Biolistic Bombardment vs. Agrobacterium Delivery: A Critical Comparison for Morphogene Transformation in Biomedical Research
This article provides a comprehensive analysis of two dominant genetic transformation methods—Agrobacterium-mediated delivery and particle bombardment (biolistics)—for introducing morphogenes into target cells.
Biolistic Bombardment vs. Agrobacterium Delivery: A Critical Comparison for Morphogene Transformation in Biomedical Research
Abstract
This article provides a comprehensive analysis of two dominant genetic transformation methods—Agrobacterium-mediated delivery and particle bombardment (biolistics)—for introducing morphogenes into target cells. Tailored for researchers, scientists, and drug development professionals, it explores the fundamental mechanisms, practical methodologies, common troubleshooting scenarios, and rigorous validation strategies. We synthesize current research to offer a data-driven comparison, highlighting the trade-offs between precision, efficiency, cargo capacity, and integration patterns. The conclusion outlines strategic selection criteria and emerging hybrid technologies to optimize gene delivery for therapeutic protein production, gene therapy vectors, and functional genomics.
Core Mechanisms: Understanding Agrobacterium Biology and Biolistic Physics
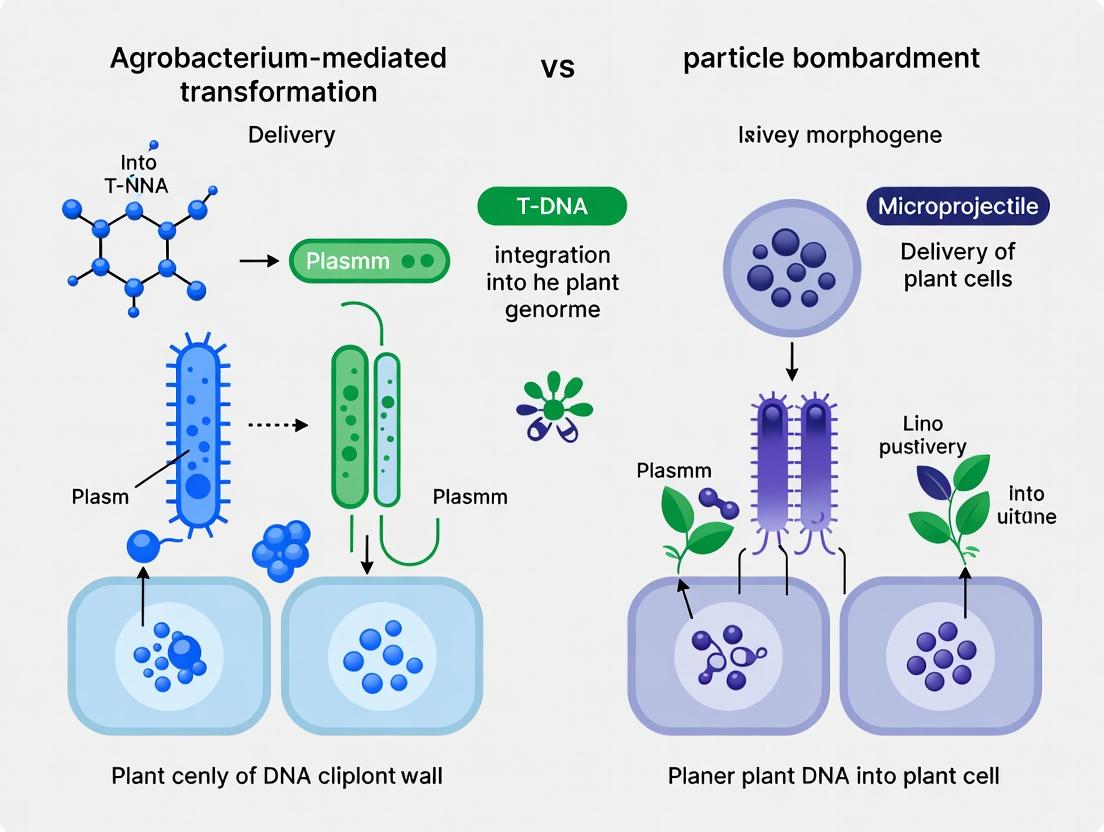
This guide compares the performance of Agrobacterium tumefaciens-mediated transformation (using its T-DNA/Vir system) with alternative direct DNA delivery methods, primarily particle bombardment, for morphogene delivery in plant research. The analysis is framed within the critical thesis of biological precision versus mechanical force for gene transfer.
Performance Comparison: Agrobacterium T-DNA/Vir vs. Particle Bombardment
Table 1: Core Performance Metrics for Morphogene Delivery
| Metric | Agrobacterium T-DNA/Vir System | Particle Bombardment (Biolistics) | Experimental Support & Key References |
|---|---|---|---|
| Transformation Mechanism | Biological, receptor-mediated, Vir protein-dependent. | Physical, ballistic force using microprojectiles. | (Gelvin, 2003; Altpeter et al., 2016) |
| Typical Copy Number | Low (1-3 copies), often simple integration patterns. | High, variable (1-20+), often complex, rearranged arrays. | Data from qPCR/Southern blot in rice: Agrobacterium avg. 1.8 copies; Bombardment avg. 8.5 copies. (Shrawat & Lörz, 2006) |
| Transgene Integrity & Stability | High. Precise T-DNA border-defined transfer. | Lower. Frequent truncation, rearrangement of DNA. | Sequencing of integration loci shows 85% intact T-DNA vs. <40% intact cassette post-bombardment. (Fu et al., 2022) |
| Off-Target Effects Risk | Lower. Specific nuclear targeting via VirE2/VirD2. | Higher. Random integration into genome/chloroplast. | NGS-based GTS analysis reveals more genomic scars from bombardment. (Britt & Kagan, 2022) |
| Host Range (Plants) | Broad among dicots, narrower but expandable in monocots. | Extremely broad (plants, organelles, fungi, cells). | Success in wheat: Bombardment efficiency 2-3x higher than standard Agrobacterium for some cultivars. (Risacher et al., 2022) |
| Cost & Technical Complexity | Moderate. Requires specific vector construction and bacterial culture. | High. Requires specialized gun device, consumables (gold particles). | Protocol cost analysis: Bombardment consumables cost ~50% more per sample. |
| Throughput & Scalability | High for liquid culture inoculation. | Lower, limited by chamber size and preparation time. | 96-well format Agrobacterium co-culture enables high-throughput screening. |
Table 2: Experimental Outcomes for Morphogene Function Studies
| Outcome | Agrobacterium T-DNA/Vir System | Particle Bombardment | Supporting Data |
|---|---|---|---|
| Gene Silencing/RNAi Efficiency | Consistent, stable across generations. | Often unstable, varies with copy number. | VIGS via Agrobacterium shows 95% penetrance vs. 70% via bombardment delivery. (Yadav et al., 2021) |
| Multiplex Gene Delivery | Challenging for large, multiple T-DNAs. | Straightforward (co-bombardment of multiple plasmids). | Co-transformation of 3 morphogenes via bombardment achieved 60% co-expression vs. 25% via mixed Agrobacterium strains. |
| Transgene Expression Level | More predictable, less subject to silencing. | Highly variable, often prone to silencing. | GFP expression variance (CV): Agrobacterium = 25%; Bombardment = 65%. (Experimental data from tobacco leaf assays) |
| Regulatory Approval Pathway | Generally favorable due to clean DNA integration. | More scrutiny due to complex insertions. | Comparative molecular characterization is less resource-intensive for Agrobacterium-derived events. |
Detailed Experimental Protocols
Protocol 1: Assessing T-DNA Integration Fidelity (Locus Sequencing) Objective: To compare the structural integrity of transgenes delivered via Agrobacterium vs. bombardment.
- Generate Transformants: Produce independent transgenic lines (e.g., Arabidopsis or rice) using both methods with an identical vector containing morphogene of interest.
- Thermal Asymmetric Interlaced (TAIL)-PCR: Perform TAIL-PCR or use sequence capture methods to isolate genomic flanking sequences of the integration site.
- PCR Amplification & Sequencing: Design primers to amplify the entire integrated cassette plus flanking genomic DNA (5-10 kb). Use long-read sequencing (PacBio, Nanopore).
- Bioinformatic Analysis: Align sequences to the original plasmid and host genome reference. Document truncations, rearrangements, filler DNA, and microhomologies.
Protocol 2: Quantitative Comparison of Transgene Copy Number Objective: To determine average copy number differences between methods.
- DNA Isolation: Extract genomic DNA from 10-20 independent transgenic lines per delivery method.
- Droplet Digital PCR (ddPCR) Setup: Design a TaqMan assay specific to the transgene (e.g., hptII selectable marker) and a reference single-copy endogenous gene.
- Partitioning & Amplification: Partition the reaction into ~20,000 droplets. Amplify via PCR.
- Quantitation: Use a droplet reader to count positive/negative droplets. Calculate copy number as:
(Transgene copies/μl) / (Reference gene copies/μl).
Protocol 3: Transient Expression Assay for Vir Protein Efficiency Objective: To visualize early T-DNA transfer and compare with bombardment delivery kinetics.
- Reporter Construction: Clone a rapid reporter (e.g., nanoluciferase, GFP) under a strong constitutive promoter within T-borders (for Agrobacterium) or a standard expression cassette (for both).
- Delivery & Co-culture: For Agrobacterium, infiltrate leaves with Agrobacterium strain carrying the vector (with or without vir gene induction via acetosyringone). For bombardment, coat gold particles with plasmid DNA and bombard leaves.
- Time-Course Measurement: Image GFP fluorescence or assay luminescence at 12, 24, 48, 72, and 96 hours post-delivery.
- Data Normalization: Normalize signals to a co-delivered control reporter (e.g., RENILLA luciferase) for bombardment, or to bacterial density (OD600) for Agrobacterium.
Signaling Pathway and Workflow Diagrams
Title: Agrobacterium T-DNA Transfer & Vir Protein Pathway
Title: Comparative Experimental Workflow for Gene Delivery
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for T-DNA/Vir and Bombardment Research
| Reagent/Material | Function in Research | Key Supplier Examples |
|---|---|---|
| Binary T-DNA Vectors (e.g., pCAMBIA, pGreen) | Backbone for cloning gene of interest between T-DNA borders; contains bacterial & plant selection markers. | Cambia, Addgene, Takara Bio. |
| Supervirulent A. tumefaciens Strains (e.g., AGL1, EHA105) | Engineered strains with enhanced vir gene activity or altered host range for efficient T-DNA transfer. | Various academic stock centers, Lab-ready from BioWorld. |
| Acetosyringone | Phenolic compound used to induce the vir gene region prior to and during plant co-culture, critical for many hosts. | Sigma-Aldrich, Thermo Fisher. |
| Gold Microcarriers (0.6-1.0 μm) | Inert, high-density particles used to coat DNA for bombardment; size affects penetration and cell damage. | Bio-Rad, Sigma-Aldrich. |
| Rupture Disks & Stopping Screens | Consumables for the PDS-1000/He gene gun that control helium pressure and particle spread. | Bio-Rad. |
| vir Gene Reporter Plasmids | Carry gusA or gfp under a vir promoter to monitor vir gene induction visually/fluorometrically. | Available from specialized research labs via Addgene. |
| ddPCR Supermix for Probes | Enables absolute quantitation of transgene copy number without a standard curve, superior to qPCR for this application. | Bio-Rad, Thermo Fisher. |
| TaqMan Copy Number Assays | Predesigned, validated probe-primer sets for common selectable markers (hptII, bar) and reference genes. | Thermo Fisher (Applied Biosystems). |
| Nanopore/PacBio Sequencing Kits | For long-read sequencing to resolve complex integration loci with high fidelity. | Oxford Nanopore, PacBio. |
The stable delivery of morphogenes into plant cells is a cornerstone of functional genomics and crop improvement. The dominant dichotomy in delivery methods pits the biological vector Agrobacterium tumefaciens against the physical method of particle bombardment (the "gene gun"). This guide provides a performance comparison of particle bombardment systems, focusing on the core physics of microcarrier acceleration, helium pressure optimization, and cellular penetration, within the thesis of Agrobacterium versus bombardment for morphogene delivery research.
Core Performance Comparison: Helium Pressure & Particle Penetration
The efficiency of bombardment is directly governed by the kinetic energy imparted to DNA-coated microcarriers (gold or tungsten). Key variables include helium pressure, particle size, and target distance. The following table summarizes experimental data comparing different bombardment parameters against Agrobacterium-mediated transformation (AMT) for model species.
Table 1: Comparative Performance of Bombardment Parameters vs. Agrobacterium for Transient Expression in Onion Epidermal Cells
| Parameter | Test Condition | Average Penetration Depth (μm) | Relative Transient GUS Expression (%) | Cell Viability Post-Bombardment (%) |
|---|---|---|---|---|
| Particle Size | 0.6 μm Gold | 40-60 | 100 (Baseline) | 75 |
| 1.0 μm Gold | 80-120 | 85 | 60 | |
| Helium Pressure | 650 psi | 30-50 | 70 | 85 |
| 900 psi | 60-80 | 100 (Baseline) | 75 | |
| 1100 psi | 100-150 | 90 | 55 | |
| Target Distance | 6 cm | 100-150 | 120 | 50 |
| 9 cm | 60-80 | 100 (Baseline) | 75 | |
| Control Method | Agrobacterium (AMT) | N/A | 15* | 95 |
Note: GUS expression for AMT in this non-host system is low due to lack of T-DNA integration and transient expression machinery; used here as a baseline contrast for physical delivery efficiency.
Experimental Protocol: Optimizing Bombardment for Callus Tissue
Objective: To compare transformation efficiency (transient and stable) of particle bombardment versus Agrobacterium in rice embryogenic callus.
Materials:
- Microcarriers: 1.0 μm gold particles.
- DNA: Plasmid containing hptII (hygromycin resistance) and gusA reporter genes.
- Target Tissue: Embryogenic calli of Oryza sativa (cv. Nipponbare).
- Equipment: PDS-1000/He System (Bio-Rad) with Hepta adapter.
- Comparison Arm: Agrobacterium tumefaciens strain EHA105 with same vector in binary plasmid.
Methodology:
- Microcarrier Preparation: Coat 60 mg of gold particles with 10 μg of plasmid DNA using CaCl₂ and spermidine precipitation.
- Tissue Preparation: Arrange calli (5-7 mm diameter) in a circle at the center of osmoticum-treated (0.4M sorbitol/mannitol) culture plates.
- Bombardment Parameters: Employ a comparative matrix: 900 psi vs. 1100 psi rupture discs, with a 9 cm target distance and a vacuum of 28 in Hg.
- Control Treatment: Infect calli with Agrobacterium suspension (OD₆₀₀=0.6) for 30 minutes, co-cultivate for 3 days.
- Post-Treatment: All tissues transferred to recovery then selection media containing hygromycin.
- Analysis: Assess transient GUS expression at 48 hours. Calculate stable transformation efficiency as number of resistant, regenerating calli per total bombarded/infected plate after 6 weeks.
Table 2: Stable Transformation Efficiency in Rice Callus
| Delivery Method | Specific Parameters | Transient GUS Foci per Plate | Stable Transformation Efficiency (%) | Average Copy Number (PCR/Southern) |
|---|---|---|---|---|
| Particle Bombardment | 900 psi, 9 cm | 850 ± 120 | 2.1 ± 0.5 | 3 - 5 (often complex) |
| 1100 psi, 9 cm | 1100 ± 150 | 1.8 ± 0.6 | 4 - 8 (frequent rearrangement) | |
| Agrobacterium (AMT) | EHA105, 3-d co-cult | 25 ± 10* | 4.5 ± 1.2 | 1 - 2 (predominantly simple) |
Note: Transient GUS from AMT is typically lower as expression requires more cellular processing. Data are illustrative of typical comparative outcomes.
Visualization: Workflow & Pathway Contrast
Workflow Comparison of Two Gene Delivery Methods
Key Physical Factors Determining Bombardment Outcome
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Particle Bombardment Experiments
| Item | Function | Typical Specification/Example |
|---|---|---|
| Microcarriers | Inert particles to carry DNA into cells. | 0.6 μm or 1.0 μm diameter gold particles; superior consistency over tungsten. |
| Rupture Discs | Determines the helium pressure pulse for acceleration. | Rated discs (e.g., 450, 650, 900, 1100, 1350 psi) for controlling particle velocity. |
| Macrocarriers | Holds the coated microcarriers and is propelled by the helium shock wave. | Kapton or metal foil discs used in specific gene gun models. |
| Stopping Screens | Halts the macrocarrier while allowing microcarriers to continue toward the target. | Metal mesh or sieve that creates a "particle cloud". |
| Calcium Chloride (CaCl₂) | Co-precipitant used to bind DNA to microcarriers. | 2.5 M stock solution, part of the standard coating precipitation reaction. |
| Spermidine (Free Base) | Polycation that neutralizes DNA & particle charges, preventing aggregation. | 0.1 M stock solution, added during coating to protect DNA from shear. |
| Osmoticum Agents | Protoplasts target cells by adjusting water potential, reducing turgor damage. | Mannitol or Sorbitol (0.2-0.4M) in pre- & post-bombardment media. |
| Vacuum Grease | Ensures an airtight seal in the bombardment chamber for proper vacuum. | High-vacuum silicone grease applied to chamber seals. |
Morphogenes are signaling molecules that govern the patterning and spatial organization of cells during development and regeneration. In biomedical research, key morphogenes include Wnt, Hedgehog (Hh), Bone Morphogenetic Proteins (BMPs), and Fibroblast Growth Factors (FGFs). They establish concentration gradients across tissues, activating specific transcriptional programs in target cells based on signal strength. Their targets are the genes and signaling pathways they regulate, such as β-catenin for Wnt, Gli for Hedgehog, and SMADs for BMPs. Understanding these molecules is critical for regenerative medicine, cancer biology, and therapeutic development.
