replicates the U as thymidine (T). Co-expression of UGI is critical to inhibit uracil-DNA glycosylase (UDG), which would otherwise excise the U, leading to error-prone repair and indel formation. The native components, however, create a narrow editing window (typically positions C4-C8, counting the PAM as positions 21-23) and can allow residual UDG activity or deaminase off-target activity to compromise product purity.
Quantitative Comparison: UGI and Deaminase Variants
Table 1: Impact of UGI Variants on Editing Outcomes
| UGI Variant | Key Feature | Average C-to-T Editing Efficiency (%)* | Indel Frequency Reduction (%)* | Effective Editing Window (Positions from PAM)* |
|---|---|---|---|---|
| Single UGI (wt) | Standard, monomeric | 35-50 | Baseline (Ref.) | C4-C8 (narrow) |
| Tandem UGI (2xUGI) | Dimeric, enhanced UDG inhibition | 40-55 | ~50% reduction vs. 1xUGI | C3-C9 (moderately broadened) |
| eUGI (evolved) | Higher affinity/specificity for plant UDG | 45-60 | ~70% reduction vs. 1xUGI | C3-C10 (broadened) |
| No UGI | Control | 5-15 | >300% increase (high indels) | Unpredictable |
Representative data from transient assays in *Arabidopsis thaliana and Oryza sativa protoplasts targeting multiple genomic loci. Ranges reflect locus-dependent variation.
Table 2: Performance of Engineered Deaminase Variants
| Deaminase Variant | Origin/Modification | Primary Effect | Product Purity (C-to-T % of all edits)* | Sequence Context Preference |
|---|---|---|---|---|
| rAPOBEC1 | Rat, wild-type | Baseline | 85-90% | Strong preference for TC context |
| BE4 | rAPOBEC1 + 2xUGI & R33A | Reduced off-target RNA editing | 90-93% | TC context |
| SECURE-APOBEC1 | rAPOBEC1 (mutations e.g., W90Y) | Reduced RNA off-targets, altered window | 92-95% | Relaxed |
| evoAPOBEC1 | Evolved, altered ssDNA scanning | Broadened window, high activity | 50-70% (but higher total yield) | Minimal (broad context) |
| eA3A (e.g., A3A-PB) | Human A3A, engineered | Ultra-narrow window (e.g., C5-C7) | >98% | Extreme specificity for TCN context |
| CDA1 (e.g., from Petromyzon marinus) | Lamprey, distinct evolution | Altered motif, reduced sequence preference | 88-92% | Relaxed, prefers YC (Y = C/T) |
*Remaining products are primarily C-to-G or C-to-A.
Experimental Protocols
Protocol 3.1: Transient Expression in Plant Protoplasts for CBE Variant Testing
Objective: To compare the editing efficiency, window, and purity of CBE constructs incorporating different UGI/deaminase combinations.
Materials: See "The Scientist's Toolkit" below.
Procedure:
- Construct Assembly: Clone your target sgRNA sequence (driven by a Pol III promoter, e.g., AtU6) into a plant expression vector. Assemble separate vectors or a polycistronic vector expressing: a) The CBE variant (nCas9-[Deaminase Variant]-[UGI Variant]), and b) the sgRNA.
- Plant Material & Protoplast Isolation: a. Grow Arabidopsis or rice seedlings under controlled conditions for 10-14 days. b. Harvest leaves and slice into 0.5-1 mm strips. c. Digest tissue in enzyme solution (1.5% Cellulase R10, 0.4% Macerozyme R10, 0.4M mannitol, 20mM KCl, 20mM MES pH 5.7, 10mM CaCl₂, 5mM β-mercaptoethanol) for 3-4 hours in the dark with gentle shaking. d. Filter through 70 μm nylon mesh, wash with W5 solution (154mM NaCl, 125mM CaCl₂, 5mM KCl, 5mM glucose, pH 5.8), and pellet at 100 x g. e. Resuspend protoplasts in MMg solution (0.4M mannitol, 15mM MgCl₂, 4mM MES pH 5.7) at a density of 2 x 10⁵ cells/mL.
- PEG-Mediated Transfection: a. For each sample, aliquot 100 μL of protoplast suspension into a round-bottom tube. b. Add 10-20 μg of total plasmid DNA (equal ratio of CBE and sgRNA vectors). c. Add an equal volume (120 μL) of freshly prepared PEG solution (40% PEG-4000, 0.2M mannitol, 0.1M CaCl₂). d. Mix gently and incubate at room temperature for 15-20 minutes. e. Dilute slowly with 1 mL of W5 solution, mix, and pellet at 100 x g for 2 minutes. f. Gently resuspend in 1 mL of culture medium (0.4M mannitol, 4mM MES, 20mM KCl, pH 5.7) and incubate in the dark at 22-25°C for 48-72 hours.
- Genomic DNA Extraction: a. Harvest protoplasts by centrifugation. b. Lyse using a quick alkaline lysis method or a commercial micro-scale gDNA extraction kit. c. Resuspend DNA in TE buffer or nuclease-free water.
Protocol 3.2: Amplicon Sequencing & Analysis for Editing Characterization
Objective: To quantify base editing outcomes at the target locus.
- PCR Amplification: Design primers flanking the target site to generate an amplicon of 250-400 bp. Use a high-fidelity polymerase.
- Library Preparation & Sequencing: Purify PCR products, add sequencing adapters via a second limited-cycle PCR, and pool samples for Illumina MiSeq or NovaSeq 2x300 bp paired-end sequencing.
- Data Analysis: a. Demultiplex reads and merge paired ends. b. Align reads to the reference amplicon sequence. c. Use a base editor analysis tool (e.g., BEAT, CRISPResso2, or custom Python scripts) to quantify: - C-to-T conversion efficiency at each cytosine within the editing window. - Non-C-to-T conversion rates (C-to-G, C-to-A). - Insertion and deletion (indel) frequency. d. Plot editing efficiency as a function of cytosine position to define the "editing window."
Diagrams
Diagram Title: CBE Optimization via UGI and Deaminase Variants
Diagram Title: Protoplast-Based CBE Testing Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function in CBE Plant Research | Example/Notes |
|---|---|---|
| nCas9 (D10A) Backbone Vector | Provides the DNA-targeting module that creates the ssDNA bubble for deaminase action. | Often from Streptococcus pyogenes (SpCas9). A plant codon-optimized version is essential. |
| Modular Deaminase Variant Cloning Kit | Allows rapid swapping of deaminase domains (e.g., APOBEC1, A3A, CDA1) into the CBE fusion. | Commercial Golden Gate or Gibson Assembly kits tailored for plant expression vectors. |
| UGI Variant Plasmids | Sources of wild-type, tandem (2x), and evolved (eUGI) sequences for fusion to CBE. | Critical control: A plasmid encoding a UGI-null (mutated) version. |
| Plant Protoplast Isolation Kit | Standardized enzymes and solutions for reproducible protoplast generation from model plants. | Contains Cellulase R10/Macerozyme R10 or similar. Mannitol for osmoticum. |
| High-Efficiency PEG Transfection Reagent | Polyethylene glycol solution optimized for plant protoplasts to deliver plasmid DNA. | 40% PEG-4000 with Ca²⁺ is standard. Commercial "Protoplast Transfection Enhancer" additives exist. |
| Plant gDNA Micro-Extraction Kit | Rapid, column-based DNA purification from small volumes of protoplasts. | Must effectively remove polysaccharides and secondary metabolites. |
| NGS Amplicon-Seq Library Prep Kit | For preparing barcoded sequencing libraries from target site PCR products. | Kits compatible with low DNA input (e.g., 10 ng) are ideal for high-throughput screening. |
| Base Editing Analysis Software | Bioinformatics tools to quantify base substitution frequencies and indels from NGS data. | CRISPResso2, BEAT, or custom pipelines (e.g., in R or Python). |
Cytosine base editors (CBEs) represent a precise genome editing tool that enables targeted C•G to T•A conversions without generating double-strand breaks. Their application in plants, however, is often hampered by plant-specific hurdles, most notably low transformation efficiency and high rates of chimerism in regenerated plants. This protocol, framed within a broader thesis on CBE applications in plant research, details optimized strategies to overcome these barriers, thereby enabling the generation of high-quality, uniformly edited plants for functional genomics and trait development.
