Evolution and Innovation: How NLR Genes Shape Plant Immunity and Biomedical Potential
This article provides a comprehensive analysis of the evolution of Nucleotide-binding Leucine-rich Repeat (NLR) genes across the plant kingdom, from early algae to modern crops.
Evolution and Innovation: How NLR Genes Shape Plant Immunity and Biomedical Potential
Abstract
This article provides a comprehensive analysis of the evolution of Nucleotide-binding Leucine-rich Repeat (NLR) genes across the plant kingdom, from early algae to modern crops. It explores the foundational principles of NLR diversification, methodological advances in genomic identification, the challenges of immune regulation, and the validation of NLR functions through comparative genomics and functional assays. Aimed at researchers and drug development professionals, the synthesis highlights how understanding the dynamic evolutionary patterns of this key gene family informs strategies for disease-resistant crop breeding and offers insights into the evolution of innate immunity mechanisms with broader biomedical relevance.
From Algae to Angiosperms: Tracing the Ancient Origins and Expansion of the NLR Gene Family
The innate immune system in plants relies heavily on nucleotide-binding leucine-rich repeat (NLR) proteins, which serve as intracellular sensors for pathogenic invaders. These sophisticated receptors recognize pathogen effector molecules and initiate robust defense responses [1]. While NLR proteins are central to plant immunity, their evolutionary origins extend deep into the history of life, predating the emergence of plants and animals. Comparative genomic analyses across diverse organisms have revealed that the core components of NLR proteins existed in prokaryotic life forms before the divergence of eukaryotes [2] [3]. This review examines the foundational evidence for the prokaryotic origins of NLR building blocks and the independent assembly events that produced modern plant immune receptors, providing a crucial evolutionary context for understanding NLR function in land plants.
Molecular Architecture of Plant NLRs
Plant NLR proteins exhibit a characteristic modular structure consisting of three core domains that define their function in immunity signaling. The central nucleotide-binding domain (NB-ARC in plants) serves as a molecular switch regulated by nucleotide binding and hydrolysis [2] [3]. The C-terminal leucine-rich repeat (LRR) domain is primarily involved in effector recognition, while the N-terminal domain, which can be a Toll/Interleukin-1 receptor (TIR) domain, coiled-coil (CC) domain, or resistance to powdery mildew 8 (RPW8) domain, mediates downstream signaling [4]. This modular architecture enables NLRs to act as sophisticated molecular switches that detect pathogen effectors and initiate immune responses.
Table 1: Core Domains of Plant NLR Immune Receptors
| Domain | Full Name | Primary Function | Structural Features |
|---|---|---|---|
| N-terminal | TIR (Toll/Interleukin-1 Receptor) | Signaling initiation | α-helices and β-strands forming a globular structure |
| CC (Coiled-Coil) | Signaling initiation | Helical bundles with heptad repeats | |
| RPW8 (Resistance to Powdery Mildew 8) | Signaling initiation | Compact helical domain unique to plants | |
| NB-ARC | Nucleotide-Binding adaptor shared with APAF-1, R proteins, and CED-4 | Molecular switch for activation | Nucleotide-binding pocket with conserved motifs (P-loop, RNBS, etc.) |
| LRR | Leucine-Rich Repeat | Effector recognition & protein interactions | Repetitive β-strand/α-helix motifs forming curved structure |
The functional integration of these domains allows NLR proteins to adopt autoinhibited conformations in the absence of pathogens and undergo dramatic conformational changes upon effector detection, ultimately leading to the activation of defense responses including programmed cell death [1] [2].
Evidence for Prokaryotic Origins
Comprehensive genome-wide comparative analyses have provided compelling evidence for the ancient origins of NLR building blocks. A landmark study by Yue et al. (as cited in [2] [3]) analyzed 38 model organisms spanning major taxonomic groups, including eubacteria, archaebacteria, protists, fungi, plants, and metazoans. This extensive analysis revealed that the core structural domains of NLR proteins—including the NB-ARC, NACHT, TIR, and LRR domains—already existed in prokaryotic genomes before the evolutionary divergence of eukaryotes.
The research methodology involved several sophisticated bioinformatic approaches:
- Large-scale data mining of genomic and transcriptomic data from 5,126 species across nine early plant lineages
- Domain architecture analysis using tools such as InterProScan and Pfam database searches
- Phylogenetic reconstruction to trace the evolutionary relationships between domains across taxa
- Motif combination analysis to determine how different domains became associated
This investigation revealed that while the individual building blocks existed in prokaryotes, they were not assembled into the multi-domain architecture characteristic of modern plant NLRs. The NB-ARC domain found in plant NLRs and the structurally similar NACHT domain present in animal NLRs showed clear phylogenetic distinctions, suggesting either an ancient divergence or completely independent origins before the separation of eukaryotes, eubacteria, and archaebacteria [2] [3].