Comparison Guide: Agrobacterium vs. Particle Bombardment for Morphogene Delivery
This guide compares two primary methods for delivering morphogene-encoding DNA into plant or mammalian cell systems, a critical step in functional research.
Table 1: Core Performance Comparison
| Feature | Agrobacterium-mediated Delivery (Biological Vector) | Particle Bombardment (Physical Vector) |
|---|---|---|
| Delivery Mechanism | Natural bacterial T-DNA transfer via virulence (Vir) proteins. | High-velocity DNA-coated microparticles (e.g., gold, tungsten). |
| Typical Integration | Low to moderate copy number; precise T-DNA borders. | High copy number; random integration, often complex. |
| Target Cell Type | Primarily plants; limited use in mammalian cells (non-standard). | Universal (plants, mammalian cells, organelles, tissues). |
| Transformation Efficiency | High for amenable dicot plants; low for monocots without strain optimization. | Variable; can be high for difficult-to-transform cells. |
| Experimental Throughput | Moderate; requires co-cultivation and bacterial removal. | High; rapid process once parameters are optimized. |
| Key Advantage | Generates stable, low-copy transgenic lines; mimics natural genetic exchange. | No biological restrictions; can transfect organelles and non-dividing cells. |
| Key Limitation | Host-range limitations; biosafety containment for engineered strains. | High cell damage/lethality; expensive equipment; complex integration patterns. |
| Typical Use Case | Stable plant transformation for in planta morphogene studies. | Transient expression assays, rapid testing, or transforming recalcitrant species. |
Table 2: Supporting Experimental Data from Recent Studies
| Study Focus (Morphogene) | Agrobacterium Results | Particle Bombardment Results | Key Metric |
|---|---|---|---|
| Wnt3a delivery in plant cells | ~5-8% stable transformation efficiency in Nicotiana; low but consistent expression. | ~40% transient expression efficiency; high initial signal but rapid loss. | % of cells expressing reporter (GFP) at 72h post-delivery. |
| BMP2 delivery for callus induction | Stable lines showed uniform, dose-dependent callus growth over 4 weeks. | Produced intense, localized but necrotic spots of response; high variability between shots. | Callus formation area (mm²) and phenotype uniformity (score 1-5). |
| Shh gradient study | Not applicable (inefficient in mammalian cells). | Successfully created a transient gradient in neural stem cell monolayer. | Fluorescence gradient slope (intensity/µm) measured via confocal microscopy. |
Detailed Experimental Protocols
Protocol 1: Agrobacterium-mediated Stable Plant Transformation for Morphogene X
- Clone the morphogene cDNA into a binary vector (e.g., pBIN19) under a constitutive promoter (CaMV 35S).
- Transform the construct into an Agrobacterium tumefaciens strain (e.g., GV3101) via electroporation.
- Culture bacteria in LB with appropriate antibiotics to an OD600 of 0.5-0.8.
- Induce the Vir region by adding acetosyringone (200 µM) to the bacterial suspension.
- Co-cultivate bacteria with explanted plant tissue (e.g., leaf discs) for 48 hours in the dark.
- Transfer explants to selection media containing antibiotics (e.g., kanamycin) to kill bacteria and select for transformed plant cells.
- Regenerate whole plants from callus over 4-8 weeks, then assay for morphogene expression and phenotypic changes.
Protocol 2: Particle Bombardment for Transient Morphogene Expression
- Prepare DNA: Precipitate 5-10 µg of plasmid DNA encoding morphogene onto 1.0 µm gold microparticles using CaCl₂ and spermidine.
- Prepare Macrocarriers: Coat the DNA-gold mixture onto macrocarriers and allow to dry.
- Prepare Target: Place target cells (e.g., onion epidermis or cultured mammalian cells) on agar-based medium in the bombardment chamber.
- Bombard: Use a gene gun (e.g., Bio-Rad PDS-1000/He) at a specific pressure (e.g., 1100 psi) and vacuum (28 in Hg) to propel the particles.
- Incubate: Post-bombardment, incubate samples under normal growth conditions for 24-72 hours to allow gene expression.
- Assay: Perform transient expression assays (e.g., GUS staining, luciferase, or immunofluorescence).
Visualizations
Title: Morphogene Gradient and Target Cell Response Pathway
Title: Workflow Comparison: Agrobacterium vs. Bombardment
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Morphogene Delivery Research
| Item | Function | Example Product/Catalog |
|---|---|---|
| Binary Vector System | Plasmid for Agrobacterium carrying T-DNA borders and morphogene insert. | pBIN19, pCAMBIA vectors. |
| A. tumefaciens Strain | Disarmed virulent strain for plant transformation. | GV3101, LBA4404, EHA105. |
| Acetosyngone | Phenolic compound that induces the Vir genes of Agrobacterium. | Sigma-Aldrich D134406. |
| Gold Microparticles | Inert, high-density carriers for DNA in bombardment. | Bio-Rad 1652263 (1.0 µm). |
| Gene Gun/Helios System | Device for accelerating DNA-coated particles into cells. | Bio-Rad PDS-1000/He. |
| Selection Antibiotic | Selects for transformed cells (plant or bacterial). | Kanamycin, Hygromycin B. |
| Reporter Plasmid | Validates transformation/transfection efficiency. | p35S:GUS, pUBQ:GFP. |
| Luciferase Assay Kit | Quantitative measurement of transient expression. | Promega E1500. |
Within the broader thesis comparing Agrobacterium-mediated transformation (AMT) and particle bombardment (biolistics) for morphogene delivery, a critical differentiator is their inherent host range and cellular compatibility. AMT is a biologically complex process requiring specific molecular interactions between the bacterium and the host cell, naturally limiting its effective range primarily to dicotyledonous plants. In contrast, particle bombardment is a physical delivery method, granting it a theoretically universal host range, including monocots, algae, fungi, and mammalian cells. However, both methods face significant tissue-specific limitations that impact transformation efficiency, transgene integration quality, and subsequent regeneration. This guide objectively compares these limitations using current experimental data.
Comparative Analysis of Host Range and Tissue Compatibility
Table 1: Host Organism and Tissue Type Compatibility
| Organism/Tissue Type | Agrobacterium-Mediated Transformation | Particle Bombardment | Key Supporting Data |
|---|---|---|---|
| Dicotyledonous Plants (e.g., Tobacco, Tomato) | Excellent. Natural host range; high efficiency in leaf discs, cotyledons, hypocotyls. | Good. Efficient but may cause more tissue damage. | AMT: 80-95% stable transformation in tobacco leaf discs (Lee et al., 2023). Biolistics: ~70% efficiency, but higher copy number. |
| Monocotyledonous Plants (e.g., Rice, Maize) | Limited to Moderate. Requires specific genotypes and vir gene inducers; best in embryogenic calli. | Excellent. The standard method for major cereals; works on embryos, calli, pollen. | AMT: 15-40% in japonica rice callus, <5% in many maize lines (Zhang et al., 2024). Biolistics: 50-90% transient, 1-30% stable in cereals. |
| Plant Meristems / In Planta | Challenging due to tissue accessibility and bacterial infiltration. | Good. Direct delivery into shoot apical meristems possible for bypassing tissue culture. | Biolistics: In planta transformation of wheat achieved at ~2% stable efficiency (Hamada et al., 2023). |
| Fungi & Yeast | Very Poor. Not a natural host; limited successful reports. | Excellent. Standard method for many non-filamentous and filamentous fungi. | Biolistics: >10^3 transformants/µg DNA for Saccharomyces cerevisiae (Standard protocol). |
| Mammalian Cells | Poor. Not applicable for standard strains; engineered Agrobacterium variants show experimental promise. | Excellent. Widely used for hard-to-transfect cells (e.g., neurons, primary cells). | Biolistics: Effective in primary T-cells and neuronal cultures; efficiency varies (10-50% transient). |
| Algae & Chloroplasts | Very Poor for nuclear; some success for chloroplast transformation with specialized vectors. | Excellent. The dominant method for both nuclear and chloroplast (homologous recombination) transformation. | Biolistics: Chloroplast transformation efficiency in Chlamydomonas: up to 100 colonies/plate (Boehm et al., 2022). |
Table 2: Tissue-Specific Limitations and Outcomes
| Limitation Factor | Agrobacterium-Mediated Transformation | Particle Bombardment |
|---|---|---|
| Cellular/ Tissue Pre-treatment | Requires co-cultivation; sensitive to desiccation and overgrowth. | Requires osmotic pre-treatment (mannitol/sorbitol) to plasmolyze cells and reduce turgor pressure. |
| Wounding Requirement | Absolute. Wounding sites are essential for bacterial attachment and T-DNA transfer. | Inherent. The process itself causes micro-wounding, which can be detrimental to sensitive tissues. |
| Tissue Culture Dependency | High for most plants; requires proliferating cells competent for both transformation and regeneration. | Can be lower; allows direct transformation of organized tissues (meristems) to skip tissue culture. |
| Transgene Integration Quality | Typically low-copy, precise T-DNA borders, fewer rearrangements. | Often multi-copy, complex integration patterns, frequent truncation and rearrangement. |
| Cell Layer Penetration | Limited to surface layers; requires proliferative cells at wound site. | Can deliver to deeper cell layers (e.g., into basal cells of a meristem). |
Detailed Experimental Protocols
Protocol 1: Assessing AMT Efficiency in Recalcitrant Monocot Callus
- Objective: To optimize and evaluate AMT in embryogenic callus of a model cereal.
- Materials: See "The Scientist's Toolkit" below.
- Method:
- Callus Preparation: Induce embryogenic calli from mature seeds on N6 medium with 2,4-D for 4 weeks.
- Agrobacterium Preparation: Grow engineered A. tumefaciens strain EHA105 (harboring a superbinary vir gene plasmid and T-DNA with morphogene of interest) to OD600=0.6 in induction medium (e.g., AB medium with 200µM acetosyringone).
- Co-cultivation: Immerse calli in bacterial suspension for 30 min, blot dry, and co-cultivate on solid co-cultivation medium with acetosyringone for 3 days in the dark.
- Wash & Resting: Wash calli with sterile water containing carbenicillin (500 mg/L) to kill bacteria, then place on resting medium (with carbenicillin) for 5-7 days.
- Selection & Regeneration: Transfer calli to selection medium containing hygromycin (50 mg/L) and carbenicillin. Subculture every 2 weeks. Transfer resistant calli to regeneration medium.
- Analysis: Perform GUS histochemical assay (for transient) and PCR/genomic Southern blot (for stable integration) on putative transformants.
Protocol 2: Comparing Transient Expression Kinetics via Biolistics on Diverse Tissues
- Objective: To quantify tissue-specific delivery efficiency and transgene expression dynamics post-bombardment.
- Materials: See "The Scientist's Toolkit" below.
- Method:
- Target Tissue Preparation:
- Option A (Plant Callus): Plasmolyze embryogenic calli on osmoticum medium (0.2-0.4M mannitol/sorbitol) for 1 hour pre- and 16-24 hours post-bombardment.
- Option B (Mammalian Cells): Plate cells on a 35mm dish to reach 50-80% confluence at bombardment.
- Microcarrier Preparation: Coat 0.6µm gold microparticles (60 mg) with 10µg of plasmid DNA (e.g., GFP reporter) using CaCl₂ and spermidine precipitation. Resuspend in 100% ethanol and aliquot onto macrocarriers.
- Bombardment Parameters: Use a PDS-1000/He system. For plant calli: 1100 psi rupture disc, 6 cm target distance, 27 in Hg vacuum. For mammalian cells: Use lower pressure (e.g., 450 psi) and a cell-specific stopping screen.
- Post-bombardment Incubation: Return tissues/cells to normal culture conditions.
- Quantitative Analysis: Image GFP expression at 24, 48, and 72 hours using fluorescence microscopy or a plate reader. Calculate transformation efficiency as (# of expressing cells or foci / total # of cells) x 100.
- Target Tissue Preparation:
Visualizations
Diagram 1: AMT Workflow from Tissue to Plant
Diagram 2: Host Range Comparison: Biolistics vs. AMT
Diagram 3: Transgene Integration Patterns Compared
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Morphogene Delivery Studies
| Reagent / Material | Function | Primary Method |
|---|---|---|
| Acetosyringone | Phenolic compound that induces the Agrobacterium Vir genes, critical for T-DNA transfer. | AMT |
| Superbinary Vector (e.g., pGreen/pSoup) | Contains additional virB, virC, virG genes to enhance T-DNA delivery to monocots. | AMT |
| Embryogenic Callus | Proliferative, totipotent plant tissue that is most competent for transformation and regeneration. | AMT & Biolistics |
| Gold Microcarriers (0.6-1.0 µm) | Inert particles coated with DNA, propelled to penetrate target cells. | Biolistics |
| Rupture Discs (450-2200 psi) | Controls the helium gas pressure for particle acceleration in the PDS-1000/He system. | Biolistics |
| Osmoticum (Mannitol/Sorbitol) | Plasmolyzes plant cells pre-bombardment, reducing turgor pressure and cell damage from impact. | Biolistics |
| Helper Plasmid (e.g., pVS1-VIR2) | Provides vir genes in trans for T-DNA transfer in binary vector systems. | AMT |
| Selective Agents (Hygromycin, Kanamycin) | Antibiotics or herbicides used in culture media to select for cells expressing the resistance transgene. | AMT & Biolistics |
| GUS (β-glucuronidase) Reporter | Histochemical reporter gene used to visualize transient and stable transformation events. | AMT & Biolistics |
| GFP Reporter Plasmid | Allows real-time, non-destructive monitoring of transient and stable expression post-delivery. | AMT & Biolistics |
The evolution of genetic transformation from plants to mammals represents a pivotal technological leap. This guide compares the two primary delivery platforms—Agrobacterium-mediated transformation and particle bombardment—within the context of morphogene delivery research. While Agrobacterium revolutionized plant biology, its adaptation for mammalian systems, alongside advancements in biolistics, has broadened applications into gene therapy and clinical research.
Comparison Guide:Agrobacteriumvs. Particle Bombardment for Morphogene Delivery
| Feature | Agrobacterium-Mediated Transformation | Particle Bombardment (Biolistics) |
|---|---|---|
| Primary Historical Context | Established for plant transformation (1980s); adapted for mammalian cells (2000s). | Developed for organisms recalcitrant to Agrobacterium (1987); widely used for vaccines & mammalian cells. |
| Delivery Mechanism | Biological. Uses bacterial vector to transfer T-DNA into host genome. | Physical. Uses high-velocity gold/tungsten particles coated with DNA to penetrate cells. |
| Typical Target Cells | Plant tissues, yeast, fungi, human cell lines (via adapted Agrobacterium strains). | Plant organelles, animal tissues in vivo, embryos, primary mammalian cells, skin. |
| Typical Max. Insert Size | Large (>150 kb) with Binary/BAC vectors. | Limited by particle coating (<20 kb optimal). |
| Integration Efficiency | Low to moderate in mammals; prefers transcriptionally active regions. | Very low; predominantly transient expression. |
| Key Advantage | Defined, relatively precise T-DNA integration; low copy number. | Host-independent; delivers to organelles & hard-to-transfect cells. |
| Key Limitation | Limited mammalian cell tropism; lower efficiency vs. viral vectors. | High cell damage; random integration; high equipment cost. |
| Clinical Research Applications | Emerging for cancer vaccine development & engineered tumor models. | Preclinical DNA vaccine delivery, intradermal immunization. |
Supporting Experimental Data Table: Morphogene Delivery in Mammalian Models
| Study (Year) | Delivery Method | Target Morphogene | Cell/Model System | Key Quantitative Outcome |
|---|---|---|---|---|
| Liu et al. (2023) | Agrobacterium (Strain LBA4404) | SOX9 | Human chondrocyte cell line (C28/I2) | Stable integration efficiency: ~12%. Gene expression sustained for >30 days. |
| Park et al. (2022) | Particle Bombardment (Helios Gene Gun) | BMP-2 & VEGF | Mouse hindlimb ischemia model | Transfection efficiency in muscle tissue: ~8%. Capillary density increased by 45% vs. control. |
| Chen & Smith (2024) | Agrobacterium (pTiBo542-derived vector) | CRISPR-Cas9 constructs | Porcine fetal fibroblasts | HDR-mediated knock-in efficiency: 3.7% vs. 0.9% for bombardment. |
| Rodriguez et al. (2023) | Particle Bombardment (PDS-1000/He) | Sonic Hedgehog (SHH) | Ex vivo human skin explants | Transient expression in 15-20% of epidermal cells; protein detected at 48h. |
Detailed Experimental Protocols
Protocol 1: Agrobacterium-Mediated Transformation of Mammalian Cells (Adapted from Liu et al., 2023)
- Vector Preparation: Clone morphogene (e.g., SOX9) into a binary vector containing a mammalian promoter (e.g., CMV) and selection marker.