The primary bottlenecks in plant CBE editing are summarized in the table below.
Table 1: Common Hurdles in Plant CBE Editing and Typical Efficiency Ranges
| Hurdle | Description | Typical Efficiency Range (Conventional Protocol) | Impact on CBE Outcomes |
|---|---|---|---|
| Transformation Efficiency | The percentage of explants that stably integrate the editing machinery. | 1-30% (Species-dependent) | Limits pool of cells available for editing. |
| Editing Efficiency in T0 | The percentage of sequenced alleles showing the desired edit in primary transformants. | 0.5-50% | Low efficiency increases screening burden. |
| Chimerism Rate | The proportion of T0 regenerants with edits in only a subset of somatic cells. | 20-90% | Complicates genotyping, requires additional generations to obtain uniform lines. |
| Plant Regeneration Rate | The ability of transformed cells to develop into whole plants. | Varies widely by genotype | Bottleneck for many crop species. |
Application Notes & Optimized Protocols
Protocol A: EnhancedAgrobacterium-Mediated Transformation for CBEs
This protocol increases the density of transformation events, providing a larger edited cell pool for regeneration.
Materials:
- Young, healthy leaf explants or embryogenic calli.
- Agrobacterium tumefaciens strain (e.g., EHA105, LBA4404) harboring CBE construct (e.g., rAPOBEC1-nCas9-UGI).
- Acetosyringone (200 µM final concentration in co-culture medium).
- Optimal plant-specific co-culture medium (see Table 2).
- Novel surfactants (e.g., Silwet L-77 at 0.005-0.01%) or physical aids (e.g., sonication, vacuum infiltration).
Procedure:
- Pre-culture Explants: Culture explants on pre-regeneration medium for 2 days.
- Agrobacterium Preparation: Grow Agrobacterium to OD₆₀₀ = 0.6-0.8. Centrifuge and resuspend in induction medium containing acetosyringone.
- Enhanced Inoculation: Immerse explants in bacterial suspension. Apply brief vacuum infiltration (30-60 seconds) or gentle sonication (10-20 seconds).
- Co-culture: Blot-dry explants and co-culture on filter paper-overlaid medium in the dark at 22°C for 3 days. Critical: Limit co-culture time to reduce overgrowth.
- Washing & Selection: Wash thoroughly with sterile water containing carbenicillin (500 mg/L). Transfer to selection medium containing appropriate antibiotic and cytokinin/auxin balance.
Protocol B: Regeneration from Single Cell-Derived Callus to Minimize Chimerism
This protocol prioritizes the regeneration of plants from a single edited cell.
Materials:
- Transformed explants after initial selection.
- Hyperosmotic pre-treatment medium (e.g., supplemented with 0.2-0.4 M Mannitol).
- Protoplasting enzymes (for amenable species).
- Liquid regeneration medium for micro-callus formation.
Procedure:
- Single Cell Induction: After initial selection for 10-14 days, transfer emerging micro-calli to a hyperosmotic medium for 3-5 days to promote single-cell state.
- Micro-callus Proliferation: Manually isolate and transfer compact, yellow micro-calli (<1 mm) to fresh proliferation medium.
- Regeneration: Induce shoot formation on high-cytokinin medium. Monitor for single-point origin of shoots.
- Rooting & Genotyping: Root individual shoots. Perform early and deep sequencing (e.g., amplicon-seq of >1000X depth) on leaf samples from multiple sectors of the T0 plant to assess chimerism.
Protocol C:In PlantaDelivery Systems to Bypass Tissue Culture
For species with recalcitrant regeneration, this approach aims to edit meristematic cells.
Materials:
- De-germinated seeds or seedling apical meristems.
- Agrobacterium strain with virulent helper plasmid (e.g., pVirG).
- Floral dip or seedling vacuum infiltration solution (5% sucrose, 0.05% Silwet L-77).
- Growth chamber for rapid seedling recovery.
Procedure (Seedling Vacuum Infiltration):
- Surface-sterilize and germinate seeds on moist filter paper.
- At the hypocotyl emergence stage, immerse seedlings in Agrobacterium suspension (OD₆₀₀ = 1.0-1.5).
- Apply vacuum (25-30 in Hg) for 3-5 minutes, then release slowly.
- Co-culture seedlings on moist paper in the dark for 48 hours.
- Transfer to soil and grow to maturity. Screen progeny (T1) for inherited edits, indicating editing of germline precursor cells.
Research Reagent Solutions Toolkit
Table 2: Essential Materials for Improving CBE Outcomes in Plants
| Reagent / Material | Function in Protocol | Example / Specification |
|---|---|---|
| CRISPR-CBE Plasmid System | Expresses guide RNA and base editor fusion protein. | pRGEB32-like vector with plant codon-optimized rAPOBEC1, nCas9 (D10A), and UGI. |
| Stem Cell Marker Gene | Visual selection of transformation events. | GFP, YFP, or RFP driven by a strong constitutive promoter (e.g., 35S, ZmUbi). |
| Virulence Inducer | Activates Agrobacterium vir genes during co-culture. | Acetosyringone (200 µM). |
| Surface Active Agent | Lowers surface tension for better bacterial infiltration. | Silwet L-77 (0.005-0.05%). |
| Selection Antibiotic | Selects for plant cells with integrated T-DNA. | Hygromycin (10-50 mg/L) or Kanamycin (50-100 mg/L), species-dependent. |
| Phytohormone Mix | Drives callus induction and shoot regeneration. | 2,4-D (auxin) for callus; BAP/TDZ (cytokinin) for shoot organogenesis. |
| Osmoticum | Promotes single-cell state from callus. | Mannitol (0.2-0.4 M) in pre-treatment medium. |
| High-Fidelity PCR Kit | Amplifies target locus for deep sequencing analysis. | KAPA HiFi HotStart ReadyMix or equivalent. |
| NGS Amplicon-Seq Service | Quantifies editing efficiency and detects chimerism at high depth. | Illumina MiSeq, >1000X sequencing depth per sample. |
Visualized Workflows & Pathways
Diagram Title: Optimized CBE Workflow to Reduce Chimerism
Diagram Title: Mechanism of CBE Action in Plant Cells
Validating and Comparing CBE Outcomes: Analysis, Benchmarking, and Alternative Tools
The precise characterization of edits introduced by CRISPR-Cas9-derived Cytosine Base Editors (CBEs) is a critical step in plant research. CBEs facilitate the direct, programmable conversion of cytosine to thymine within a defined window without creating double-strand breaks, enabling single-nucleotide alterations. Within the broader thesis on "Optimizing CBE Protocols for Horticultural Crop Improvement," robust genotyping is essential to: 1) Determine editing efficiency across plant lines, 2) Identify the spectrum of on-target edits (C-to-T conversions) and potential byproducts (e.g., indels, C-to-G/A edits), and 3) Detect the presence of the transgene for segregation in subsequent generations. This application note details two complementary genotyping approaches—Sanger sequencing and High-Throughput Amplicon Sequencing (Amplicon-Seq)—alongside the bioinformatic tools required for accurate analysis.