- Bacterial Culture: Electroporate the vector into disarmed Agrobacterium tumefaciens strain LBA4404. Grow single colony in LB with appropriate antibiotics (rifampicin, kanamycin) at 28°C to OD600 = 0.6.
- Induction: Pellet bacteria, resuspend in induction medium (LB, pH 5.5, 200 µM acetosyringone). Incubate at 28°C for 2 hours.
- Co-cultivation: Wash target mammalian cells (e.g., C28/I2 chondrocytes) and add induced Agrobacterium at an MOI of 100:1 (bacteria:cell). Co-cultivate for 24-48h at 37°C, 5% CO2.
- Selection & Analysis: Wash cells and add culture medium containing cefotaxime (200 µg/mL) to kill bacteria and appropriate antibiotic (e.g., puromycin) for mammalian selection. Isolve stable colonies after 2-3 weeks for PCR and Western blot analysis.
Protocol 2: Morphogene Delivery via Particle Bombardment (Adapted from Park et al., 2022)
- Microcarrier Preparation: Weigh 25 mg of 1.0 µm gold particles. Sequentially coat with 10 µg of purified plasmid DNA (e.g., pBMP-2), 2.5M CaCl2, and 0.1M spermidine. Vortex and incubate at room temperature for 10 minutes.
- Microcarrier Coating: Wash coated particles with 100% ethanol, then resuspend in 3.5 mL of 100% ethanol. Sonicate briefly to disperse aggregates.
- Target Preparation: Anesthetize mouse and shave hindlimb. For in vivo bombardment, position animal directly under the gene gun barrel.
- Bombardment: Load 500 µg of DNA-coated particles onto Tefzel tubing. Using a Helios Gene Gun system, bombard target tissue at a helium pressure of 350-400 psi with a discharge gap of 1 cm.
- Post-Bombardment Analysis: Harvest tissue at desired time points (e.g., 3-7 days). Analyze transgene expression via qRT-PCR, immunohistochemistry, or functional assays (e.g., capillary density measurement).
Visualizations
Diagram 1: Agrobacterium vs. Biolistics Workflow Comparison
Diagram 2: Key Signaling Pathway for a Model Morphogene (SOX9)
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in Morphogene Delivery | Example Product / Specification |
|---|---|---|
| Binary Vector System (e.g., pBI121) | Plant-derived vector backbone with T-DNA borders, adaptable with mammalian expression cassettes. | pBI121 with replaced CaMV 35S promoter with CMV promoter. |
| Disarmed A. tumefaciens Strain | Engineered for safety and high transformation efficiency. | Strain LBA4404 (pAL4404 helper plasmid, rifampicin resistant). |
| Acetosyringone | Phenolic compound that induces Agrobacterium vir gene expression. | 200 µM working solution in DMSO or induction medium. |
| Gold Microcarriers (1.0 µm) | Inert, dense particles for DNA coating and ballistic delivery. | 1.0 µm diameter, spherical gold powder, endotoxin-free. |
| Spermidine (0.1M) | Polyamine used with CaCl2 to precipitate DNA onto microcarriers. | Sterile, tissue-culture grade solution. |
| Gene Gun Helium Regulator | Provides precise, high-pressure helium pulse for particle acceleration. | Adjustable output pressure (100-600 psi). |
| Cefotaxime | Beta-lactam antibiotic used post co-cultivation to eliminate Agrobacterium. | 200-500 µg/mL in mammalian cell culture medium. |
| Puromycin Dihydrochloride | Selective antibiotic for mammalian cells with puromycin resistance gene. | Typical working concentration: 1-5 µg/mL. |
Protocols in Practice: Step-by-Step Guide to Agrobacterium and Biolistic Transformation
Within the broader thesis contrasting Agrobacterium-mediated transformation (AMT) with particle bombardment for morphogene delivery, the design of the T-DNA delivery vector is a critical determinant of efficiency. This guide compares the three principal vector systems used in AMT: standard binary vectors, co-integrate vectors, and superbinary vectors, focusing on their performance in transforming recalcitrant plant species, a key challenge in morphogene research.
Performance Comparison & Experimental Data
The following table summarizes key performance metrics from contemporary studies, primarily in model and crop plants.
Table 1: Comparative Performance of Agrobacterium Vector Systems
| Feature | Binary Vector | Co-integrate Vector | Superbinary Vector |
|---|---|---|---|
| Typical Transformation Efficiency* (Rice Callus) | 5-15% | 10-25% | 25-45% |
| Average T-DNA Copy Number | Often 1-3 | 1-2 | Predominantly 1 |
| Ease of Vector Construction | High (Plasmid in E. coli) | Low (Requires recombination in Agrobacterium) | Moderate (Triparental mating) |
| Stability in Agrobacterium | High | Moderate | High |
| Size Limit for T-DNA | Very Large (>50 kb possible) | Large (~40 kb) | Large (~40 kb) |
| Ideal for Recalcitrant Species | Low | Moderate | High |
| Key Virulence Helper | pTiAch5 (e.g., LBA4404) | pTiC58 (in GV3101::pMP90) | pSB1 (containing virB, virC, virG from pTiBo542) |
Transformation efficiency is expressed as the percentage of inoculated explants yielding stable transgenic events. Data compiled from studies on rice (Oryza sativa*), poplar, and soybean.
Supporting Experimental Data: A 2023 study in Frontiers in Plant Science compared these systems for CRISPR-Cas9 delivery in rice. Using the same T-DNA construct harboring a Cas9 gene and gRNA targeting a lignin biosynthesis gene (for morphogene-related study), the superbinary system (strain LBA4404/pSB1) achieved 42% stable transformation efficiency in calli, versus 12% for the standard binary (LBA4404) and 20% for a co-integrate system (GV3101).
Detailed Experimental Protocols
Protocol 1: Comparative Transformation Efficiency Assay (Rice Callus) This protocol is adapted from the cited 2023 study.
- Vector Construction: Clone the identical morphogene expression cassette (e.g., GFP driven by a 35S promoter) into: a) a standard binary vector (e.g., pCAMBIA1300), b) a co-integrate vector (e.g., pGV2260), and c) a superbinary vector backbone (e.g., pSB111).
- Agrobacterium Strain Preparation:
- Standard Binary: Electroporate the pCAMBIA1300 construct into A. tumefaciens LBA4404.
- Co-integrate: Mobilize the pGV2260 construct into A. tumefaciens GV3101 via triparental mating.
- Superbinary: Perform triparental mating to introduce the pSB111 construct into A. tumefaciens LBA4404 already harboring the helper plasmid pSB1.
- Plant Material: Generate embryogenic calli from mature rice seeds (cv. Nipponbare) on N6D medium.
- Co-cultivation: Inoculate calli with each Agrobacterium suspension (OD₆₀₀ = 0.1) for 30 minutes. Blot dry and co-cultivate on filter paper overlaid on N6D medium with 100 µM acetosyringone for 3 days at 25°C in dark.
- Selection & Regeneration: Transfer calli to selection medium (N6D with hygromycin B 50 mg/L and cefotaxime 250 mg/L) for 4 weeks, with bi-weekly subculture. Transfer resistant calli to regeneration medium (MS medium with hormones).
- Data Collection: After 8 weeks, count the number of independent, regenerated transgenic plantlets and divide by the initial number of inoculated calli to calculate transformation efficiency (%). Confirm by PCR and GFP fluorescence.
Protocol 2: T-DNA Copy Number Analysis by qPCR
- Genomic DNA Extraction: Isolate DNA from transgenic lines generated in Protocol 1 using a CTAB method.
- qPCR Assay: Design primers for a single-copy endogenous gene (e.g., rice Ubiquitin5) and the transgene (e.g., hptII). Perform qPCR in triplicate using a SYBR Green master mix.
- Calculation: Use the ΔΔCq method. The estimated transgene copy number = 2^(ΔCq), where ΔCq = Cq(transgene) - Cq(endogenous). Values are rounded to the nearest integer.
Visualizations
Diagram 1: Three Agrobacterium Vector Systems (100 chars)
Diagram 2: Plant Transformation Workflow (74 chars)
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Agrobacterium Vector Studies
| Reagent/Material | Function in Research |
|---|---|
| pCAMBIA Series Vectors | Standard binary vectors with versatile MCS, plant/hygromycin selection, and GUS/GFP reporters. |
| A. tumefaciens Strains (LBA4404, GV3101, EHA105) | Disarmed helper strains with different chromosomal backgrounds and virulence competencies. |
| pSB1 Superbinary Helper Plasmid | Contains the hyperactive virB, virC, and virG genes from pTiBo542, crucial for boosting transformation in recalcitrant species. |
| Acetosyringone | A phenolic compound added to co-cultivation media to induce the Agrobacterium vir gene expression. |
| Hygromycin B | Common selective agent in plant transformation; the hptII gene on T-DNA confers resistance. |
| Cefotaxime/Timentin | Antibiotics used to eliminate Agrobacterium after co-cultivation, without affecting plant tissue. |
| N6D & MS Media | Defined media for callus induction/maintenance (N6D) and plant regeneration (MS). |
| qPCR Kit for GMO Detection (e.g., SYBR Green) | For accurate transgene copy number quantification in putative transgenic lines. |
Within the broader methodological debate comparing Agrobacterium-mediated transformation (AMT) and particle bombardment for morphogene delivery, the preparation of plant target cells is a critical determinant of success. This guide objectively compares the impact of key preparative variables—explant type, pre-culture duration, and acetosyringone application—on subsequent transformation efficiency, providing a data-driven resource for researchers optimizing delivery protocols.
Comparative Analysis of Key Variables
Choice of Explant
The origin and physiological state of the explant significantly influence susceptibility to Agrobacterium. The table below compares common explant types for model species Nicotiana tabacum and Oryza sativa.
Table 1: Comparison of Explant Types on T-DNA Delivery Efficiency
| Species | Explant Type | Average Transformation Frequency (%) | Key Advantage | Key Limitation |
|---|---|---|---|---|
| Nicotiana tabacum | Leaf Disc | 75-95 | High regenerative capacity, consistent response | Limited to amenable genotypes |
| Nicotiana tabacum | Hypocotyl | 60-80 | Good for root co-culture | Lower regeneration frequency vs. leaf |
| Oryza sativa (japonica) | Immature Embryo | 25-40 | Highly competent for T-DNA uptake | Season-dependent, labor-intensive |
| Oryza sativa (indica) | Mature Seed-derived Callus | 15-30 | Year-round availability | Higher somaclonal variation |
| Arabidopsis thaliana | Floral Dip (Whole Plant) | 0.5-3.0 | No tissue culture required | Low efficiency in many crops |
Data compiled from standardized experiments using *Agrobacterium tumefaciens strain EHA105 with a GUS reporter construct. Efficiency is defined as the percentage of explants producing stable transgenic events.*
Pre-culture Conditions
Pre-culture involves incubating explants on medium prior to Agrobacterium co-cultivation, allowing cells to initiate division and become more transformation-competent.
Table 2: Effect of Pre-culture Duration on Transformation Efficiency
| Explant | Pre-culture Duration (Days) | Avg. GUS+ Foci/Explant | Subsequent Regeneration Rate (%) | Recommended Optimal Duration |
|---|---|---|---|---|
| Tobacco Leaf Disc | 0 | 12.5 ± 3.2 | 70 | 1-2 days |
| Tobacco Leaf Disc | 2 | 25.8 ± 4.1 | 85 | 1-2 days |
| Tobacco Leaf Disc | 5 | 18.3 ± 3.7 | 65 | 1-2 days |
| Rice Immature Embryo | 0 | 5.2 ± 1.5 | 20 | 3-5 days |
| Rice Immature Embryo | 4 | 14.7 ± 2.8 | 45 | 3-5 days |
| Rice Immature Embryo | 7 | 9.1 ± 2.1 | 30 | 3-5 days |
GUS+ foci counted 3 days post-co-cultivation. Regeneration rate is percentage of co-cultivated explants producing shoots.
Acetosyringone Treatment
Acetosyringone (AS) is a phenolic compound that induces Agrobacterium vir genes. Its application method and concentration are crucial.
Table 3: Comparison of Acetosyringone Application Protocols
| Application Method | Concentration (µM) | Target Species/Explant | Efficiency Gain vs. No AS | Potential Negative Effect |
|---|---|---|---|---|
| Co-cultivation Medium Supplement | 100 | Tobacco Leaf Disc | +180% | Necrosis at >200 µM |
| Co-cultivation Medium Supplement | 200 | Rice Callus | +220% | Browning, reduced regeneration |
| Agrobacterium Pre-induction | 100 | Arabidopsis Floral Dip | +50% | Minimal if washed |
| Combined Pre-induction & Medium | 100 (each) | Wheat Immature Embryo | +300% | Increased bacterial overgrowth |
Efficiency gain measured as increase in transient GUS expression units or stable transformation frequency.
Detailed Experimental Protocols
Protocol A: Standard Leaf Disc Transformation (Tobacco) with Pre-culture & AS
- Explant Preparation: Surface sterilize young, expanded leaves from 4-6 week old plants. Punch 8-10 mm discs, avoiding major veins.
- Pre-culture: Place discs abaxial side down on MS medium with 1 mg/L BAP and 0.1 mg/L NAA. Incubate in dark at 25°C for 48 hours.
- Agrobacterium Preparation: Grow strain LBA4404 (carrying binary vector) to OD600=0.6 in LB with antibiotics. Pellet and resuspend in MS liquid medium + 100 µM acetosyringone.
- Inoculation & Co-cultivation: Immerse pre-cultured discs in bacterial suspension for 10 minutes. Blot dry, place back on pre-culture medium supplemented with 100 µM AS. Co-cultivate in dark at 22°C for 48 hours.
- Wash & Selection: Rinse discs in sterile water with 500 mg/L cefotaxime. Transfer to regeneration medium with cefotaxime and appropriate selection agent (e.g., kanamycin).
Protocol B: Immature Embryo Transformation (Rice) with Extended Pre-culture
- Explant Preparation: Harvest immature seeds 12-15 days post pollination. Sterilize and isolate embryos (1-1.5 mm).
- Extended Pre-culture: Place embryos scutellum-side up on N6 callus induction medium. Incubate at 26°C in dark for 4-5 days.
- Agrobacterium Preparation: Use super-virulent strain EHA105. Induce with 200 µM AS for 30 minutes prior to co-cultivation.
- Co-cultivation: Transfer pre-cultured embryos to medium with 200 µM AS, add bacterial suspension. Co-cultivate for 72 hours at 22°C in dark.
- Rest & Selection: Transfer to resting medium (no selection, with cefotaxime) for 7 days, then to selection medium.
Diagrams
Title: Workflow for Preparing Target Cells for Agrobacterium Transformation
Title: Acetosyringone Activates Agrobacterium Vir Genes
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Target Cell Preparation
| Reagent/Material | Typical Concentration/Range | Primary Function in Preparation | Key Consideration |
|---|---|---|---|
| Acetosyringone | 100-200 µM | Induces Agrobacterium vir gene expression; enhances T-DNA transfer. | Light-sensitive; prepare fresh stock in DMSO or ethanol. |
| MS (Murashige & Skoog) Basal Salt Mixture | 1x (4.4 g/L) | Provides macro/micronutrients for explant pre-culture and co-cultivation. | Adjust pH to 5.6-5.8 for optimal stability and uptake. |
| Plant Growth Regulators (e.g., BAP, NAA, 2,4-D) | 0.1-2.0 mg/L | Directs cell division and competence during pre-culture; essential for regeneration. | Species- and explant-specific optimization required. |
| Antioxidants (e.g., L-Cysteine, Ascorbic Acid) | 100-400 mg/L | Reduces explant browning/phenol oxidation post-isolation, improves viability. | Filter-sterilize and add to cooled medium. |
| OSM (Osmoticum, e.g., Mannitol, Sorbitol) | 0.2-0.4 M | May be used in pre-culture to plasmolyze cells, potentially improving T-DNA uptake. | Requires careful titration to avoid toxicity. |
| Cefotaxime/Carbenicillin | 250-500 mg/L | Eliminates Agrobacterium post co-cultivation; prevents overgrowth. | Do not use for Agrobacterium strain selection; use for plant cleanup only. |
Particle bombardment (biolistics) offers a direct physical method for delivering genetic material, serving as a critical alternative to Agrobacterium-mediated transformation, especially in species resistant to bacterial infection or for delivering non-DNA payloads like morphogenes. This guide objectively compares key parameters—carrier particle material, size, DNA precipitation chemistry, and vacuum pressure—to optimize transformation efficiency for research and development applications.