The choice of genotyping method depends on the experimental stage, required resolution, and sample throughput.
Table 1: Comparison of Genotyping Methods for CBE-Edited Plants
| Feature | Sanger Sequencing | High-Throughput Amplicon Sequencing (Amplicon-Seq) |
|---|---|---|
| Primary Application | Initial screening, low-sample-number validation, transgene segregation. | High-throughput characterization of edit diversity, efficiency, and precision in pooled populations. |
| Resolution | Identifies edits in pooled PCR products from a plant or tissue; reveals major alleles but masks minor variants (<15-20%). | Single-nucleotide resolution, capable of detecting variants present at <1% allele frequency in a pooled sample. |
| Throughput | Low to medium (tens to hundreds of samples). | Very high (hundreds to thousands of amplicons across many samples). |
| Key Output Metrics | Chromatogram quality, base-calling, inferred edit types. | Edit efficiency (% C-to-T), edit purity (% intended edits vs. byproducts), allele frequency tables. |
| Typical Cost per Sample | $5 - $15 | $20 - $100 (highly dependent on multiplexing level and sequencing depth) |
| Best For | Rapid confirmation of edits in T0/T1 plants, checking for homozygous/biallelic edits, segregating transgene-free lines. | Comprehensive profiling of edit outcomes in a population, detecting rare editing byproducts, analyzing complex tissue samples (e.g., chimeric leaves). |
Protocols and Application Notes
Protocol A: Sanger Sequencing for Initial CBE Edit Screening
Objective: To amplify and sequence the target genomic locus from individual CBE-treated plants for rapid identification of base edits.
Materials (Research Reagent Solutions):
- Plant DNA Extraction Kit: (e.g., CTAB-based or commercial kits like NucleoSpin Plant II). Function: Isolates high-quality, PCR-amplifiable genomic DNA.
- High-Fidelity DNA Polymerase: (e.g., Q5, Phusion). Function: Ensures accurate amplification of the target locus with minimal PCR errors.
- Target-Specific Primers: Designed to amplify a 300-500 bp region surrounding the CBE target site.
- PCR Purification Kit: (e.g., NucleoSpin Gel and PCR Clean-up). Function: Removes primers, dNTPs, and enzymes prior to sequencing.
- Sanger Sequencing Service: Utilizing BigDye Terminator chemistry.
Detailed Workflow:
- Genomic DNA Isolation: Extract DNA from ~100 mg of leaf tissue from wild-type (WT) and putative edited (T0) plants. Quantify DNA using a fluorometer.
- PCR Amplification: Set up a 25-50 µL reaction with high-fidelity polymerase. Use a touchdown PCR program to enhance specificity. Include a no-template control.
- Typical Cycle: 98°C 30s; (98°C 10s, 65-55°C 30s [-0.5°C/cycle], 72°C 30s/kb) for 15 cycles; (98°C 10s, 55°C 30s, 72°C 30s/kb) for 25 cycles; 72°C 2 min.
- Amplicon Verification: Run 5 µL of PCR product on a 1.5% agarose gel. A single, bright band of the expected size should be present.
- PCR Clean-up: Purify the remaining PCR product according to the purification kit protocol. Elute in 20-30 µL of nuclease-free water.
- Sequencing Submission: Submit purified amplicons for Sanger sequencing using one of the PCR primers as the sequencing primer. Sequencing from both ends is recommended for high-confidence base calling.
- Analysis: Align the returned sequencing chromatogram to the reference WT sequence using tools like SnapGene, BioEdit, or online platforms like DSDecodeM or ICE (Synthego). Look for overlapping peaks (double peaks) starting at the CBE activity window, indicating successful editing.
Protocol B: High-Throughput Amplicon Sequencing for Population-Wide Analysis
Objective: To deeply sequence PCR amplicons from many plants (or pooled tissues) to quantify editing efficiency and characterize the full spectrum of edit alleles.
Materials (Research Reagent Solutions):
- Multiplexing-Compatible PCR Master Mix: (e.g., KAPA HiFi HotStart ReadyMix with unique dual-indexing primers). Function: Enables pooled sequencing of hundreds of amplicons with sample-specific barcodes.
- Next-Generation Sequencing Platform: (e.g., Illumina MiSeq, NovaSeq 6000). Function: Provides the high-throughput, short-read sequencing capability.
- SPRIselect Beads: (e.g., Beckman Coulter). Function: For post-PCR clean-up and accurate amplicon size selection, removing primer dimers.
- Library Quantification Kit: (e.g., KAPA Library Quantification Kit for Illumina). Function: Accurately measures the concentration of the final sequencing library for optimal cluster density.
Detailed Workflow:
- DNA Isolation & Primary PCR: Isolate DNA and perform initial target amplification as in Protocol A, Step 1-3, but in a smaller volume (e.g., 15 µL).
- Indexing (Barcoding) PCR: Use a second, short-cycle PCR to append unique dual-index sequences (i5 and i7) and full Illumina adapter sequences to each amplicon. This allows multiplexing.
- Library Purification & Pooling: Clean up indexing PCR products with SPRIselect beads (0.8x ratio). Quantify each library fluorometrically, then pool equimolar amounts of all samples into a single tube.
- Sequencing: Submit the pooled library for sequencing on an Illumina platform (e.g., MiSeq v2, 2x250 bp). Target a minimum depth of 10,000 reads per amplicon sample for reliable variant detection.
- Bioinformatic Analysis:
- Demultiplexing: The sequencing facility provides FASTQ files separated by sample index.
- Primary Processing: Use a pipeline like CRISPResso2 or AmpliconDIVider. Steps include: 1) Align reads to the reference amplicon sequence (Bowtie2/BWA). 2) Identify the CBE target window. 3) Quantify the percentage of reads with C-to-T (and other nucleotide) conversions at each position. 4) Generate summary statistics and visualizations.
Analysis Tools and Data Interpretation
Table 2: Key Bioinformatic Tools for CBE Genotyping Data
| Tool Name | Primary Use Case | Input | Key Output |
|---|---|---|---|
| ICE (Inference of CRISPR Edits) - Synthego | Analysis of Sanger traces from edited, potentially heterogeneous samples. | Sanger .ab1 chromatogram file, WT reference sequence. | Editing efficiency graph, inferred allele composition, quality score. |
| CRISPResso2 | Comprehensive analysis of NGS amplicon data from CRISPR-edited pools. | Paired-end FASTQ files, amplicon & guide sequences. | Nucleotide substitution plots, allele frequency tables, quantification of editing efficiency and byproducts. |
| AmpliconDIVider | Specialized for base editor analysis from NGS amplicon data. | Aligned BAM file, target site location. | Detailed breakdown of base conversion frequencies within the editing window. |
| Geneious Prime | Integrated platform for manual inspection of Sanger/NGS alignments. | Sequence files, reference. | Alignments, variant calling, primer design. |
Visualized Workflows
Title: Sanger Sequencing Workflow for CBE Plants
Title: High-Throughput Amplicon Sequencing Workflow
Title: Genotyping Method Selection Guide
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for CBE Plant Genotyping
| Reagent / Material | Supplier Examples | Function in Genotyping |
|---|---|---|
| High-Fidelity PCR Kit | NEB (Q5), Thermo Fisher (Phusion), KAPA Biosystems | Ensures accurate, specific amplification of genomic target for both Sanger and NGS library prep. |
| Plant DNA Isolation Kit | Macherey-Nagel (NucleoSpin), Qiagen (DNeasy), CTAB Protocol | Reliable extraction of PCR-ready DNA from diverse, polysaccharide-rich plant tissues. |
| SPRIselect Beads | Beckman Coulter | Size-selection and clean-up of amplicon libraries; critical for removing primer dimers before NGS. |
| Dual-Indexed Primer Sets | Integrated DNA Technologies (IDT), Twist Bioscience | Allows multiplexing of hundreds of samples in one NGS run by attaching unique barcode combinations. |
| NGS Library Quant Kit | KAPA Biosystems (qPCR), Invitrogen (Qubit dsDNA HS) | Accurate quantification of the final sequencing library is essential for optimal Illumina cluster generation. |
| Sanger Sequencing Kit | Thermo Fisher (BigDye Terminator v3.1) | Industry-standard chemistry for capillary electrophoresis-based sequencing of single amplicons. |
Phenotypic validation is the critical step in functional genomics and plant breeding that confirms a causal link between a genetic modification (genotype) and an observable characteristic (trait). Within the broader thesis on Cytosine Base Editor (CBE) protocols in plants, this process is paramount. It moves beyond confirming successful DNA editing to demonstrating that the precise C→T (or G→A) substitution produces a functionally relevant phenotypic outcome, such as herbicide resistance, altered flowering time, or improved nutritional content. This document provides application notes and detailed protocols for designing and executing robust phenotypic validation experiments following CBE-mediated genome editing in plants.
Core Quantitative Data: Key Metrics for Phenotypic Validation
Table 1: Quantitative Metrics for Phenotypic Validation in CBE-Edited Plants
| Metric Category | Specific Parameter | Typical Measurement Method | Data Output Example (e.g., Herbicide Resistance Gene) | Significance for Validation |
|---|---|---|---|---|
| Editing Efficiency | Target Site Modification Rate | NGS Amplicon Sequencing | 92% C→T conversion in T1 generation | Establishes the penetrance of the genotype in the population. |
| Homozygosity | Percentage of Homozygous Edited Plants | PCR/RE Digestion or NGS | 70% of T1 plants are homozygous for the W574L allele | Links a stable, non-segregating genotype to the trait. |
| Protein Effect | Amino Acid Change & Protein Level | Western Blot / ELISA | W574L substitution confirmed; protein abundance at 110% of WT. | Confirms the edit translates to the expected molecular product. |
| Primary Phenotype | Trait of Interest (e.g., Survival Rate) | Herbicide Spray Assay | 95% survival at 2x field dose of herbicide vs. 0% WT survival. | Direct evidence of the engineered trait's function. |
| Secondary/Agronomic Traits | Plant Height, Yield, Flowering Time | Digital Phenotyping (RGB/IR imaging), manual measurement | No significant difference in yield components vs. WT under control conditions. | Ensures the edit does not have unforeseen negative pleiotropic effects. |
| Segregation Correlation | Co-segregation of Genotype & Phenotype | Chi-squared Test on T2 Population | χ² = 0.15, p > 0.05 (fit to expected Mendelian ratio for dominant trait). | Statistically reinforces the causal genotype-phenotype link. |
Table 2: Common Plant CBE Targets and Associated Phenotypic Assays
| Target Gene | Intended Amino Acid Change | Resulting Trait | Key Phenotypic Validation Protocol |
|---|---|---|---|
| Acetolactate Synthase (ALS) | Proline 186 to Serine / Tryptophan 574 to Leucine | Herbicide (e.g., Chlorsulfuron) Resistance | Root growth or whole-plant spray assay with selective herbicide. |
| Acc1 / accD (plastid) | Various, to inactivate enzyme | Resistance to Aryloxyphenoxypropionate (FOP) herbicides | Seed germination or seedling growth assay on media containing herbicide. |
| EPSPS | Threonine 102 to Isoleucine + Alanine 103 to Serine (double) | Glyphosate Tolerance | Callus or whole-plant assay with graded glyphosate concentrations. |
| Flowering Locus T (FT) | Cys/Tyr in key functional domain | Altered Flowering Time | Days to flowering under controlled long-day/short-day conditions. |
| GBSSI (Waxy) | Introduction of premature stop codon | High-amylose, low-viscosity starch | Iodine staining of pollen/seeds; rheological measurement of starch. |
Detailed Experimental Protocols
Protocol 3.1: Herbicide Resistance Phenotyping for ALS-Edited Plants
A. Seed-Based Root Inhibition Assay (Primary Screen)
- Surface Sterilization: Sterilize seeds (T1 or T2 generation) from edited and wild-type (WT) lines.
- Plating: Sow seeds on ½ MS media plates supplemented with a sub-lethal concentration of the target herbicide (e.g., 50 nM Chlorsulfuron). Include control plates without herbicide.
- Growth Conditions: Stratify at 4°C for 48h, then incubate vertically in a growth chamber (22°C, 16h light).
- Data Collection (7-10 days post-germination):
- Measure primary root length for each seedling.
- Record germination rate and visible signs of chlorosis/necrosis.
- Analysis: Calculate percentage root growth inhibition relative to control plates. Compare edited and WT populations statistically (t-test).
B. Whole-Plant Spray Assay (Confirmatory)
- Plant Growth: Grow edited and WT plants in soil under standard conditions until the 4-6 leaf stage.
- Herbicide Application: Apply herbicide (e.g., Chlorsulfuron at 1x and 2x field-recommended rate) using a precision spray chamber. Include a water-only control group.
- Scoring & Monitoring: Visually score plants at 3, 7, and 14 days after treatment (DAT) using a standardized injury scale (0% = no effect, 100% = complete death). Capture digital images.
- Biomass Measurement: At 21 DAT, harvest and dry shoots to measure dry weight. Resistance is validated by significantly higher survival and biomass in edited lines versus WT.
Protocol 3.2: Validation of Altered Flowering Time
- Genotyping & Plant Selection: Identify homozygous edited and WT plants (T2 or later generation) via PCR/sequencing.
- Synchronized Growth: Sow seeds directly in soil. After germination, thin to one plant per pot. Use a minimum of 12 plants per genotype.
- Environmental Control: Place plants in a tightly controlled growth chamber or cabinet with specific photoperiods (e.g., long days: 16h light/8h dark).
- Phenotyping: Record the following:
- Days to flowering: From sowing to first visible bud emergence.
- Leaf number at flowering: Count total rosette leaves at flowering as a developmentally stable metric.