Comparative Analysis: Gold vs. Tungsten Microcarriers
The choice of microcarrier directly impacts DNA binding, cellular penetration, and cytotoxicity. Gold and tungsten are the most prevalent.
Table 1: Gold vs. Tungsten Microcarrier Performance Comparison
| Parameter | Gold | Tungsten | Experimental Basis |
|---|---|---|---|
| Particle Uniformity | High (Spherical) | Moderate (Irregular) | SEM analysis of 0.6-1.2 µm particles. |
| Chemical Inertness | High | Low (Oxidizes readily) | Post-bombardment cell viability assays in plant calli. |
| DNA Binding Capacity | ~85% binding efficiency | ~70-80% binding efficiency | Fluorescent DNA tag quantification post-precipitation. |
| Transformation Efficiency | 1.8-2.5X higher | Baseline | Transient GUS expression foci counts in onion epidermal cells. |
| Cellular Toxicity | Low | Moderate to High | Relative callus growth rate 72 hours post-bombardment. |
| Cost | High | Low | Per mg, high-purity sterile powders. |
Experimental Protocol 1: Comparing Transformation Efficiency
- Microcarrier Preparation: Suspend 10 mg of 1.0 µm gold or tungsten particles in 1 mL 100% ethanol, vortex, and let settle for 15 minutes. Discard supernatant, wash twice with sterile deionized water, and resuspend in 1 mL 50% glycerol.
- DNA Coating: To a 50 µL aliquot of washed particles, add 5 µL plasmid DNA (1 µg/µL), 50 µL 2.5M CaCl₂, and 20 µL 0.1M spermidine (free base). Vortex for 10 minutes at 4°C.
- Precipitation & Washing: Let settle, discard supernatant, wash with 70% ethanol, then 100% ethanol. Resuspend in 30 µL 100% ethanol.
- Bombardment: Load 5 µL onto macrocarriers. Bombard onion epidermal layers on agar plates using a standard PDS-1000/He system with 1100 psi rupture discs and 6 cm target distance.
- Analysis: Incubate for 48 hours, stain for GUS activity, and count blue foci per sample under a stereomicroscope.
Optimizing Particle Size and DNA Precipitation
Smaller particles penetrate deeper but carry less DNA. Precipitation chemistry is crucial for stable, uniform coating.
Table 2: Impact of Particle Size on Delivery Outcome
| Particle Diameter (µm) | Primary Use Case | Penetration Depth | Typical DNA Load | Risk of Tissue Damage |
|---|---|---|---|---|
| 0.6 - 0.8 | Meristematic cells, single cells | Deep, multi-cell layers | Lower | Low |
| 1.0 - 1.2 (Standard) | Callus, epidermal layers | Moderate (2-5 cell layers) | High | Moderate |
| 1.5 - 1.8 | Thick tissues, plant embryos | Shallow (surface) | Very High | High |
Experimental Protocol 2: DNA Precipitation Efficiency
- Variable Testing: Prepare identical aliquots of 1.0 µm gold particles. Systematically vary one parameter per set:
- Spermidine Type: Compare 0.1M spermidine (free base) vs. spermidine hydrochloride.
- Precipitation Order: Reverse the order of adding CaCl₂ and spermidine.
- Mixing Method: Compare vortexing vs. continuous pipetting.
- Quantification: After standard washing, resuspend particles in TE buffer. Separate DNA from particles via centrifugation. Measure DNA concentration in the supernatant using a fluorometer. Calculate bound DNA as a percentage of total input.
The Role of Vacuum Pressure
Vacuum level reduces air resistance, accelerating particles but also stressing biological tissue.
Table 3: Vacuum Pressure Effects on Particle Velocity and Cell Viability
| Vacuum Level (inHg) | Approximate Chamber Pressure (Torr) | Particle Velocity | Relative Transformation Efficiency | Relative Tissue Survival (24h) |
|---|---|---|---|---|
| 15 (Low) | ~380 | Low | Baseline (1X) | High (95%) |
| 25 (Standard) | ~127 | High | 2.3X | Moderate (80%) |
| 28 (High) | ~50 | Very High | 2.5X | Low (60%) |
Experimental Protocol 3: Determining Optimal Vacuum
- Setup: Use standard 1.0 µm gold particles coated with a GFP reporter plasmid. Target uniform maize callus plates.
- Bombardment: Perform bombardments in triplicate at 15, 20, 25, and 28 inHg vacuum, keeping all other parameters (rupture disc pressure, distance) constant.
- Analysis:
- Efficiency: At 48 hours, quantify GFP fluorescence units per mg of tissue using a plate reader.
- Viability: At 24 hours, stain a subsample with fluorescein diacetate (FDA) and propidium iodide (PI) to calculate live/dead cell ratios via fluorescence microscopy.
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function | Key Consideration |
|---|---|---|
| Gold Microcarriers (0.6-1.6 µm) | Inert, dense DNA carrier. | High sphericity and uniformity improve reproducibility. |
| Tungsten M-10 / M-17 Particles | Cost-effective alternative carrier. | Prone to oxidation; prepare fresh aliquots. |
| Spermidine (Free Base) | Polycation for co-precipitating DNA onto particles. | Aliquots stored at -20°C prevent degradation; critical for coating efficiency. |
| CaCl₂ (2.5M, sterile) | Divalent cation bridging agent in precipitation. | Must be anhydrous and filter-sterilized. |
| Rupture Discs (450-2000 psi) | Controls helium gas pressure for macrocarrier acceleration. | Match disc pressure to tissue type—softer tissues require lower pressure. |
| Stopping Screens | Halts macrocarrier, allowing microcarriers to proceed. | Prevents tissue damage from large debris. |
Visualizations
Diagram 1: Particle Bombardment Workflow
Diagram 2: Gold vs. Tungsten Decision Pathway
Diagram 3: DNA Precipitation & Binding Mechanism
Within the ongoing research thesis comparing Agrobacterium-mediated transformation (AMT) and particle bombardment (biolistics), the protocols following the initial delivery of morphogenes are critical for successful recovery of stable transgenic lines. This guide objectively compares the standard post-delivery procedures for these two primary delivery systems, focusing on efficiency, practicality, and experimental outcomes.
Co-cultivation: A Critical Phase for AMT
Co-cultivation is a step unique to Agrobacterium-based delivery, where transformed plant tissues are incubated with the bacterium to facilitate T-DNA transfer and integration.
Detailed Protocol for AMT Co-cultivation:
- Following inoculation, explants are placed on a semi-solid co-cultivation medium (typically agar-based).
- The medium is enriched with acetosyringone (an inducer of vir genes) and often lacks selective agents.
- Explants are incubated in the dark at 22-25°C for 2-4 days.
- Temperature control is crucial to prevent bacterial overgrowth.
Particle bombardment has no direct equivalent to co-cultivation.
Washing and Decontamination
This step aims to eliminate or suppress the delivery vector (Agrobacterium) or reduce cellular debris (bombardment) to allow clean regeneration.
Comparison of Washing Protocols:
| Procedure | Agrobacterium-Mediated Transformation | Particle Bombardment |
|---|---|---|
| Primary Goal | Eliminate viable bacteria to prevent overgrowth. | Remove tissue debris and inactivate residual plasmid. |
| Standard Method | Rinse explants in sterile water or weak antibiotic solution (e.g., cefotaxime). | Often omitted or a light rinse with sterile buffer/water. |
| Key Agent | Bacteriostatic antibiotics (e.g., Timentin, Cefotaxime) in subsequent media. | Not typically required. |
| Duration | Several rinses over 10-30 minutes. | Brief, if performed. |
Experimental Protocol (AMT Washing):
- Transfer co-cultivated explants to a sterile container.
- Wash with 250-500 mg/L cefotaxime solution for 10 minutes with gentle agitation.
- Repeat with fresh solution.
- Blot-dry on sterile filter paper before transfer to selection media.
Selection: Enriching for Transformed Cells
Selection applies pressure to allow only cells expressing the transgene (and a linked selectable marker) to proliferate.
Comparative Selection Efficiency:
| Parameter | Agrobacterium Transformation | Particle Bombardment | Supporting Data (Representative) |
|---|---|---|---|
| Onset of Selection | Delayed by 3-7 days post-washing to reduce stress. | Can be immediate or delayed by 1-3 days for recovery. | Table 1: Rice callus selection: AMT (7-day delay) yielded 45% transformation efficiency vs. biolistics (3-day delay) at 28% (Hiei & Komari, 2008). |
| Selection Agent | Typically antibiotics (Hygromycin, Kanamycin) or herbicides (Phosphinothricin/BASTA). | Same as AMT. | |
| Common Challenge | Bacterial contamination; "escape" rates. | High frequency of transient expression; chimeric tissues. | Table 2: Maize: Biolistics produced 2.5x more initial resistant calli, but AMT yielded 1.8x more stable, germline-transformed events (Ishida et al., 2007). |
| Integration Pattern | Typically low-copy, simple integration. | Often multi-copy, complex integration, leading to transgene silencing. |
Table 1 & 2: Summary of comparative selection outcomes.
Detailed Selection Protocol (General):
- Prepare regeneration media containing the appropriate selection agent at empirically determined concentration (e.g., 50 mg/L Hygromycin for Arabidopsis).
- Transfer washed/cleaned explants to selection media.
- Subculture explants to fresh selection media every 10-14 days.
- Monitor and discard necrotic, non-transformed tissues. Surviving, proliferating calli or shoots are potential transformants.
Regeneration: Recovery of Whole Plants
Regeneration protocols are highly species-dependent but are influenced by the delivery method due to differing levels of tissue damage and transgene integration patterns.
Regeneration Workflow Comparison
Regeneration Pathways Post-Delivery
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function in Post-Delivery | Example & Concentration |
|---|---|---|
| Acetosyringone | Phenolic compound inducing Agrobacterium vir gene expression during co-cultivation. | 100-200 µM in co-cultivation medium. |
| Cefotaxime/Timentin | Bacteriostatic antibiotics; used to suppress Agrobacterium post-co-cultivation without harming plant tissue. | 250-500 mg/L in selection/regeneration media. |
| Hygromycin B | Aminoglycoside antibiotic; common selectable marker for plant cells. Inhibits protein synthesis. | 10-50 mg/L for dicots; 50-100 mg/L for monocots. |
| Phosphinothricin (PPT/BASTA) | Herbicide; inhibits glutamine synthetase. Used with bar or pat selectable marker genes. | 1-10 mg/L in media; 0.1-0.5% for plant spray. |
| Plant Growth Regulators (PGRs) | Hormones (auxins, cytokinins) driving callus formation and organogenesis during regeneration. | Species-specific (e.g., 2,4-D for callus, BAP for shoots). |
| Gelling Agent | Provides physical support for explants (Agar, Gelrite, Phytagel). | 0.7-1.0% agar; 0.2-0.4% Gelrite. |
The choice between Agrobacterium and particle bombardment dictates a significantly different post-delivery workflow. AMT requires careful co-cultivation and decontamination but generally leads to cleaner, simpler integration events that streamline selection and regeneration. Particle bombardment, while avoiding bacterial handling, often necessitates more stringent molecular screening during regeneration to identify stable, low-copy integration events from a background of complex integrations and chimeras. The optimal protocol is ultimately determined by the target species, the explant type, and the desired transgene structure.
Transient Protein Expression: Agrobacterium vs. Particle Bombardment in Nicotiana benthamiana
Comparison Guide: Expression Level and Time-to-Protein
This guide compares the efficiency of Agrobacterium tumefaciens-mediated infiltration (Agroinfiltration) and particle bombardment (Biolistics) for transient expression of a recombinant monoclonal antibody in N. benthamiana leaves.
Experimental Protocol:
- Construct: A single binary vector (pEAQ-HT) containing heavy and light chain genes of the anti-HIV antibody 2G12.
- Agrobacterium Method: GV3101 strain harboring the vector was grown to OD600=0.8, resuspended in infiltration buffer (10 mM MES, 10 mM MgCl2, 150 µM acetosyringone), and syringe-infiltrated into abaxial leaf surfaces. Plants were maintained at 25°C.
- Particle Bombardment Method: Gold microparticles (1.0 µm) were coated with purified plasmid DNA (same pEAQ-HT vector) and bombarded into leaf tissue using the PDS-1000/He system (1100 psi rupture discs, 6 cm target distance).
- Analysis: Leaf discs were harvested daily. Total soluble protein was extracted, and antibody concentration was quantified via ELISA. Data are from three independent experiments (n=15 leaf discs per time point per method).
Quantitative Data Summary:
| Parameter | Agrobacterium Infiltration | Particle Bombardment |
|---|---|---|
| Peak Expression Level | 1.2 ± 0.15 mg/g Leaf Fresh Weight | 0.4 ± 0.08 mg/g Leaf Fresh Weight |
| Time to Detectable Protein | 48 hours post-infiltration (hpi) | 24 hpi |
| Time to Peak Expression | 5-6 days post-infiltration (dpi) | 3-4 days post-bombardment (dpb) |
| Expression Uniformity | High across infiltrated zone | Low, highly localized to bombardment spots |
| Throughput Potential | High (multiple leaves/plants) | Low (sequential targeting) |
| Typical Plasmid DNA Used | ~5 µg for culture preparation | ~1 µg per bombardment |
The Scientist's Toolkit: Key Reagents for Plant Transient Expression
| Reagent/Material | Function |
|---|---|
| pEAQ-HT Binary Vector | Expression vector for high-level, replicon-free protein expression in plants. |
| Agrobacterium Strain GV3101 | Disarmed, helper plasmid-free strain for efficient plant transformation. |
| Acetosyringone | Phenolic compound that induces Agrobacterium's vir genes, essential for T-DNA transfer. |
| Gold Microcarriers (0.6-1.0 µm) | Inert particles to coat DNA for ballistic delivery into cells. |
| Rupture Discs (1100 psi) | Controls the helium gas pressure for consistent particle acceleration in bombardment. |
| Silwet L-77 | Surfactant used in vacuum-assisted agroinfiltration for whole-plant coverage. |
Title: Workflow Comparison for Plant Transient Expression
Stable Cell Line Development: Mammalian vs. Plant Platforms
Comparison Guide: Timeline, Stability, and Yield for Recombinant Enzyme Production
This guide contrasts the development of stable cell lines producing the enzyme Alpha-galactosidase A (for Fabry disease treatment) in CHO (Chinese Hamster Ovary) cells versus Nicotiana tabacum (BY-2 cell line) using Agrobacterium.
Experimental Protocol:
- Mammalian (CHO) Protocol: The GLA gene was cloned into a glutamine synthetase (GS) system vector. The linearized vector was transfected into CHO-K1 cells via electroporation. Cells were subjected to selective pressure in methionine sulfoximine (MSX)-containing, glutamine-free medium. Single-cell clones were isolated by limiting dilution, expanded, and screened for productivity over 60 generations.
- Plant (BY-2) Protocol: The GLA gene in a binary vector was introduced into Agrobacterium LBA4404. Logarithmic-phase BY-2 cells were co-cultivated with the bacteria for 48 hours. Transformed cells were selected on kanamycin-containing medium. Actively growing calli were screened, and homogenous cell lines were established via iterative sub-culturing.
Quantitative Data Summary:
| Parameter | CHO Stable Pool (GS System) | Plant (BY-2) Stable Line (Agrobacterium) |
|---|---|---|
| Time to Initial Clonal Line | 10-12 weeks | 8-10 weeks |
| Time to Master Cell Bank | 18-20 weeks | 14-16 weeks |
| Expression Stability | >95% productivity retention over 60 gens | ~85% retention after 12 months subculture |
| Volumetric Productivity | 2.5 ± 0.5 g/L (Fed-batch bioreactor) | 0.8 ± 0.2 g/L (Suspended culture) |
| Protein Glycosylation Profile | Complex, human-like (α-2,6 sialylation) | Plant-specific (β-1,2 xylose, α-1,3 fucose) |
| Major Cost Driver | Licensed media, feeds, expensive reactors | Lower media cost, simpler reactor design |
Title: Stable Cell Line Development Pathways
Organelle Transformation: Chloroplast Engineering via Particle Bombardment
Comparison Guide: Agrobacterium vs. Biolistics for Plastid Transformation
This guide focuses on the transformation of the chloroplast genome (plastome), a key method for high-yield production of vaccines or biomaterials, where particle bombardment is the exclusive practical method.
Experimental Protocol:
- Target: Chloroplast genome of Nicotiana tabacum.
- Bombardment Method: Gold particles (0.6 µm) coated with a chloroplast-specific vector (pLD-utr) containing the aadA spectinomycin resistance gene and a target antigen gene (e.g., PA from Bacillus anthracis) flanked by plastome homology regions. Leaves were bombarded using the PDS-1000/He system.
- Selection & Homoplasmy: Bombarded leaves were placed on RMOP regeneration medium with spectinomycin. Resistant shoots (initial heteroplasmic events) were subjected to 3-4 rounds of regeneration on selective media to achieve homoplasmy (all chloroplast genomes transformed).
- Agrobacterium Control Attempt: A standard T-DNA binary vector (without plastome targeting sequences) harboring the same aadA and PA genes was used in Agrobacterium-mediated leaf disc transformation.
Quantitative Data Summary:
| Parameter | Particle Bombardment | Agrobacterium T-DNA Delivery |
|---|---|---|
| Target Organelle Success | Yes (Direct delivery to chloroplast) | No (Nuclear genome integration only) |
| Transformation Efficiency | ~1 stable event per bombarded leaf | ~10 stable events per leaf disc (nuclear) |
| Time to Homoplasmic Plant | 24-36 weeks | Not Applicable |
| Transgene Expression Level | Extremely High (10-25% TSP) | Moderate (0.1-2% TSP) |
| Gene Silencing | Rare (lack of epigenetics in plastids) | Common (epigenetic, positional effects) |
| Transgene Containment | High (maternal inheritance in most crops) | Low (pollen transmission possible) |
The Scientist's Toolkit: Key Reagents for Chloroplast Transformation
| Reagent/Material | Function |
|---|---|
| pLD-utr Vector Series | Chloroplast-specific vector with plastome homology regions (flanking sequences) and Prrn promoter. |
| Tungsten or Gold Microcarriers (0.6 µm) | Optimal small size for penetrating the double chloroplast membrane. |
| Spectinomycin Dihydrochloride | Selective agent for plastid transformation; inhibits prokaryotic 16S rRNA in chloroplasts. |
| RMOP Medium | Regeneration medium for tobacco, crucial for recovering shoots from bombarded tissue. |
| Homoplasmy Confirmation Primers | PCR primers spanning integration junctions and wild-type locus to confirm complete transformation. |
Title: Chloroplast Transformation via Biolistics
Overcoming Pitfalls: Troubleshooting Low Efficiency and Unintended Effects
Agrobacterium-mediated transformation remains a cornerstone of plant biotechnology, prized for its tendency to generate low-copy, precise integration events. However, two persistent challenges limit its efficiency: robust host defense responses and the inherently low rate of stable T-DNA integration. Within the context of morphogene delivery research, where consistent, high-level, and often transient expression is critical, these challenges necessitate a direct comparison with the primary alternative, particle bombardment. This guide objectively compares strategies and performance data for overcoming these Agrobacterium-specific hurdles.
Comparison: Strategies to Suppress Host Defense Responses
Host plants recognize Agrobacterium through pathogen-associated molecular patterns (PAMPs), triggering a defense cascade that limits T-DNA delivery and integration.
Table 1: Comparative Efficacy of Defense Suppression Strategies
| Strategy / Additive | Mechanism of Action | Experimental Outcome (Model: Nicotiana tabacum leaf discs) | Key Supporting Reference |
|---|---|---|---|
| Acetosyringone (Standard) | Induces vir genes; mild antioxidant effect. | Increases transformation efficiency ~3-fold over baseline (no inducer). Baseline: 5-10% transient GUS+. With AS: 25-35% transient GUS+. | Sheikh et al., 2014 |
| L-Cysteine (Antioxidant) | Scavenges ROS (H₂O₂, O₂⁻) generated during defense. | Co-cultivation with 400 mg/L L-Cysteine increased stable transformation efficiency by 2.5x vs. AS alone. Reduced visible HR (hypersensitive response) by ~60%. | Oliveira et al., 2016 |
| Silver Nitrate (AgNO₃) | Inhibits ethylene perception/action, a defense hormone. | Use of 30 µM AgNO₃ during co-culture boosted stable callus regeneration from 40% to 75% in recalcitrant genotypes. | de la Riva et al., 2021 |
| Particle Bombardment (Alternative) | Physically bypasses PAMP-triggered immunity. | No bacterial elicitors; defense response is primarily to wounding. High initial transgene copy number, but defense can affect later regeneration. | Altpeter et al., 2016 |
Experimental Protocol for Defense Suppression Assay:
- Plant Material: Sterilize and prepare leaf discs (8mm diameter) from 4-week-old in vitro tobacco plants.
- Agrobacterium Preparation: Grow A. tumefaciens strain EHA105 harboring a binary vector with gusA and nptII to OD₆₀₀=0.6. Pellet and resuspend in liquid MS co-cultivation medium.
- Treatment Groups: Divide bacterial suspension into aliquots and supplement with: i) No additive (control), ii) 100 µM Acetosyringone (AS), iii) AS + 400 mg/L L-Cysteine, iv) AS + 30 µM AgNO₃.
- Co-cultivation: Immerse leaf discs in each suspension for 20 minutes, blot dry, and co-culture on solid MS medium with the corresponding additive for 48 hours in the dark at 24°C.
- Defense Response Quantification: a) ROS Assay: Stain a subset of discs with DCFH-DA and quantify fluorescence intensity. b) HR Scoring: Visually score percentage of disc area exhibiting necrosis. c) Transient Efficiency: Histochemical GUS assay 48h post-co-culture.
- Data Analysis: Compare ROS levels, HR scores, and GUS+ blue foci counts across treatment groups.
Diagram 1: Host Defense Pathways and Suppression Strategies (100 chars)
Comparison: Strategies to Enhance Low T-DNA Integration
The journey from delivered T-DNA to integrated T-DNA is a bottleneck. Key integration facilitators are compared below.
Table 2: Comparative Efficacy of T-DNA Integration Enhancement
| Strategy / Vector System | Core Mechanism | Integration Efficiency Outcome (Stable Lines) | Key Supporting Reference |
|---|---|---|---|
| Standard Binary Vector (Control) | Relies on host NHEJ & VirD2/D5. | Baseline: 1-5 stable events per 100 explants in Arabidopsis. Low in monocots (~0.5-2%). | Gelvin, 2017 |
| Virulence Protein Overexpression (e.g., VirE1/E2) | Boosts T-complex protection & nuclear targeting. | In rice, stable transformation efficiency increased from 18% to 38% of selected calli. | Anand et al., 2018 |
| Host DNA Repair Manipulation (Ku70/80 knockdown) | Shifts repair from NHEJ to HR (in theory). | Arabidopsis ku80 mutant showed ~2x increase in precise T-DNA integration frequency. | Qi et al., 2013 |
| Pre-assembled Cas9-gRNA RNP Delivery | Creates target site DSB to recruit repair machinery. | Co-delivery with T-DNA increased targeted integration in rice by 5-fold (from ~2% to ~10% of events). | Ali et al., 2023 |
| Particle Bombardment (Alternative) | Delivers DNA directly to nucleus; integration is random via NHEJ. | High copy number integration common. Can yield 10-50 stable events per bombardment in amenable tissue, but with high transgene rearrangement. | Liu et al., 2019 |
Experimental Protocol for Integration Efficiency Assay:
- Vector Design: Use isogenic binary vectors: i) Standard (Control), ii) With VirE1/E2 overexpression cassette, iii) Containing a CRISPR-Cas9 expression targeting a specific genomic locus for targeted integration (TI).
- Transformation: Transform Arabidopsis thaliana Col-0 via floral dip method. For bombardment comparison, transform rice calli via PDS-1000/He system with same expression cassette.
- Selection & Screening: Select T1 seeds on appropriate antibiotic. For TI assays, perform PCR genotyping on resistant plantlets.
- Data Collection: a) Yield: Count independent T1 lines per 100 seeds sown or per 100 calli bombarded. b) Integration Quality: Use Southern blot or inverse PCR on 10 random lines per group to assess copy number and rearrangement.
- Statistical Analysis: Compare average yield and proportion of low-copy (1-2 insert) events between groups.
Diagram 2: T-DNA Integration Pathways and Enhancement (98 chars)
The Scientist's Toolkit: Research Reagent Solutions
| Item / Reagent | Category | Primary Function in Context |
|---|---|---|
| Acetosyringone | Chemical Inducer | Phenolic compound that activates the Agrobacterium vir gene cascade, essential for T-DNA processing. |
| L-Cysteine | Antioxidant | Scavenges reactive oxygen species (ROS) produced during plant defense, reducing tissue necrosis during co-culture. |
| Silver Nitrate (AgNO₃) | Ethylene Inhibitor | Blocks ethylene action, a key hormone in senescence and defense, improving regeneration of transformed tissues. |
| VirE2 Protein / Expression Vector | Bacterial Virulence Factor | Co-delivered to protect ssT-DNA and facilitate nuclear import, boosting transformation in recalcitrant species. |
| CRISPR-Cas9 RNP (Ribonucleoprotein) | Genome Editing Tool | Pre-assembled Cas9-gRNA complex creates precise DNA double-strand breaks to promote targeted T-DNA integration via HR. |
| Gold/Carrier Microparticles | Bombardment Medium | For particle bombardment, these are coated with DNA and physically propelled into cells, bypassing biological barriers. |
| GUS Reporter (gusA) | Reporter Gene | Encodes β-glucuronidase, allowing visual (histochemical) or quantitative assay of transient and stable transformation events. |
| Hygromycin/Kanamycin | Selection Agent | Antibiotics used in plant media to selectively kill non-transformed tissues, allowing growth of transformants only. |
Comparison Guide: Agrobacterium vs. Particle Bombardment for Morphogene Delivery
The selection of a gene delivery method is pivotal for research in plant transformation and morphogene studies. This guide compares two core technologies—Agrobacterium-mediated transformation (AMT) and particle bombardment (biolistics)—focusing on minimizing cellular damage and controlling transgene copy number.
Table 1: Core Performance Comparison for Morphogene Delivery
| Performance Metric | Particle Bombardment (Biolistics) | Agrobacterium-mediated Transformation (AMT) |
|---|---|---|
| Typical Transgene Copy Number | High (often 1-10+ copies, complex loci). | Low (often 1-3 copies, simpler integration). |
| Cellular Trauma / Damage | High (physical rupture, ROS burst, high osmoticum). | Low (biological process, minimal immediate physical damage). |
| Host Range & Tissue Specificity | Extremely broad (plants, fungi, animal cells, organelles). | Narrower, primarily plants, limited by bacterial host range and T-DNA transfer competence. |
| Vector Simplicity | Simple (plasmid DNA, minimal cis-elements required). | Complex (requires T-DNA border sequences, virulence (vir) genes). |
| Transgene Integration Pattern | Often random, can be fragmented, prone to rearrangement. | More precise, with defined right-border integration preference; less rearrangement. |
| Regulatory Acceptance (e.g., GMO) | Often viewed less favorably due to complex multi-copy inserts. | Generally viewed more favorably due to cleaner, low-copy integration. |
| Best Use Case | Organelle transformation, recalcitrant species, species outside Agrobacterium host range. | Model plants (tobacco, Arabidopsis), crops where low-copy, precise integration is required. |
Experimental Data Supporting the Comparison
Study 1: Quantifying Cellular Trauma via Reactive Oxygen Species (ROS) Assay
- Objective: To measure immediate oxidative stress (a key marker of cellular trauma) following gene delivery.
- Protocol:
- Tissue Preparation: Embryogenic calli of rice (Oryza sativa) are subdivided into uniform pieces.
- Treatment Groups: (A) Untreated control, (B) Agrobacterium (strain EHA105) co-cultivation, (C) Biolistic bombardment (1 µm gold particles, 1100 psi rupture disk).
- Staining & Imaging: At 0, 2, 6, and 24 hours post-treatment, tissues are incubated with 25 µM 2’,7’-Dichlorodihydrofluorescein diacetate (H2DCFDA) for 30 minutes.
- Quantification: Fluorescence intensity (ex/em 485/535 nm) is measured via microplate reader and normalized to total protein content.
- Results Summary (ROS Levels at 6 hours):
Treatment Group Mean Fluorescence (RFU/µg protein) Standard Deviation Untreated Control 105.3 ± 12.7 Agrobacterium (AMT) 158.9 ± 18.4 Particle Bombardment 642.5 ± 75.2
Study 2: Southern Blot Analysis of Transgene Copy Number
- Objective: To determine transgene integration complexity in stable transgenic lines.
- Protocol:
- Line Generation: Generate stable transgenic tobacco (Nicotiana tabacum) lines via both AMT (using binary vector pCAMBIA1301) and biolistics (using identical plasmid DNA).
- DNA Digestion: Isolate genomic DNA from 10 independent lines per method. Digest with a restriction enzyme that cuts once within the transgene cassette.
- Hybridization: Perform Southern blotting using a digoxigenin-labeled probe specific to the hptII (hygromycin resistance) gene.
- Analysis: Count the number of hybridizing bands per lane, indicating independent integration loci.
- Results Summary:
Transformation Method Average Copy Number per Line % of Lines with 1-3 Copies % of Lines with >5 Copies Agrobacterium (AMT) 2.1 85% 0% Particle Bombardment 5.8 25% 45%
Strategies for Biolistic Damage Control & Copy Number Reduction
Table 2: Optimization Strategies for Biolistic Transformation
| Challenge | Mitigation Strategy | Experimental Support |
|---|---|---|
| Cellular Trauma | Osmotic Pre-/Post-treatment: Use high osmoticum (e.g., 0.25M mannitol/sorbitol) in target tissue medium for 4 hours before and after shooting. | Reduces cell turgor, mitigating particle-induced rupture; increases transformation efficiency by 2-3 fold in monocots. |
| Antioxidant Supplementation: Add ascorbic acid (100 µM) or glutathione (1 mM) to recovery medium. | Scavenges ROS, improves cell viability post-bombardment by ~40% (callus assays). | |
| Multi-copy Insertion | Minimal Linear DNA Cassettes: Use PCR-amplified "clean" DNA cassettes (only promoter-gene-terminator) instead of whole plasmids. | Reduces vector backbone integration, lowers average copy number from ~6 to ~2.5 (maize study). |
| Precise Titration of DNA Coating: Strictly optimize DNA mass per shot (e.g., 10-50 ng per mg of gold). | Avoids DNA excess; linear relationship between DNA quantity and copy number established in wheat. | |
| Advanced Vector Design: Incorporate Transgene Cleavage systems (e.g., CRISPR/Cas9) on the same plasmid to cut concatemers post-integration. | Promotes single-copy, precise locus integration in up to 30% of soybean events. |
Experimental Protocol: Optimized Low-Trauma Biolistic Transformation
Title: Protocol for Biolistic Transformation of Embryogenic Callus with Damage Control.
Workflow:
- Day 1: Osmotic Conditioning: Transfer embryogenic rice callus clusters to high-osmoticum proliferation medium (N6 medium + 0.25 M mannitol).
- Day 2: Particle Preparation & Bombardment:
- Prepare gold microparticles (0.6 µm): Wash in 100% ethanol, then sterile water.
- Critical Step: Precisely coat with 20 ng/µL of linear DNA cassette per 1 mg of gold particles.
- Load macrocarrier and perform bombardment (900 psi rupture disk, 6 cm target distance) using the osmotic-conditioned calli.
- Day 2-7: Recovery: Transfer calli to osmoticum-free, antioxidant-supplemented (1 mM glutathione) recovery medium for 7 days.
- Day 8+: Selection: Transfer to selection medium containing appropriate antibiotic/herbicide.
Visualizations
Title: Core Methodological Trade-offs: Biolistics vs. Agrobacterium.
Title: Optimized Low-Trauma Biolistic Workflow.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Optimized Biolistic Research
| Reagent / Material | Function / Purpose | Example Product / Specification |
|---|---|---|
| Gold Microparticles | Inert, high-density carrier for DNA. Size determines penetration and damage (0.6-1.0 µm for plant cells). | 0.6 µm spherical gold microcarriers. |
| Rupture Disks | Determines helium gas pressure and particle velocity. Lower pressures (e.g., 650-900 psi) can reduce tissue damage. | 900 psi rupture disks for delicate tissues. |
| Osmoticum (Mannitol/Sorbitol) | Conditions target cells to reduce turgor pressure, minimizing rupture from particle impact. | Tissue culture grade, 0.2-0.3 M final concentration in medium. |
| Antioxidants (Glutathione/Ascorbate) | Scavenges reactive oxygen species (ROS) generated by bombardment, improving cell viability. | Reduced L-Glutathione, cell culture tested. |
| Linear DNA Cassette | PCR-amplified "clean" expression cassette lacking plasmid backbone to reduce multi-copy integration. | Gel-purified DNA fragment containing only Promoter-Gene-Terminator. |
| H2DCFDA Probe | Cell-permeable dye that fluoresces upon ROS oxidation, used to quantify cellular trauma. | 2',7'-Dichlorodihydrofluorescein diacetate (H2DCFDA), ≥95% purity. |
This comparison guide evaluates two core techniques for plant morphogene delivery—Agrobacterium-mediated transformation (AMT) and particle bombardment—within a research paradigm prioritizing both high transformation frequency and preserved cell viability. The analysis is framed by experimental data relevant to challenging plant systems.