- Bolting height: Measure stem length at weekly intervals.
- Statistical Analysis: Compare means between edited and WT groups using ANOVA. Plot survival curves for time-to-flower event data.
Visualization of Workflows and Relationships
Diagram Title: Phenotypic Validation Workflow for CBE-Edited Plants
Diagram Title: Linking CBE Mechanism to Final Trait
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for Phenotypic Validation
| Category | Item / Reagent | Function / Application | Example Product / Specification |
|---|---|---|---|
| Genotyping | High-Fidelity PCR Mix | Accurate amplification of target locus for sequencing analysis. | Thermo Scientific Phusion Green Hot Start Mix. |
| Restriction Enzymes (if applicable) | Cleavage-based assay for detecting loss/gain of a restriction site via edit. | New England Biolabs (NEB) enzymes. | |
| Sanger Sequencing Service | Confirmation of edits in initial lines. | In-house capillary sequencer or commercial service. | |
| Deep Sequencing | NGS Amplicon-Seq Kit | Library prep for high-throughput sequencing of target amplicons. | Illumina DNA Prep with Unique Dual Indexes. |
| Target-Specific Primers (with overhangs) | Amplification of genomic region for NGS library. | HPLC-purified primers with Illumina adapter sequences. | |
| Phenotyping Assays | Purified Herbicide (e.g., Chlorsulfuron) | Active ingredient for selective pressure in resistance assays. | Sigma-Aldrich, PESTANAL grade. |
| Controlled Environment Growth Chamber | Standardized light, temperature, humidity for plant growth. | Percival or Conviron chamber with programmable settings. | |
| Digital Imaging System | Quantitative measurement of plant growth, color, architecture. | RGB/IR camera setup with analysis software (e.g., ImageJ, PlantCV). | |
| General | Tissue Culture Media (MS Basal Salts) | For in vitro propagation and selection of edited plants. | PhytoTechnology Laboratories Murashige & Skoog Basal Salt Mixture. |
| Soil Matrix | Consistent substrate for whole-plant phenotyping. | SunGro Horticulture Sunshine Mix #1. |
Within the broader thesis on developing a high-fidelity Cytosine Base Editor (CBE) protocol for plants, a rigorous assessment of off-target effects is a critical validation step. CBEs, which consist of a Cas9 nickase fused to a cytidine deaminase, can induce unwanted deamination at sites beyond the intended on-target locus. These off-target edits, if present in the germline, can lead to heritable mutations with potentially detrimental phenotypic consequences. This document outlines complementary application notes and protocols for two principal assessment strategies: in silico computational prediction and empirical detection via whole-genome sequencing (WGS).
Application Notes & Protocols
A. Computational Prediction of Potential Off-Target Sites
Application Note: This in silico approach is used prior to experimental transformation to predict and rank potential off-target sites across the genome. It informs guide RNA (gRNA) selection and identifies loci for targeted deep sequencing validation.
Protocol: In Silico Off-Target Prediction Using Cas-OFFinder and CBE-Specific Scoring
Principle: Identify genomic loci with sequence similarity to the intended gRNA spacer, allowing for mismatches and DNA/RNA bulge formations, then apply a CBE-specific scoring model that weighs the sequence context (e.g., presence of required NG, NNG, or NCN protospacer adjacent motif (PAM) for nCas9 and the preferred editing window).
Materials & Software:
- Reference genome sequence of the target plant species (e.g., Arabidopsis thaliana TAIR10, Oryza sativa IRGSP-1.0).
- Intended gRNA spacer sequence (20-nt).
- Cas-OFFinder software (standalone or web tool).
- Command-line terminal or web browser.
Detailed Methodology:
Parameter Definition:
- Specify the PAM sequence corresponding to your nCas9 variant (e.g., 5'-NGG-3' for SpCas9).
- Set the search parameters: maximum number of DNA/RNA bulge sizes (typically 0-2) and maximum number of mismatches (typically 0-5).
- Define the genome to search against.
Execution:
- Input the gRNA sequence (without the PAM) into Cas-OFFinder.
- Run the search algorithm. The output is a list of genomic coordinates with alignment details.
CBE-Specific Filtering & Ranking:
- Filter results to retain only sites with a canonical PAM.
- Extract the sequence context for each potential off-target site, focusing on the "editing window" (typically positions 3-10 within the protospacer, relative to the PAM).
- Rank sites using a combined score considering:
- Alignment Score: Number and position of mismatches/bulges (fewer and distal mismatches score higher risk).
- Sequence Context Score: Preference for cytidines within a 5'-TC-3' or 5'-AC-3' context (for common APOBEC-based deaminases) over other contexts.
- Generate a prioritized list of top ~20-50 potential off-target sites for experimental validation.
Data Presentation: Table of Predicted Off-Target Sites
Table 1: Example Output of Predicted Off-Target Sites for a CBE gRNA Targeting the *Arabidopsis PDS3 Gene.*
| Rank | Chromosome | Position (Strand) | Predicted Off-Target Sequence (PAM underlined) | Mismatches | Bulges | CBE Context Score (1-10) | Recommended for Validation? |
|---|---|---|---|---|---|---|---|
| 1 | Chr3 | 12548765 (+) | GATCCTCCAGAGTCACCTGA TGG | 0 | 0 | 9.2 | Yes (On-Target) |
| 2 | Chr1 | 23897412 (-) | GATCCTGCAGAGTCACCTCA AGG | 2 | 0 | 7.1 | Yes |
| 3 | Chr5 | 18965433 (+) | GATCCTCCAGAGGCACCTTA TGG | 3 | 0 | 4.5 | Yes |
| 4 | Chr2 | 45632178 (+) | GATCCTCCGAAGTCACCTGA CGG | 4 | 0 | 2.1 | No |
The Scientist's Toolkit: Key Reagents & Resources for Computational Prediction
| Item | Function/Description |
|---|---|
| Cas-OFFinder | Algorithm for genome-wide search of potential off-target sites with user-defined mismatch/bulge tolerance. |
| Plant Reference Genome (FASTA) | High-quality, chromosome-level assembled genome for the species of interest. Essential for accurate mapping. |
| CRISPRseek/Biostrings (R) | R/Bioconductor packages for additional gRNA specificity analysis and sequence manipulation. |
| Custom Python/R Script | For implementing CBE-specific context scoring and filtering of raw Cas-OFFinder results. |
Diagram: Computational Off-Target Prediction Workflow (79 chars)
B. Empirical Detection via Whole-Genome Sequencing (WGS)
Application Note: This unbiased, genome-wide experimental approach is used after regenerating plants from edited tissue to identify all de novo single nucleotide variants (SNVs), providing the most comprehensive profile of CBE-mediated off-target activity.
Protocol: Whole-Genome Sequencing for De Novo SNV Detection in CBE-Edited Plants
Principle: Sequence the genomes of an edited plant (T1 generation) and its parental wild-type control to high coverage. Use a robust bioinformatics pipeline to call variants, then filter aggressively to isolate CBE-induced, non-hybrid SNVs—specifically C•G to T•A transitions within a defined sequence context.
Materials:
- Biological Material: High-molecular-weight genomic DNA (gDNA) from a single, independent CBE-edited T1 plant and its unedited parental line.
- Library Prep Kit: PCR-free WGS library preparation kit (to avoid PCR-introduced errors).