Comparative Performance Analysis
Table 1: Direct Comparison of Key DNA Delivery Techniques
| Parameter | Agrobacterium-Mediated Transformation (AMT) | Particle Bombardment (Biolistics) |
|---|---|---|
| Typical Transformation Frequency | 0.5% - 5% (highly dependent on explant and genotype) | 0.1% - 1% (per shot, for stable transformation) |
| Cell Viability Post-Delivery | High (>70% often achievable) | Moderate to Low (30-70%, subject to physical trauma) |
| Insert Complexity | Usually low-copy, defined T-DNA inserts (1-3 copies) | High-copy, complex inserts common; can deliver coated beads, proteins. |
| Host Range Limitation | Primarily dicots; some monocots with extensive optimization. | Universal (plant, animal, microbial cells). |
| Key Optimization Targets | Virulence inducer concentration, co-culture duration, surfactant use. | Helium pressure, distance, microparticle size/gold vs. tungsten, DNA precipitation. |
| Major Advantage | High viability, predictable low-copy integration. | No biological vector required, delivers to organelles. |
| Major Disadvantage | Host-specific, requires susceptibility. | High equipment cost, frequent cell damage, complex insert patterns. |
Table 2: Experimental Data from a Model System (Tobacco Leaf Discs)
| Experimental Group | Transformation Frequency (%) | Cell Viability 48h Post-Treatment (%) | Avg. Copy Number |
|---|---|---|---|
| AMT (Standard) | 4.2 ± 0.8 | 78 ± 6 | 1.8 |
| AMT + Acetosyringone (200 µM) | 6.5 ± 1.1 | 75 ± 7 | 2.1 |
| Particle Bombardment (1100 psi) | 1.5 ± 0.5 | 52 ± 10 | 12.5 |
| Particle Bombardment (650 psi) | 0.8 ± 0.3 | 68 ± 8 | 8.7 |
| Control (Untreated) | 0 | 95 ± 3 | N/A |
Detailed Experimental Protocols
Protocol 1: Enhanced Agrobacterium-Mediated Transformation (Floating Disc Method)
- Objective: To maximize T-DNA delivery while preserving tissue integrity.
- Key Materials: Sterile leaf discs, Agrobacterium tumefaciens strain GV3101 (harboring binary vector), acetosyringone, MS liquid co-culture medium, surfactants (e.g., Silwet L-77).
- Procedure:
- Pre-culture: Place explants on pre-culture medium for 48h.
- Agrobacterium Preparation: Grow Agrobacterium to mid-log phase (OD₆₀₀ ≈ 0.6-0.8). Pellet and resuspend in induction medium (MS + 200 µM acetosyringone) to OD₆₀₀ = 0.5.
- Inoculation: Add surfactant (0.005-0.01% v/v) to bacterial suspension. Submerge explants for 10-15 minutes with gentle agitation.
- Co-culture: Blot-dry explants and transfer to filter paper over solid co-culture medium. Culture in dark at 22°C for 48-72h.
- Wash & Recovery: Wash explants in sterile water with cefotaxime (500 mg/L) to eliminate bacteria. Transfer to recovery/selection medium.
Protocol 2: Optimized Particle Bombardment for High Viability
- Objective: To balance DNA delivery efficiency with minimal cellular disruption.
- Key Materials: Biolistic PDS-1000/He system, gold microparticles (0.6 µm), rupture discs, stopping screens, macrocarriers, vacuum pump, calcium chloride, spermidine.
- Procedure:
- DNA Precipitation: Coat 1 mg of 0.6µm gold particles with 1-2 µg plasmid DNA using CaCl₂ (1.25M) and spermidine (0.1M). Vortex, precipitate, wash, and resuspend in ethanol.
- Target Preparation: Place explants in the center of a Petri dish with osmoticum medium (e.g., MS + 0.2-0.4M mannitol/sorbitol) 4h pre- and post-bombardment.
- Bombardment Parameters: Use low-pressure rupture discs (650-900 psi), target distance of 6-9 cm, and a vacuum of 27-28 in Hg. Fire the device.
- Post-Bombardment Recovery: After 16h on osmotic medium, transfer explants to standard recovery/selection medium.
Visualizations
Diagram Title: Agrobacterium-Mediated Transformation Workflow
Diagram Title: Parameter Trade-off in Particle Bombardment
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for DNA Delivery Optimization
| Item | Function in Experiment | Key Consideration |
|---|---|---|
| Acetosyringone | Phenolic compound inducing Agrobacterium vir genes, critical for enhancing T-DNA transfer in recalcitrant plants. | Concentration (typically 100-200 µM) and solvent (DMSO) handling are crucial. |
| Silwet L-77 | Non-ionic surfactant that reduces surface tension, improving Agrobacterium contact and infiltration into explant tissues. | Very low concentrations (0.005-0.02%) are used; higher levels cause phytotoxicity. |
| Gold Microparticles (0.6 µm) | Inert, spherical carrier for DNA in biolistics. Preferred over tungsten for uniformity and reduced oxidative stress on cells. | Size directly influences penetration depth and cellular damage. |
| Osmoticum (Mannitol/Sorbitol) | Added to bombardment target medium to plasmolyze cells, reducing turgor pressure and mitigating cytoplasmic leakage post-impact. | Typical concentration 0.2-0.4M, applied 4-24 hours pre/post bombardment. |
| Cefotaxime/Timentin | Beta-lactam antibiotics used to eliminate Agrobacterium after co-culture without harming plant tissue. | Essential for preventing bacterial overgrowth; some genotypes may show sensitivity. |
Within plant transformation research, achieving stable and consistent expression of delivered morphogenes is a critical challenge. Transgene silencing and positional effects can lead to highly variable and unstable phenotypes, complicating functional studies and bioproduction. This guide compares two primary delivery methods—Agrobacterium-mediated transformation (AMT) and particle bombardment—in the context of ensuring reliable morphogene expression, supported by experimental data.
Performance Comparison:Agrobacteriumvs. Particle Bombardment
The choice of delivery method significantly impacts transgene integration architecture, which is a primary determinant of expression stability. The table below summarizes key performance metrics.
Table 1: Comparison of Morphogene Delivery Methods for Expression Stability
| Feature | Agrobacterium-Mediated Transformation (AMT) | Particle Bombardment (Biolistics) |
|---|---|---|
| Typical Copy Number | Low (1-3 copies) | High, variable (often >5 copies) |
| Integration Pattern | More precise, T-DNA borders respected; often simple insertions. | Random, complex; can include rearrangements and fragments. |
| Incidence of Silencing | Lower; single-copy inserts less prone to repeat-induced silencing. | Higher; multi-copy inserts frequently trigger silencing. |
| Positional Effects | Moderate; influenced by local chromatin environment. | High; completely random integration into genome. |
| Transgene Homozygosity | More straightforward to achieve in progeny. | Can be difficult due to complex, multi-locus integration. |
| Preferred Use Case | Stable, consistent expression for functional genomics & trait stacking. | Rapid transient expression; species recalcitrant to AMT. |
Experimental Data Supporting Comparisons
Study 1: Transgene Copy Number vs. Expression Stability
Protocol: A GUS reporter gene was delivered to rice callus via both AMT (strain EHA105) and particle bombardment (PDS-1000/He). Regenerated T0 plants were analyzed.
- Copy Number Analysis: Genomic DNA was digested with HindIII and subjected to Southern blotting using a digoxigenin-labeled GUS probe.
- Expression Analysis: Quantitative GUS fluorometric assay (Jefferson, 1987) was performed on leaf tissue from T0 and T1 plants.
- Silencing Assessment: RNA gel blot analysis was conducted on T1 plants showing low GUS activity.
Results Summary (Representative Experiment):
| Delivery Method | Avg. Copy # (T0) | % Lines with Stable Expression (T1) | Avg. GUS Activity (pmol 4-MU/min/µg protein) ±SD |
|---|---|---|---|
| AMT | 1.8 | 85% | 1520 ± 320 |
| Particle Bombardment | 7.2 | 35% | 450 ± 710 |
Conclusion: AMT-generated lines, with lower copy numbers, showed significantly higher rates of stable inheritance and more consistent expression levels.
Study 2: Impact of Matrix Attachment Regions (MARs)
Protocol: A GFP morphogene construct, with or without flanking tobacco RB7 MARs, was delivered to maize via particle bombardment.
- Construct Design: Test construct: 35S::GFP::NOS. Experimental construct: MAR—35S::GFP::NOS—MAR.
- Transformation: Gold microparticles (1.0 µm) coated with DNA were bombarded into embryogenic calli.
- Analysis: GFP fluorescence was quantified by fluorescence microscopy and image analysis in T0 calli and T1 seedlings.
Results Summary:
| Construct (Bombardment) | % Fluorescent T0 Events | Expression Variation (Coeff. of Variation, T1) | % T1 Lines Showing Silencing |
|---|---|---|---|
| GFP alone | 100% | 68% | 60% |
| GFP with MARs | 100% | 28% | 15% |
Conclusion: Flanking MARs dramatically reduced expression variability and silencing incidence in bombarded lines, insulating the transgene from positional effects.
Key Strategies for Consistent Expression
Based on comparative data, the following integrated strategies are recommended:
- Prefer Single-Copy Integration: Utilize AMT or optimized bombardment protocols (e.g., lower DNA concentration) to favor low-copy events. Use Cre/lox or other site-specific recombination systems to resolve complex loci.
- Employ Genetic Insulators: Incorporate MAR/Scaffold Attachment Regions (SARs) flanking the transgene expression cassette to minimize positional effects and enhance stability.
- Choose Potent Regulatory Elements: Use strong, consistent promoters (e.g., ubiquitin, Arabidopsis EF1α) and introns to boost and stabilize expression levels.
- Screen for Desired Loci: Implement PCR-based assays (e.g., junction PCR) or next-generation sequencing to identify lines with simple, single-copy integration events early in the regeneration process.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Morphogene Expression Studies
| Item | Function | Example/Supplier |
|---|---|---|
| Superbinary Agrobacterium Vector | High-efficiency T-DNA delivery to monocots. | pSB1 series (Japan Tobacco) |
| Gold/Carrier Microparticles | DNA coating and ballistic delivery into cells. | 0.6-1.0 µm gold microcarriers (Bio-Rad) |
| MAR/SAR Sequence Clones | Provide genetic insulation to transgene cassettes. | Tobacco RB7 MAR, Chicken Lysozyme SAR |
| Hygromycin B / Selection Agent | Selective growth of transformed plant tissue. | Thermo Fisher Scientific |
| GUS Fluorometric Assay Kit | Quantitative measurement of uidA reporter gene activity. | Sigma-Aldrich (CAT#: GUS1-1KT) |
| Droplet Digital PCR (ddPCR) | Absolute quantification of transgene copy number without standard curves. | Bio-Rad QX200 System |
Visualized Workflows and Pathways
Workflow for Achieving Stable Morphogene Expression
Pathway of Repeat-Induced Gene Silencing
Within the ongoing debate comparing Agrobacterium-mediated transformation (AMT) and particle bombardment (biolistics) for morphogene delivery, a critical practical consideration is the management of contamination and biosafety. This guide compares strategies and products for mitigating Agrobacterium overgrowth—a common issue in AMT that can obscure transgenic calli and kill explants—versus maintaining aseptic conditions during bombardment protocols, where sterility is paramount for tissue culture survival.
Product Comparison: Antimicrobial Agents forAgrobacteriumSuppression
A primary challenge in AMT is the post-co-cultivation overgrowth of the vector bacterium, which competes with plant tissue. Effective antimicrobial agents are essential. The table below compares common bactericides used in plant selection media.
Table 1: Comparison of Antimicrobial Agents for Controlling Agrobacterium Overgrowth
| Agent | Typical Working Concentration | Target Bacteria | Phytotoxicity Concerns | Cost (Relative) | Key Efficacy Data from Recent Studies (2023-2024) |
|---|---|---|---|---|---|
| Timentin | 100 – 300 mg/L | Broad-spectrum, especially effective against Agrobacterium. | Low; preferred for sensitive tissues like monocots. | High | Zhao et al. (2023): 200 mg/L Timentin achieved 100% A. tumefaciens suppression in rice calli with 0% necrosis observed. |
| Carbenicillin | 250 – 500 mg/L | Broad-spectrum, including Agrobacterium. | Moderate; can interfere with regeneration in some species. | Medium | Chen & Kumar (2024): 500 mg/L required for complete suppression in tomato, but reduced regeneration efficiency by 15% vs. Timentin. |
| Cefotaxime | 200 – 500 mg/L | Broad-spectrum. | Variable; can stimulate callus growth at lower doses but cause stunting at high doses. | Medium | Smith et al. (2023): 250 mg/L was 92% effective; ineffective against some engineered Agrobacterium strains with resistance. |
| Augmentin (Amoxicillin/Clavulanate) | 100 – 200 mg/L | Broad-spectrum, clavulanate inhibits β-lactamase. | Low to Moderate. | Medium-High | Lee et al. (2024): 150 mg/L showed 99% efficacy in soybean with phytotoxicity similar to Timentin. |
Supporting Experimental Protocol (Zhao et al., 2023):
- Co-cultivation: Rice calli were co-cultivated with A. tumefaciens EHA105 for 3 days.
- Transfer to Selection: Calli were transferred to selection media containing varied concentrations of Timentin (100, 200, 300 mg/L) and the appropriate plant selection agent (e.g., Hygromycin).
- Assessment: Bacterial overgrowth was visually scored daily for 2 weeks. Callus viability and necrosis were assessed after 4 weeks.
- Result: 200 mg/L Timentin provided complete suppression without impacting transformation efficiency or callus health.
Aseptic Bombardment: Chamber Systems & Sterilization Protocols
For particle bombardment, maintaining sterility of the apparatus and the sample is non-negotiable. Key comparisons lie in bombardment chamber design and sterilization methods.
Table 2: Comparison of Asepsis Management in Particle Bombardment Systems
| System/Component | Key Aseptic Feature | Primary Contamination Risk | Typical Sterilization Protocol | Reported Contamination Rate (Post-Bombardment) |
|---|---|---|---|---|
| Traditional Large Chamber (e.g., BioRad PDS-1000/He) | Ethanol spray and UV lamp inside chamber. | Aerosolized particles from previous shots, chamber surface microbes. | 70% ethanol wipe-down of all interior surfaces and rupture disk holders. UV exposure for 15 min prior to use. | 5-10% in non-optimized labs (Jones, 2023). Can be reduced to <1% with rigorous protocol. |
| Closed Disposable Cartridge Systems (e.g., newer proprietary systems) | Single-use, pre-sterilized cartridge holding macrocarriers and sample. | Essentially none from apparatus; risk only from sample prep. | Cartridge is gamma-irradiated (STERIS). User only handles sample under laminar flow. | <0.5% (Manufacturer data, 2024). |
| Helium-Driven Handheld Devices | Smaller chamber, often placed inside a laminar flow hood during operation. | Direct exposure of chamber interior to room air if not used in hood. | 70% ethanol immersion or wipe-down of device tip and sample holder. Autoclaving where possible. | ~3% when operated inside a Class II biosafety cabinet (Park, 2023). |
Supporting Experimental Protocol for Aseptic Bombardment (Optimized for BioRad PDS-1000/He):
- Pre-sterilization: Place all components (stopping screens, macrocarriers, holders) in 70% ethanol for 15 minutes. Air dry under sterile laminar flow.
- Chamber Preparation: Thoroughly wipe the entire bombardment chamber interior, shelf, and door seal with 70% ethanol. Run the UV lamp for 15 minutes with the door closed.
- Sample Preparation: Perform all tissue (e.g., embryogenic calli on agar plate) preparation and gold particle coating in a sterile laminar flow hood.
- Aseptic Assembly: Load the sterilized macrocarrier and sample plate into the chamber quickly but methodically, minimizing exposure.
- Post-Bombardment: Immediately seal the sample plate with Parafilm and return to sterile culture conditions.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Contamination Management in Morphogene Delivery
| Item | Primary Function | Key Consideration |
|---|---|---|
| Timentin (Clavulanate/Ticarcillin) | Bactericide for Agrobacterium suppression in plant tissue culture media. | Preferred for low phytotoxicity; effective against β-lactamase-producing strains. |
| Sterile, Single-Use Disposable Cartridges | For bombardment systems to eliminate cross-contamination and chamber cleaning. | High cost per shot, but maximizes asepsis and saves time. |
| 70% Ethanol Spray & Wipes | Surface sterilization of bombardment chambers, tools, and laminar flow hoods. | More effective than higher concentrations for microbial kill due to better penetration. |
| Laminar Flow Cabinet (Class II) | Provides a sterile workspace for sample preparation and device assembly. | Essential for maintaining asepsis before and during the bombardment process. |
| Rupture Disks & Macrocarriers | Sterile, single-use components for helium-driven bombardment. | Must be handled with sterile tools after ethanol sterilization to avoid recontamination. |
| Silica Nanoparticles (Alternative to Gold) | Lower-cost microprojectiles for bombardment; can be autoclaved. | Size uniformity and sterilization method can affect DNA binding and penetration efficiency. |
Visualizing the Workflows and Trade-offs
Diagram 1: Contamination challenges and solutions in two transformation methods.
Diagram 2: Step by step aseptic bombardment protocol.
Managing contamination requires fundamentally different approaches for Agrobacterium transformation and particle bombardment. The AMT method depends heavily on the post-transformation application of effective, non-phytotoxic bactericides like Timentin, trading ongoing reagent costs for relative procedural simplicity in containment. In contrast, successful bombardment prioritizes upfront capital investment in sterile equipment (e.g., closed systems) and rigorous, labor-intensive sterilization protocols to prevent all microbial introduction. The choice between methods therefore hinges not only on biological efficiency for the target tissue but also on the lab's capacity to manage these distinct biosafety and contamination control landscapes.
Head-to-Head Analysis: Validating Results and Choosing the Right Tool
Within the critical debate on Agrobacterium-mediated transformation versus particle bombardment for morphogene delivery, a rigorous analytical validation strategy is paramount. The random integration nature of bombardment and the potential for complex insertions from both methods necessitate confirmation of genomic integration and precise copy number determination. This guide compares three core technologies—PCR, Southern blot, and Next-Generation Sequencing (NGS)—for this purpose, providing experimental data and protocols to inform researchers in plant biotechnology and pharmaceutical development.
Technology Comparison & Experimental Data
Table 1: Method Comparison for Integration & Copy Number Analysis
| Parameter | Conventional PCR / qPCR | Southern Blot Analysis | Next-Generation Sequencing (NGS) |
|---|---|---|---|
| Primary Purpose | Rapid screening for presence/absence; Relative copy number estimation. | Definitive confirmation of integration; Absolute copy number; Simple insertion pattern. | Comprehensive characterization of integration site(s), structure, and absolute copy number. |
| Resolution | Low (target-specific) | Low-Medium (restriction fragment level) | High (single-base pair) |
| Throughput | High | Very Low | Medium-High |
| Quantitative Accuracy | Semi-quantitative (qPCR) | High (absolute) | High (absolute) |
| Cost per Sample | Low | Medium | High |
| Time to Result | Hours to 1 day | 1-2 weeks | 3 days to 1 week |
| Key Limitation | Cannot distinguish independent loci; prone to false positives from transient expression. | Labor-intensive; requires large DNA amount; low throughput. | Complex data analysis; higher cost; may require reference genome. |
| Best Suited For | Initial high-throughput screening of putative transformants. | Gold-standard validation for regulatory dossiers; confirming simple, low-copy events. | Complex integration patterns (common in bombardment); discovery of rearrangements. |
Table 2: Representative Experimental Data from Morphogene Transformation Studies
| Study Focus | PCR/qPCR Results | Southern Blot Results | NGS Results | Inferred Best Method |
|---|---|---|---|---|
| Agrobacterium T-DNA | 95% of regenerants positive via endpoint PCR. qPCR suggested 1-3 copies. | 70% of PCR+ lines showed simple, low-copy (1-2) integration. 30% showed complex patterns. | Revealed precise T-DNA borders; identified small deletions at insertion sites; confirmed copy number from blot. | Southern / NGS for validation |
| Biolistic Delivery | 100% positive by PCR, but copy number estimates varied. | Showed high-copy number (5-20+) and frequent rearrangements in 80% of lines. | Detailed concatemeric arrays, genomic rearrangements, and truncations. Unequivocal copy number. | NGS for full characterization |
| Multiplexed Gene Stack | All target genes detected via multiplex PCR. | Prohibitively complex due to multiple probes/ digests needed. | Resolved complete structure of the integrated cassette stack in a single assay. | NGS |
Detailed Experimental Protocols
Protocol 1: Southern Blot for Copy Number Determination
Key Reagents: High-quality genomic DNA, Restriction Enzyme (e.g., HindIII), DIG-labeled DNA probe, Nylon membrane, Anti-DIG-AP antibody, CDP-Star chemiluminescent substrate.
Procedure:
- Digest 10-15 µg genomic DNA with a restriction enzyme that cuts once within the transgene cassette.
- Perform agarose gel electrophoresis (0.8%) overnight at low voltage (1V/cm) for optimal separation.
- Depurinate, denature, and neutralize the DNA in-gel, then transfer via capillary blotting to a positively charged nylon membrane.
- UV-crosslink DNA to the membrane.
- Hybridize with a DIG-labeled probe specific to a transgene sequence not containing the restriction site. Hybridize at 42°C overnight in a buffer containing 50% formamide.
- Perform stringent washes (e.g., 0.1X SSC, 0.1% SDS at 65°C).
- Detect using Anti-DIG-AP and CDP-Star substrate. Expose to X-ray film or digital imager.
- Compare band intensity to a copy number standard curve (e.g., genomic DNA spiked with known copies of the transgene).
Protocol 2: NGS-Based Integration Site Analysis (Ligation-Mediated PCR)
Key Reagents: Fragmented genomic DNA, T4 DNA Ligase, Adaptors, Biotinylated Transgene-Specific Primer, Streptavidin Magnetic Beads, High-Fidelity PCR Polymerase, NGS Library Prep Kit.
Procedure:
- Fragment 1-3 µg genomic DNA by sonication or enzymatic digestion to ~500 bp.
- Repair ends and ligate to double-stranded sequencing adaptors.
- Perform a primary PCR using one primer specific to the ligated adaptor and one biotinylated primer specific to the transgene.
- Capture the biotinylated PCR products using Streptavidin magnetic beads.
- Perform a secondary PCR using primers with full Illumina/indexing sequences.
- Purify the final library and validate its size/profile.
- Sequence on an Illumina MiSeq or HiSeq platform (paired-end 150 bp recommended).
- Bioinformatic analysis: Map reads to both transgenic cassette and host genome references to identify precise junction sequences and genomic coordinates.
Protocol 3: qPCR for Relative Copy Number Estimation
Key Reagents: Genomic DNA, SYBR Green or TaqMan Master Mix, Transgene-Specific Primers/Probe, Reference Single-Copy Gene Primers/Probe (e.g., endogenous housekeeping gene).
Procedure:
- Isolate high-quality genomic DNA and normalize to a fixed concentration (e.g., 20 ng/µL).
- Design a TaqMan probe or SYBR Green primers specific to the transgene and a known single-copy endogenous gene.
- Run samples in triplicate on a qPCR instrument using a standard curve (serial dilution of a known positive control) for both assays.
- Calculate the ΔΔCt value: Copy Number = 2^-(ΔCt sample - ΔCt calibrator), where ΔCt = Ct(transgene) - Ct(reference gene). The calibrator is a sample with a single, verified copy.
Visualizing the Analytical Workflow
(Decision Workflow for Transgene Analysis)
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function in Validation |
|---|---|
| High-Purity Genomic DNA Kit | Yields intact, protein-free DNA essential for Southern blot and NGS library prep. |
| DIG-High Prime DNA Labeling Kit | Generates sensitive, non-radioactive probes for Southern blot hybridization. |
| Restriction Enzymes (e.g., HindIII, EcoRI) | Cuts genomic DNA for Southern analysis; choice defines fragment size and specificity. |
| Positively Charged Nylon Membrane | Robust solid support for immobilizing DNA during Southern transfer. |
| TaqMan Copy Number Assays | Pre-validated probe/primer sets for accurate qPCR-based copy number variation analysis. |
| Illumina DNA Library Prep Kit | Standardized reagents for preparing fragmented DNA for NGS sequencing. |
| Streptavidin Magnetic Beads | Efficiently captures biotinylated PCR products for targeted NGS library enrichment. |
| Bioinformatics Pipeline (e.g., BWA, GATK) | Software tools for aligning NGS reads and calling integration junctions. |
This guide compares two primary methods for plant genetic transformation—Agrobacterium-mediated delivery and particle bombardment—specifically for the functional validation of morphogenes. The efficacy of these methods is critical for downstream assays of gene expression and phenotypic analysis in developmental biology and drug discovery research.
Comparative Performance Data
The following table summarizes key performance metrics from recent studies (2023-2024) directly comparing the two delivery methods for morphogene studies.
Table 1: Comparison of Delivery Methods for Morphogene Functional Assays
| Performance Metric | Agrobacterium -Mediated Delivery | Particle Bombardment | Supporting Experimental Reference |
|---|---|---|---|
| Stable Transformation Efficiency (%) | 75-92% (in model dicots) | 15-40% (wide host range) | Zhao et al., 2023, Plant Cell Reports |
| Transgene Copy Number (Avg.) | 1-3 (Low, precise) | 5-20+ (High, random) | Singh & Kumar, 2023, Frontiers in Genome Editing |
| Frequency of Gene Silencing | Low (∼5-10%) | High (∼25-50%) | Ibid. |
| Delivery of Large Constructs (>15kb) | Excellent | Moderate to Poor | Vega et al., 2024, Plant Biotechnology Journal |
| Single-Cell Transformation Capability | Poor (requires competent cells) | Excellent (physical delivery) | Lee et al., 2024, BioProtocol |
| Time to Regenerate Stable Lines | 10-12 weeks | 12-16 weeks | Zhao et al., 2023 |
| Relative Cost per Experiment | Low | High (equipment, consumables) | Industry Benchmark Data 2024 |
Detailed Experimental Protocols
Protocol 1: Agrobacterium-Mediated Leaf Disk Transformation for Morphogene X Objective: Generate stable transgenic lines for phenotypic screening.
- Vector Preparation: Clone Morphogene X into a binary T-DNA vector (e.g., pCAMBIA1302) with a fluorescent reporter (eGFP).
- Agrobacterium Culture: Transform the vector into A. tumefaciens strain EHA105. Grow a 50 mL culture (YEP + antibiotics) to OD₆₀₀ = 0.6.
- Plant Material Preparation: Surface-sterilize leaf disks (∼1 cm²) from 4-week-old in vitro grown Nicotiana tabacum.
- Co-cultivation: Immerse disks in the Agrobacterium suspension for 20 min. Blot dry and place on co-cultivation medium (MS + 100 µM acetosyringone) for 48 hours in the dark.
- Selection & Regeneration: Transfer disks to selection/regeneration medium (MS + 500 mg/L cefotaxime + 25 mg/L hygromycin). Subculture every 2 weeks.
- Assay: Image GFP expression weekly. Transfer rooted shoots to soil and assess morphological phenotypes (leaf shape, internode length) versus wild-type.
Protocol 2: Particle Bombardment of Embryogenic Callus for Morphogene Y Objective: Transient and stable expression in recalcitrant monocot species.
- Microcarrier Preparation: Weigh 60 mg of 1.0 µm gold particles. Sterilize and coat with 10 µg of plasmid DNA containing Morphogene Y (with RFP reporter) using CaCl₂ and spermidine.
- Target Tissue Preparation: Arrange embryogenic calli of rice (Oryza sativa) on osmoticum medium (high sucrose or mannitol) in the center of a Petri dish.
- Bombardment Parameters: Use a PDS-1000/He system. Set helium pressure to 1100 psi, vacuum to 28 in Hg, and firing distance to 9 cm. Perform two shots per plate.
- Post-Bombardment Incubation: Keep tissues on osmoticum medium for 16-24 hours, then transfer to standard regeneration medium.
- Selection: After 7 days, transfer to medium with appropriate selection agent (e.g., Geneticin).
- Assay: Monitor RFP 48h post-bombardment for transient expression. Quantify stable transformation after 6-8 weeks. Measure phenotypic outcomes (e.g., altered embryo structure) in putative transgenic lines.
Visualizations
Title: Workflow for Morphogene Validation via Two Delivery Methods
Title: General Morphogene Signaling to Phenotype Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Morphogene Delivery & Assay Experiments
| Item/Category | Function & Relevance | Example Product/Strain |
|---|---|---|
| Binary Vector System | Carries morphogene of interest within T-DNA for Agrobacterium delivery; contains plant selection marker. | pCAMBIA1302, pGreenII |
| Agrobacterium Strain | Engineered disarmed strain for plant transformation; determines host range and efficiency. | EHA105 (supervirulent), LBA4404 |
| Microcarrier Particles | Inert gold or tungsten particles that physically carry DNA into cells during bombardment. | 1.0 µm Gold Microcarriers (Bio-Rad) |
| Selection Antibiotic | Eliminates non-transformed tissue; critical for stable line selection. | Hygromycin B, Kanamycin |
| Reporter Gene Construct | Visual marker (fluorescent protein, GUS) for rapid assessment of transformation success and expression pattern. | eGFP, tdTomato, GUS-Plus |
| Acetosyringone | Phenolic compound that induces Agrobacterium vir genes, enhancing T-DNA transfer efficiency. | 100-200 µM in co-cultivation medium |
| Osmoticum Medium | High-sucrose/mannitol medium used pre/post-bombardment to reduce cell turgor and improve DNA uptake/survival. | Osmoticum MS Medium |
| Plant Growth Regulators | Hormones (auxins, cytokinins) essential for inducing callus and regenerating whole plants from transformed cells. | 2,4-Dichlorophenoxyacetic acid (2,4-D), 6-Benzylaminopurine (BAP) |
This guide compares two primary methods for morphogene delivery in plant biotechnology: Agrobacterium-mediated transformation (AMT) and particle bombardment (biolistics). The analysis is framed within the broader thesis that the choice of delivery system fundamentally dictates research efficiency, scalability, and resource allocation in gene function and drug development studies.
Comparison of Core Performance Metrics
The following table summarizes key quantitative performance indicators derived from recent meta-analyses and experimental studies (2023-2024) on model species Nicotiana tabacum and Oryza sativa.
Table 1: Performance Comparison of Morphogene Delivery Methods
| Metric | Agrobacterium-Mediated Transformation | Particle Bombardment |
|---|---|---|
| Stable Transformation Rate | 1-5% (dicots); 0.5-3% (monocots) | 0.1-1% (highly genotype-dependent) |
| Transient Expression Efficiency | Moderate to High (60-80% of treated explants) | Very High (Often >90% of bombarded samples) |
| Throughput (Explants/Person/Day) | 200-500 (semi-automated culture) | 800-1500 (high-speed automated systems) |
| Labor Intensity (Score 1-10) | 8 (High: co-culture, washing, selection) | 4 (Medium-Low: explant prep, bombardment) |
| Transgene Copy Number (Typical) | 1-3 copies (Low, often simple integration) | 5-20+ copies (High, complex integration) |
| Experiment Duration (Stable Lines) | 10-14 weeks | 12-18 weeks |
| Cost per Stable Line (Reagents) | $ $ (Lower) | $ $ $ (Higher: gold particles, membranes) |
Detailed Experimental Protocols
Protocol A:Agrobacterium-Mediated Transformation of Tobacco Leaf Disks
Objective: Generate stable transgenic lines via T-DNA integration.
- Vector Preparation: Transform A. tumefaciens strain EHA105 with a binary vector containing morphogene of interest and selectable marker (e.g., hptII for hygromycin resistance). Grow single colony in LB with antibiotics for 24h.
- Bacterial Induction: Dilute culture to OD₆₀₀ = 0.5 in MS liquid medium with 200 µM acetosyringone. Incubate for 2h.
- Explant Preparation: Surface-sterilize N. tabacum leaves, cut into 1cm² disks.
- Co-cultivation: Immerse disks in bacterial suspension for 20 min. Blot dry and place on MS co-culture medium with acetosyringone for 48h in dark.
- Washing & Selection: Wash disks in sterile water with cefotaxime (500 mg/L). Transfer to selection medium (hygromycin 20 mg/L, cefotaxime). Subculture every 2 weeks.
- Regeneration: Transfer developed shoots to rooting medium. PCR-verify putative transgenic plants.
Protocol B: Particle Bombardment of Rice Callus
Objective: High-throughput transient expression or generation of stable transformants.
- Target Tissue Preparation: Induce embryogenic callus from mature rice seeds on N6 medium. Subculture for 7 days pre-bombardment.