- Sequencing Platform: Illumina NovaSeq or equivalent for >30x coverage.
Detailed Methodology:
DNA Extraction & QC:
- Extract gDNA using a method optimized for long fragments (e.g., CTAB with RNase A).
- Assess purity (A260/A280 ~1.8, A260/A230 >2.0) and integrity (Fragment Analyzer/TapeStation: DIN >7.0).
- Quantify via fluorometry (Qubit).
Library Preparation & Sequencing:
- Use a PCR-free library preparation protocol to minimize sequencing artifacts.
- Perform paired-end sequencing (2x150 bp) to achieve a minimum of 30x coverage for both edited and control samples.
Bioinformatic Analysis Pipeline:
- Quality Control & Trimming: Use FastQC and Trimmomatic.
- Alignment: Map reads to the reference genome using BWA-MEM or HiSat2.
- Variant Calling: Call SNVs using GATK HaplotypeCaller in "GVCF" mode for both samples.
- Variant Filtration (Critical Step):
- Combine samples using GATK CombineGVCFs and perform joint genotyping.
- Apply hard filters: QD < 2.0 || FS > 60.0 || MQ < 40.0 || SOR > 3.0.
- Isolate de novo variants: Present as homozygous/heterozygous in the edited sample but homozygous reference in the control.
- CBE-Specific Filter: Retain only C•G to T•A (or G•C to A•T) transitions.
- Context Filter: Filter for SNVs occurring within a 5-nucleotide window (e.g., T-C-N-A-G) matching the deaminase's preference.
- Remove Common Variants: Compare against available plant variome databases to exclude natural polymorphisms.
Data Presentation: Summary of WGS Off-Target Analysis
Table 2: Example Summary Statistics from WGS Off-Target Analysis of a CBE-Edited *Oryza sativa Plant.*
| Metric | Edited Sample (T1) | Control Sample (Parent) |
|---|---|---|
| Sequencing Depth (Mean) | 35x | 32x |
| Genome Coverage (>1x) | 98.5% | 98.7% |
| Total SNVs Called (Pre-Filter) | 124,507 | 119,832 |
| De novo SNVs (vs Control) | 89 | - |
| De novo C•G to T•A Transitions | 67 | - |
| C•G to T•A in Preferred Context | 41 | - |
| High-Confidence CBE Off-Target Candidates | 3 | 0 |
| On-Target Editing Efficiency | 92% | 0% |
The Scientist's Toolkit: Key Reagents & Resources for WGS Detection
| Item | Function/Description |
|---|---|
| PCR-Free WGS Library Prep Kit | Minimizes amplification bias and errors, crucial for accurate de novo variant detection. |
| High-Fidelity DNA Polymerase | For optional minimal-cycle amplification if required; reduces PCR errors. |
| BWA-MEM / HiSat2 | Aligners for mapping sequencing reads to a reference genome. |
| GATK (Genome Analysis Toolkit) | Industry-standard suite for variant discovery and genotyping. |
| SAMtools/Bcftools | For processing alignment files and variant calls. |
| Integrated Genome Viewer (IGV) | Visual validation of called putative off-target sites against sequence reads. |
Diagram: WGS Off-Target Detection Pipeline (58 chars)
Integration into the CBE Protocol Thesis
These protocols form the critical validation chapter of a CBE thesis. The computational prediction (Protocol A) is used for gRNA design optimization and creating a targeted amplicon sequencing panel for initial screening of potential off-target sites in multiple edited lines. The WGS approach (Protocol B) is the definitive, gold-standard experiment conducted on a small number of final, edited lines to provide a genome-wide safety profile. The convergence of in silico prediction with empirical WGS data provides robust evidence for the fidelity of a novel CBE system or optimized delivery protocol in plants, a necessary precursor to agricultural application.
This application note provides a comparative analysis of three primary precision genome editing tools—Cytosine Base Editors (CBEs), Adenine Base Editors (ABEs), and Prime Editors (PEs)—within the context of plant research. The analysis is framed by the overarching thesis that optimizing a CBE protocol for plants requires an understanding of its operational constraints relative to other editors. While CBEs enable C•G to T•A conversions, ABEs facilitate A•T to G•C changes, and PEs offer a broader range of targeted small insertions, deletions, and all 12 possible base-to-base conversions. The choice of editor is critical for achieving desired genotypes without double-strand breaks (DSBs) and depends on the target base change, required precision, and specific plant system.
Quantitative Performance Comparison Table
Table 1: Comparative Overview of Base Editors in Plants
| Feature | Cytosine Base Editors (CBEs) | Adenine Base Editors (ABEs) | Prime Editors (PEs) |
|---|---|---|---|
| Core Component | Cas9 nickase (nCas9) fused to cytidine deaminase (e.g., rAPOBEC1) and uracil glycosylase inhibitor (UGI). | Cas9 nickase (nCas9) fused to an engineered adenine deaminase (e.g., TadA8e). | Cas9 nickase (nCas9) fused to an engineered reverse transcriptase (RT). |
| Primary Edit | C•G to T•A | A•T to G•C | All 12 base substitutions, small insertions, deletions. |
| Editing Window | ~Protospacer positions 4-10 (from PAM, for SpCas9). | ~Protospacer positions 4-10 (from PAM, for SpCas9). | Positions 3-16 in the PAM-containing strand (- strand) are most efficient. |
| Typical Efficiency in Plants | 1% to 60%, highly variable by species and target. | 0.5% to 40%, often lower than CBEs in plants. | 0.1% to 10% in primary transformations; generally lower than CBEs/ABEs. |
| Purity (Desired Edit %) | Moderate; prone to undesired C-to-G, C-to-A, and indels. | High; fewer byproducts than CBEs. | Very High; minimal indels or bystander edits. |
| Key Byproducts | Random indels, C-to-G transversions, bystander C edits within window. | Fewer byproducts; primarily indels. | Primarily large deletions at high PE expression levels. |
| Delivery in Plants | Agrobacterium T-DNA, particle bombardment, ribonucleoprotein (RNP) complexes. | Agrobacterium T-DNA, particle bombardment, RNP. | Agrobacterium T-DNA; size poses delivery challenges. |
| Major Advantage | Efficient C-to-T conversion. | Efficient, clean A-to-G conversion. | Versatility; can install precise edits beyond single-base changes. |
| Major Limitation | Off-target DNA/RNA editing, bystander edits within window. | Limited to A-to-G edits. | Lower efficiency, larger construct size, complex pegRNA design. |
Experimental Protocols
Protocol 1: Designing and Testing a CBE for Targeted Gene Knockout in Rice
Objective: Create a premature stop codon via C•G to T•A conversion.
- Target Selection: Identify a target gene exon. Using a SpCas9-based CBE (e.g., BE3), select a 20-nt protospacer with a 5'-NGG PAM. Ensure the spacer contains a cytidine (C) at positions 4-10 relative to the PAM.
- Construct Assembly: Clone the sgRNA expression cassette (U3/U6 promoter-driven) and the nCas9-cytidine deaminase-UGI expression cassette (Ubiquitin or 35S promoter-driven) into a binary T-DNA vector.
- Plant Transformation: Transform Oryza sativa japonica calli via Agrobacterium tumefaciens strain EHA105. Co-cultivate for 3 days, select on hygromycin for 4 weeks.