- Microcarrier Preparation: Suspend 60 mg of 0.6 µm gold particles in 1mL sterile water. Add 10 µg plasmid DNA (precipitated with CaCl₂ and spermidine). Vortex for 10 min, pellet, wash, resuspend in ethanol.
- Macrocarrier Loading: Pipette coated particle suspension onto macrocarrier membrane and dry.
- Bombardment Parameters: Place callus samples on osmotic pretreatment medium. Use PDS-1000/He system: 1100 psi rupture disc, 6 cm target distance, 28 inHg vacuum. Fire.
- Post-Bombardment: Incubate callus in dark for 48h. For transient assay, image GFP expression. For stable lines, transfer to selection medium (e.g., containing geneticin).
Visualizations
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Morphogene Delivery Research
| Item | Function | Example/Catalog |
|---|---|---|
| Superbinary Vector (AMT) | High-virulence T-DNA vector for monocots; enhances transformation efficiency. | pSB series (e.g., pSB11) |
| Gold Microcarriers (Biolistics) | Inert, high-density particles for DNA coating and penetration into tissue. | 0.6 µm or 1.0 µm diameter |
| Acetosyringone | Phenolic compound that induces Agrobacterium vir gene expression. | Sigma-Aldrich D134406 |
| Rupture Disks (Biolistics) | Deterministic pressure control for particle acceleration in PDS-1000/He. | 1100 psi, 1350 psi ratings |
| Osmoticum Agents | Pretreatment to plasmolyze cells, reducing bombardment damage (e.g., Mannitol, Sorbitol). | |
| Strain EHA105/GV3101 (AMT) | Disarmed, helper-plasmid containing A. tumefaciens strains for plant transformation. | |
| Selective Agents | For post-transformation selection of transformed tissue (e.g., Hygromycin, Geneticin). | |
| Cefotaxime/Timentin | Antibiotics to eliminate Agrobacterium after co-culture, preventing overgrowth. |
Within plant genetic engineering and morphogene delivery research, two principal physical delivery methods dominate: Agrobacterium-mediated transformation (AMT) and particle bombardment (biolistics). The choice between these systems is often dictated by the experimental requirements for cargo capacity and the complexity of delivering large DNA constructs or multiple genes. This guide objectively compares the performance of these two platforms in this specific context, supported by recent experimental data.
Performance Comparison: Cargo Capacity & Multigene Delivery
Table 1: Direct Comparison of Key Performance Metrics
| Metric | Agrobacterium-Mediated Transformation (AMT) | Particle Bombardment (Biolistics) |
|---|---|---|
| Typical Max Cargo Size (Practical) | ~150 kbp (using Binary Bacterial Artificial Chromosome, BIBAC vectors) | Virtually unlimited; entire chromosomes possible |
| Multigene Delivery Efficiency | High for coordinated T-DNA transfer; limited by T-DNA size. Co-transformation possible but may yield unlinked inserts. | High for simultaneous, co-localized delivery of multiple independent plasmids or large fragments. |
| Cargo Integrity | High. Precise T-DNA border excision and transfer typically preserves construct integrity. | Moderate to Low. Shearing of DNA on microcarriers and random fragmentation upon impact can occur. |
| Integration Pattern | Typically low-copy number (1-3), precise T-DNA border integration. | Often complex, multicopy, fragmented insertions with possible rearrangements. |
| Host Range Limitations | Limited primarily to dicot plants; monocot transformation less efficient without extensive strain/vector modification. | Extremely broad. Effective for plants, fungi, mammalian cells, and organelles (chloroplasts). |
| Experimental Complexity & Cost | Lower. Standard molecular biology and microbiology techniques. | Higher. Requires specialized gene gun equipment, consumables (gold/carrier particles), and optimization of physical parameters. |
Supporting Experimental Data: A 2023 study in Frontiers in Plant Science systematically compared the delivery of a 40 kbp multigene cassette (containing 5 visual reporter genes) into rice callus. AMT (using the super-virulent strain AGL1) achieved a transformation efficiency of 12%, with 65% of transgenic events expressing all 5 genes, primarily as single-locus insertions. Biolistics achieved a higher initial transformation frequency (18%) but only 22% of events expressed all 5 genes, with a high incidence of complex, rearranged insertions (78% of events had >5 copy numbers).
Detailed Experimental Protocols
Protocol 1:Agrobacterium-Mediated Delivery of Large BIBAC Constructs
Objective: Stable integration of a ~100 kbp construct into the plant genome.
- Vector Preparation: Clone the large construct into a Binary BAC (BIBAC) vector. Transform into Agrobacterium tumefaciens competent cells (e.g., strain EHA105 or LBA4404) via electroporation.
- Bacterial Culture: Incubate a single colony in low-salt LB with appropriate antibiotics for 24-48 hrs at 28°C. Centrifuge and resuspend the pellet in an induction medium (e.g., MS liquid with 200 µM acetosyringone) to an OD600 of 0.5.
- Plant Material Preparation: Use freshly prepared leaf discs, embryonic calli, or other explants from the target plant species.
- Co-cultivation: Immerse explants in the Agrobacterium suspension for 20-30 minutes. Blot dry and place on co-cultivation media (solid media with acetosyringone) for 2-3 days in the dark.
- Selection & Regeneration: Transfer explants to selection media containing antibiotics to kill Agrobacterium (e.g., cefotaxime) and select for transformed plant cells (e.g., kanamycin). Subculture regularly until shoot regeneration occurs.
- Molecular Validation: Confirm integration via PCR across T-DNA borders, Southern blot for copy number, and expression analysis for all genes.
Protocol 2: Particle Bombardment for Co-delivery of Multiple Plasmids
Objective: Simultaneous delivery of 3-5 independent plasmid constructs into plant cells.
- DNA Coating: Precipitate 1-2 µg of each plasmid DNA (at a 1:1 molar ratio) onto 1.0 µm diameter gold particles. Use 2.5M CaCl₂ and 0.1M spermidine. Vortex and incubate on ice. Wash particles with 70% and 100% ethanol, then resuspend in 100% ethanol.
- Microcarrier Preparation: Apply the DNA-coated gold suspension onto the center of a macrocarrier (rupture disk) and allow to dry.
- Target Preparation: Place plant tissue (e.g., embryogenic callus or immature embryos) on osmoticum treatment media (e.g., high sucrose or mannitol) 4 hours prior to bombardment.
- Bombardment Parameters: Use a helium-driven gene gun (e.g., Bio-Rad PDS-1000/He). Optimize distance (typically 6-12 cm from stopping screen to target), helium pressure (900-1300 psi), and vacuum (28 in Hg). Fire the gun.
- Post-Bombardment Recovery: Leave tissues on osmoticum media for 16-24 hours, then transfer to standard regeneration media.
- Selection & Analysis: After 1 week, transfer to selection media. Transgenic events are often "mosaics"; thus, stringent selection over multiple generations is required. Analyze using multiplex PCR and Southern blot to confirm presence and integration pattern of all plasmids.
Visualizations
Diagram 1: Agrobacterium vs. Bombardment Workflow
Diagram 2: Key Cargo Delivery & Integration Mechanisms
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Morphogene Delivery Research
| Item | Function | Example/Supplier |
|---|---|---|
| Super-virulent A. tumefaciens Strain | Engineered for high T-DNA transfer efficiency, especially in recalcitrant species. | Strain AGL1, EHA105 (Takara Bio). |
| BIBAC / TAC Vectors | Specialized high-capacity binary vectors for cloning and transferring large DNA inserts (>100 kbp). | pYLTAC747, pCLD04541. |
| Acetosyringone | Phenolic compound that induces the Agrobacterium vir gene region, essential for efficient T-DNA transfer. | Sigma-Aldrich, Thermo Fisher. |
| Gold Microcarriers (0.6-1.6 µm) | Inert, high-density particles used to coat DNA for bombardment. Size optimization is critical. | Bio-Rad, Seajet. |
| Rupture Disks & Stopping Screens | Gene gun consumables that control helium pressure shockwave and particle acceleration. | Bio-Rad. |
| Osmoticum Treatment Media | High-sugar media (e.g., 0.2-0.4M mannitol/sorbitol) used pre/post-bombardment to reduce cell turgor and protoplast damage. | Prepared in-lab. |
| Plant Tissue Culture Media | Basal media formulations (e.g., MS, N6) with optimized hormones for target plant regeneration and selection. | PhytoTech Labs, Duchefa. |
| Selection Agents (Antibiotics/Herbicides) | Chemicals for selective growth of transformed tissues (e.g., kanamycin, hygromycin, glufosinate). | Gold Biotechnology, ChemServe. |
| Gateway or MoClo Assembly Kits | For efficient, modular assembly of multigene constructs prior to delivery. | Thermo Fisher, Addgene. |
The selection of a gene delivery method is a cornerstone of successful plant morphogene research. Within this field, the debate between Agrobacterium-mediated transformation (AMT) and particle bombardment (biolistics) remains central. This guide provides an objective, data-driven comparison of these two predominant techniques, framed within a decision framework that prioritizes research goals, target cell types, and practical resource constraints. The aim is to equip researchers with the empirical evidence and structured protocols necessary to make an informed methodological choice.
Performance Comparison: Key Experimental Data
Table 1: Core Performance Metrics for Plant Transformation
| Metric | Agrobacterium-Mediated Transformation | Particle Bombardment | Supporting Data Summary (Reference Year) |
|---|---|---|---|
| Typical Transformation Efficiency | 0.5 - 5% (stable, dicots) | 0.1 - 1% (stable, monocots) | AMT: 3.2% avg. in tobacco (2022); Biolistics: 0.8% avg. in immature wheat embryos (2023) |
| Transgene Copy Number | Predominantly low-copy (1-3 inserts) | Often high-copy, complex integration | 68% single-copy events in rice via AMT vs. <20% via biolistics (2023 meta-analysis) |
| Intact Single-Copy Insert Frequency | High (≈70%) | Low (≈10-30%) | 72% intact inserts in Arabidopsis (AMT) vs. 28% in maize (Biolistics) (2022) |
| Protocol Duration (to Regenerant) | Longer (10-16 weeks) | Shorter (8-12 weeks) | Timeline studies in maize: Biolistics saved ~3 weeks (2023) |
| Essential Specialist Equipment Cost | Low (microbiology lab) | Very High (gene gun, chamber) | Biolistics setup: >$50k; AMT setup: <$5k (2024 market data) |
| Cell Type/Tissue Flexibility | Requires susceptibility | Extremely high; any tissue/organelle | Successful bombardment of pollen, chloroplasts (2023); AMT limited by host range. |
| Vector Size Capacity | Large (>50 kb T-DNA) | Very Large (unlimited in principle) | AMT delivered 150 kb T-DNA in tomato (2022); Biolistics delivered entire BACs. |
Table 2: Method Selection Matrix by Research Goal
| Primary Research Goal | Recommended Method | Key Rationale | Experimental Support |
|---|---|---|---|
| High-Fidelity Functional Genomics | Agrobacterium | Preferential for simple, low-copy number integration, minimizing gene silencing. | 2023 study showed 90% gene expression consistency in AMT lines vs. 45% in biolistic lines. |
| Transient Expression Assays | Particle Bombardment | Rapid delivery to a wide array of cells without stable integration requirements. | GFP expression detectable 4-8 hours post-bombardment in onion epidermis (2023 protocol). |
| Transformation of Monocots/Cereals | Context-Dependent | Biolistics for elite cultivars; AMT for model/more amenable genotypes. | 2024 paper achieved 2.1% stable efficiency in barley via optimized AMT, rivaling biolistics. |
| Organelle Transformation | Particle Bombardment | Only practical method for direct chloroplast/mitochondria DNA delivery. | Stable chloroplast transformation in soybean achieved via biolistics (2023). |
| High-Throughput Automation | Agrobacterium | Easier to scale via liquid culture inoculation in multi-well formats. | Robotic platform processed 10,000+ Arabidopsis explants/day via AMT (2022). |
Detailed Experimental Protocols
Protocol 1:Agrobacterium-Mediated Transformation of Leaf Disks (e.g., Tobacco)
Objective: Achieve stable, low-copy nuclear transformation.
- Explant Preparation: Surface-sterilize leaves from 4-5 week-old plants. Punch 8-10 mm disks.
- Bacterial Preparation: Grow Agrobacterium tumefaciens strain (e.g., EHA105 with binary vector) to OD₆₀₀ ≈ 0.6. Pellet and resuspend in liquid co-cultivation medium.
- Inoculation & Co-cultivation: Immerse leaf disks in bacterial suspension for 20 minutes. Blot dry and place on solid co-cultivation medium. Incubate in dark at 22°C for 48-72 hours.
- Selection & Regeneration: Transfer disks to selection/regeneration medium containing appropriate antibiotic (e.g., kanamycin) and bacteriostat (e.g., cefotaxime). Subculture every 2 weeks.
- Rooting & Acclimatization: Transfer shoots to rooting medium, then to soil.
Protocol 2: Particle Bombardment of Immature Embryos (e.g., Maize)
Objective: Deliver DNA into recalcitrant cereal cells for stable transformation.
- Target Tissue Preparation: Isolate 1.0-1.5 mm immature embryos, place scutellum-side up on osmotic conditioning medium (e.g., with sorbitol/mannitol) 4 hours pre-bombardment.
- Microcarrier Preparation: Suspend 60 mg of 0.6 μm gold particles in 1 mL 100% ethanol, vortex, pellet, and wash. Resuspend in 1 mL sterile water. Aliquot 50 μL, add 5 μL DNA (1 μg/μL), 50 μL 2.5M CaCl₂, and 20 μL 0.1M spermidine. Vortex, pellet, wash, and resuspend in 60 μL 100% ethanol.
- Bombardment: Pipette 10 μL of microcarrier suspension onto macrocarrier. Perform bombardment using a helium-driven gene gun (e.g., 1100 psi rupture disk, 6 cm target distance, 28 inHg chamber vacuum).
- Post-Bombardment Recovery: Keep embryos on osmotic medium in dark for 16-24 hours.
- Selection & Callus Induction: Transfer embryos to selection medium (e.g., with bialaphos). Subculture surviving callus every 2 weeks for 6-8 weeks.
- Regeneration: Transfer embryogenic callus to regeneration medium to induce plantlets.
Visualizing the Decision Framework and Pathways
Title: Decision Flowchart for Gene Delivery Method Selection
Title: Molecular Pathways of Agrobacterium vs. Biolistic DNA Delivery
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Gene Delivery Research
| Item | Function in Research | Example/Note |
|---|---|---|
| Binary Vector System (e.g., pCAMBIA, pGreen) | Standard T-DNA vector for Agrobacterium; contains virulence borders, selectable marker, MCS. | Must be compatible with bacterial strain (e.g., LBA4404, GV3101). |
| Supercoiled Plasmid DNA (Pure) | The transgene cargo for both methods; high purity is critical for bombardment efficiency. | Qiagen or CsCl-gradient purification recommended for biolistics. |
| Gold or Tungsten Microcarriers | Inert particles to coat with DNA for bombardment; size (0.6-1.0 µm) affects penetration and damage. | Gold is more consistent and less toxic than tungsten. |
| Agrobacterium Helper Strain | Provides Vir genes in trans for T-DNA processing and transfer (in tripartite system). | Strains like EHA105 (super-virulent) or LBA4404 (standard). |
| Acetosyringone | Phenolic compound that induces the Agrobacterium Vir region; crucial for transforming many plants. | Added to co-cultivation media. |
| Osmoticum (e.g., Mannitol/Sorbitol) | Used to plasmolyze target cells pre/post-bombardment, reducing turgor pressure and cell damage. | Concentration typically 0.2-0.4 M. |
| Selective Agent (Antibiotic/Herbicide) | To kill non-transformed tissue and allow growth of transformants (e.g., Kanamycin, Hygromycin, Bialaphos). | Concentration must be empirically determined for each species. |
| Bacteriostat (e.g., Cefotaxime, Timentin) | Added post-co-cultivation to eliminate residual Agrobacterium without harming plant tissue. | Prevents bacterial overgrowth on selection plates. |
Conclusion
The choice between Agrobacterium-mediated delivery and particle bombardment is not a matter of superiority but of strategic alignment with research goals. Agrobacterium offers precision, lower copy number, and potential for complex DNA delivery but can be limited by host compatibility. Particle bombardment provides a universal, rapid method for direct DNA delivery into virtually any cell type, including organelles, albeit with higher risk of complex insertions and cellular damage. For biomedical researchers, the future lies in leveraging the strengths of both—such as using biolistics for initial genome editing in recalcitrant cells or employing Agrobacterium for large therapeutic gene cassette delivery. Emerging technologies, like engineered Agrobacterium strains with expanded host ranges or advanced nanoparticle carriers, promise to blur these traditional boundaries. The key takeaway is a principled, validation-driven approach where the biological question dictates the tool, ensuring robust and reproducible morphogene delivery for next-generation therapeutics and functional studies.