- Genotype Screening: Isolate genomic DNA from regenerated T0 plantlets. Amplify the target region by PCR and subject to Sanger sequencing. Use tracking of indels by decomposition (TIDE) or ICE analysis to quantify editing efficiency.
- Analysis: Identify plants with homozygous C-to-T changes creating a STOP codon. Screen for potential bystander edits at neighboring Cs within the editing window.
Protocol 2: Installing a Precision Point Mutation with ABE in Arabidopsis
Objective: Convert an A•T to G•C to create an herbicide-tolerant allele.
- pegRNA/ngRNA Design: For ABE8e, design a standard sgRNA targeting the genomic locus, ensuring the target adenine (A) is within positions 4-10.
- Delivery: Use the floral dip method. Transform Arabidopsis thaliana Col-0 with Agrobacterium (GV3101) carrying the ABE8e T-DNA construct.
- Selection & Harvest: Select T1 seeds on BASTA plates. Collect leaf tissue from resistant seedlings.
- High-Throughput Genotyping: Perform PCR on leaf DNA. Use a restriction enzyme site created or destroyed by the A-to-G edit for initial screening. Confirm precise edits via Sanger sequencing of PCR amplicons.
- Segregation Analysis: Grow T2 progeny to identify transgene-free, precisely edited lines.
Protocol 3: Targeted Small Insertion using Prime Editing in Wheat Protoplasts
Objective: Validate PE functionality by installing a 6-bp insertion.
- pegRNA Design: Design a pegRNA containing: a) spacer sequence (13-20 nt), b) primer binding site (PBS, ~13 nt), c) RT template encoding the desired 6-bp insertion. Use web tools (PE-Designer) for optimization.
- Construct Assembly: Clone the pegRNA (U6-driven) and the nCas9-RT fusion (35S-driven) into a single plasmid.
- Protoplast Transfection: Isolate protoplasts from wheat mesophyll cells. Transfect with the PE plasmid using PEG-mediated transformation.
- DNA Extraction & Analysis: Harvest protoplasts after 48 hours. Extract genomic DNA and perform deep sequencing (amplicon-seq) of the target locus.
- Data Processing: Analyze sequencing data with CRISPResso2 or similar to quantify precise insertion efficiency and byproduct spectrum.
Diagrams
Title: Editor Selection & Experimental Workflow
Title: Core Molecular Mechanisms of CBEs, ABEs, and PEs
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for Plant Base Editing
| Reagent/Material | Function/Explanation |
|---|---|
| Binary T-DNA Vectors (e.g., pCAMBIA, pGreen) | Plant transformation backbones for Agrobacterium-mediated delivery of editor constructs. |
| Cas9-Nickase Variants (SpnCas9, SaCas9) | Catalytically impaired Cas9 creating a single-strand nick, the foundation for all three editors. |
| Deaminase Domains (rAPOBEC1 for CBE, TadA8e for ABE) | Enzymatic cores that catalyze the direct conversion of C to U, or A to I, in DNA. |
| Uracil Glycosylase Inhibitor (UGI) | Critical CBE component that prevents uracil excision, increasing edit yield and purity. |
| Engineered Reverse Transcriptase (RT) | PE core component that synthesizes DNA from the pegRNA template. |
| Plant Codon-Optimized Genes | Editor genes optimized for plant expression to improve translation and efficiency. |
| Tissue-Specific or Inducible Promoters (e.g., pUbi, p35S, pDD45) | Control spatial/temporal expression of editors to improve efficiency or reduce somatic off-targets. |
| Plant Selection Agents (Hygromycin, BASTA/Glufosinate) | Allow selection of transformed tissue expressing the editor construct. |
| High-Fidelity Polymerase for Amplicon-Seq | Essential for generating unbiased PCR products for deep sequencing analysis of editing outcomes. |
| PEG Solution for Protoplast Transfection | Enables direct delivery of plasmid or RNP complexes into plant cells for rapid testing. |
Application Notes
Cytosine base editors (CBEs) enable targeted C•G to T•A conversions without inducing double-strand breaks, making them invaluable for plant research and therapeutic development. This evaluation benchmarks the latest CBE platforms—including first-generation rAPOBEC1-based systems and advanced versions like BE-PLUS, SECURE-BE, and evoAPOBEC1-BE—on key parameters: editing efficiency, product purity (reducing undesired indels and stochastic C•G to G•C/A•T transversions), and specificity (minimizing off-target DNA and RNA edits). Recent studies indicate that while rAPOBEC1-BE3 shows robust on-target activity in plants, it can cause substantial genome-wide and transcriptome-wide off-target effects. Newer systems address these limitations: BE-PLUS incorporates bacteriophage Mu Gam protein to reduce indel formation, and SECURE-BE variants (e.g., SECURE-BE2, SECURE-BE3) use engineered, partially detached rAPOBEC1 variants to drastically lower DNA off-target editing while maintaining efficiency. Plant codon-optimized versions and delivery methods (e.g., Agrobacterium-mediated transformation, ribonucleoprotein complexes) are critical for performance.
Table 1: Benchmarking Key CBE Systems in Plants and Mammalian Cells
| CBE System | Deaminase | Efficiency Range (On-Target) | Key Indel Reduction Feature | Off-Target DNA Editing Reduction | Off-Target RNA Editing Reduction | Primary Plant Applications |
|---|---|---|---|---|---|---|
| BE3 (rAPOBEC1) | rAPOBEC1 | 5-50% (varies by locus) | None (baseline) | Baseline (high) | High (rAPOBEC1 activity) | Gene knockouts, directed evolution |
| BE-PLUS | rAPOBEC1 | 10-60% | Gam protein fusion | Moderate | High | High-efficiency editing with reduced indels |
| SECURE-BE3 | engineered rAPOBEC1 (P4/P5) | 10-40% | Partially detached deaminase | High (>90% reduction) | High | Sensitive applications requiring high specificity |
| evoAPOBEC1-BE4max | evoAPOBEC1 | 15-70% | Enhanced activity window | Moderate | Low (evoAPOBEC1 has low RNA off-targets) | High-efficiency base editing |
| ABE (Adenine Base Editor) | TadA-8e | 10-80% | N/A (A•T to G•C edits) | Generally low | Minimal | Correcting TA-to-CG mutations, precise A•T to G•C conversion |
Table 2: Quantitative Comparison of Specificity Metrics
| Metric | BE3 | BE-PLUS | SECURE-BE3 | evoAPOBEC1-BE4max |
|---|---|---|---|---|
| Average C•G to T•A Efficiency (%) | 30.5 | 38.2 | 22.7 | 45.8 |
| Indel Frequency (%) | 1.8 | 0.5 | 0.9 | 1.2 |
| DNA Off-Target Index (Relative to BE3) | 1.0 | 0.6 | 0.08 | 0.4 |
| RNA Off-Target Sites (Number) | >100 | >100 | >100 | <10 |
Experimental Protocols
Protocol 1:Agrobacterium-Mediated CBE Delivery inNicotiana benthamianafor Transient Efficiency Assay
Materials (Research Reagent Solutions)
- pBE121-CBE Expression Vector: Binary T-DNA vector containing plant codon-optimized CBE (e.g., rAPOBEC1-BE3) driven by a 35S promoter, and a guide RNA expression cassette.
- LB Medium & Antibiotics: For bacterial selection (Kanamycin, Spectinomycin).
- Infiltration Buffer (10 mM MES, 10 mM MgCl₂, 150 µM Acetosyringone, pH 5.6): Facilitates Agrobacterium infection.
- Agrobacterium tumefaciens Strain GV3101: Disarmed strain for plant transformation.
- CTAB DNA Extraction Buffer: For high-yield genomic DNA isolation from plant leaves.
- PCR Primers & Sanger Sequencing Reagents: For amplifying target locus and analyzing edits.
- T7 Endonuclease I or ICE Analysis Software: For initial efficiency estimation if using pooled samples.
Procedure
- Vector Construction: Clone your target 20-nt spacer sequence into the sgRNA scaffold of pBE121-CBE via BsaI Golden Gate assembly.
- Transform Agrobacterium: Introduce the assembled plasmid into GV3101 via electroporation. Select on LB plates with appropriate antibiotics.
- Culture Preparation: Grow a single colony in 5 mL LB with antibiotics at 28°C, shaking for 24-48 hrs. Pellet cells and resuspend in infiltration buffer to an OD₆₀₀ of 0.5. Incubate at room temperature for 2-4 hours.
- Plant Infiltration: Use a needleless syringe to infiltrate the bacterial suspension into the abaxial side of 3-4 week-old N. benthamiana leaves. Mark infiltration zones.
- Sample Harvest: Harvest leaf discs from infiltrated zones at 3-5 days post-infiltration. Flash-freeze in liquid nitrogen.
- DNA Extraction: Grind tissue, extract genomic DNA using CTAB method.
- PCR Amplification: Amplify target region (~300-500 bp) using high-fidelity polymerase.
- Editing Analysis: Purify PCR product and submit for Sanger sequencing. Use sequencing trace decomposition software (e.g., BE-Analyzer, EditR) or next-generation sequencing (NGS) for precise quantification of C•G to T•A conversion efficiency and indel frequency.
Protocol 2: NGS-Based Assessment of Off-Target DNA Editing (GOTI, GUIDE-seq adapted for plants)
Materials
- Predicted Off-Target Site List: Generated using tools like Cas-OFFinder.
- Multiplex PCR Primers: Designed with overhangs for Illumina indexing.
- High-Fidelity PCR Master Mix: For accurate amplification of multiple loci.
- Illumina DNA Library Prep Kit & Indexing Reagents: For NGS library construction.
- Bioinformatics Pipeline (CRISPResso2, custom scripts): For aligning sequences and quantifying edits.
Procedure
- Identify Potential Off-Target Sites: Input your sgRNA sequence and plant genome into Cas-OFFinder (allow up to 5 mismatches).
- Design Multiplex PCR: Design primers to amplify all potential off-target loci (150-250 bp amplicons). Include a positive control (on-target) locus.
- Genomic DNA Extraction: Extract gDNA from edited plant tissue and a wild-type control.
- Two-Step PCR Amplification: a) First PCR: Amplify all loci in separate reactions using locus-specific primers. b) Second PCR: Add Illumina flowcell binding sites and dual indices via a limited-cycle PCR.
- Library Purification & Pooling: Purify amplicons, quantify, and pool equimolarly.
- Sequencing & Analysis: Run on an Illumina MiSeq (2x250 bp). Use CRISPResso2 with appropriate parameters to quantify base edits at each locus. Compare to wild-type to filter background polymorphisms.
Protocol 3: In Vitro Deaminase Activity Assay (Specificity Screening)
Materials
- Purified CBE Protein: Express and purify CBE (e.g., rAPOBEC1-BE3 nickase, SECURE-BE3) from E. coli.
- Fluorescently-Labelled dsDNA Substrate: 50-nt dsDNA containing a target TC motif within a protospacer-like sequence.
- Reaction Buffer (20 mM HEPES, 150 mM KCl, 1 mM DTT, pH 7.5).
- Uracil-DNA Glycosylase (UDG) & Endonuclease VIII (APE1): For processing deaminated cytosines to strand breaks.
- Denaturing Polyacrylamide Gel Electrophoresis (PAGE) Setup.
Procedure
- Assemble Reaction: Mix 100 nM dsDNA substrate with 200 nM purified CBE protein in reaction buffer. Incubate at 37°C for 60 min.
- Reaction Quenching: Add Proteinase K to digest the editor.
- UDG/APE1 Treatment: To convert deaminated cytosines (uracils) into single-strand breaks, treat an aliquot of the product with UDG and APE1.
- Analysis: Run products on a denaturing PAGE gel. Compare untreated, CBE-treated, and UDG/APE1-treated lanes. Cleavage bands indicate deaminase activity. Quantify band intensity to calculate initial kinetics and compare deamination rates between different CBEs on identical substrates.
Visualization
Diagram 1: CBE Mechanism and Key Components
Diagram 2: Experimental Workflow for CBE Benchmarking in Plants
Diagram 3: Specificity Enhancement in SECURE-BE Systems
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for CBE Plant Research
| Item | Function | Example/Catalog |
|---|---|---|
| Plant Codon-Optimized CBE Plasmids | Ensures high expression of editor components in plant cells. | pBE121 series, pRCS series. |
| Golden Gate Assembly Kit (BsaI) | Modular, efficient cloning of sgRNA spacers into expression vectors. | NEB Golden Gate Assembly Kit (BsaI-HFv2). |
| Agrobacterium tumefaciens GV3101 | Disarmed strain for transient or stable plant transformation. | Common lab strain, commercially available. |
| Acetosyringone | Phenolic compound that induces Agrobacterium vir genes for T-DNA transfer. | Sigma-Aldrich D134406. |
| CTAB DNA Extraction Buffer | Effective isolation of high-molecular-weight genomic DNA from polysaccharide-rich plant tissue. | Standard lab formulation: CTAB, NaCl, EDTA, Tris, PVP. |
| High-Fidelity PCR Polymerase | Accurate amplification of target loci for sequencing analysis. | Q5 High-Fidelity DNA Polymerase (NEB). |
| BE-Analyzer or EditR Software | Quantifies base editing efficiency from Sanger sequencing chromatograms. | Free web tools (EditR, BE-Analyzer). |
| Cas-OFFinder Software | Identifies potential off-target sites in a plant genome. | Web-based or command-line tool. |
| Illumina DNA Library Prep Kit | Prepares amplicons for high-throughput sequencing to quantify edits. | Illumina DNA Prep Kit. |
| CRISPResso2 Software | Analyzes NGS data to quantify base editing and indel frequencies. | Open-source Python package. |
Conclusion
Cytosine base editors represent a transformative tool for precise genome engineering in plants, enabling single-base changes without the complications of double-strand breaks. Successful implementation hinges on a solid understanding of CBE mechanics, a robust and optimized protocol tailored to the plant species, vigilant troubleshooting to maximize on-target efficiency and minimize off-target effects, and rigorous validation of editing outcomes. As the technology evolves with improved specificity and expanded targeting ranges, its implications for biomedical and clinical research are profound—particularly in creating plant models of human diseases and engineering plants for the production of therapeutic compounds. Future directions will focus on developing novel deaminases with cleaner editing profiles, refining delivery methods for recalcitrant species, and integrating base editing with multiplexing strategies to accelerate both fundamental plant biology and the development of next-generation crops with enhanced nutritional and medicinal value.