Table 2: Evolutionary Distribution of NLR Building Blocks Across Life
| Taxonomic Group | NB-ARC Domain | NACHT Domain | LRR Domain | TIR Domain | Assembled NLR |
|---|---|---|---|---|---|
| Eubacteria | Present | Present | Present | Present | Absent |
| Archaebacteria | Present | Present | Present | Present | Absent |
| Early Eukaryotes | Present | Present | Present | Present | Absent |
| Early Land Plants | Present | Absent | Present | Present | Present |
| Flowering Plants | Present | Absent | Present | Present | Present |
The presence of these individual domains in prokaryotic organisms suggests they served fundamental cellular functions related to stress response, nucleotide sensing, and protein-protein interactions before being co-opted for immune functions in multicellular organisms.
Independent Fusion Events
The assembly of individual prokaryotic domains into complete NLR proteins represents a fascinating case of convergent evolution between plants and animals. Evidence indicates that the fusion events that created functional NLRs occurred independently in the evolutionary lineages leading to plants and animals after their divergence [2] [3].
In plants, the fusion between an ancestral NB-ARC domain and an LRR domain created the foundational structure that would later diversify into the various NLR subtypes. Similarly, in animals, a fusion event between an ancestral NACHT domain and an LRR domain produced the basic animal NLR architecture. These independent fusion events can be dated to a period coinciding with the emergence of multicellularity, suggesting that the evolution of complex multicellular organisms created selective pressures for sophisticated immune recognition systems [2].
The phylogenetic and motif combination analyses conducted in these studies provide strong support for independent origins rather than shared ancestry of the fully formed NLR proteins. This conclusion is further strengthened by the observation that the signaling domains at the N-terminus of plant NLRs (TIR, CC, RPW8) are distinct from those found in animal NLRs, indicating different evolutionary trajectories in how these immune receptors acquired their signaling capabilities [2] [3].
Figure 1: Independent Evolution of NLR Proteins in Plants and Animals. The diagram illustrates how plant and animal NLRs evolved through separate fusion events from prokaryotic domain precursors.
Evolution in Land Plants
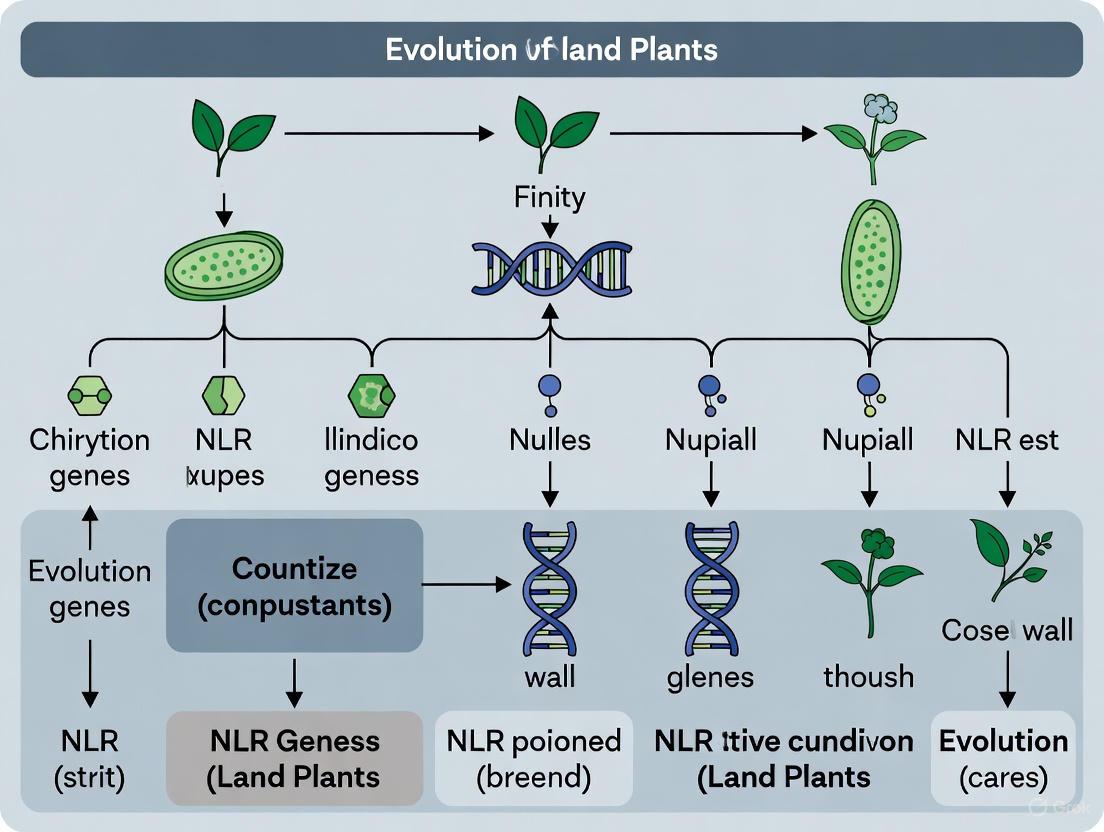
Following the initial fusion events, NLR genes underwent dramatic expansion in flowering plants, resulting in the highly diverse and species-specific NLR repertoires observed today. Genomic analyses have revealed that early land plants such as the bryophyte Physcomitrella patens and the lycophyte Selaginella moellendorffii possess relatively small NLR repertoires of approximately 25 and 2 genes respectively, indicating that the major expansion of NLR genes occurred primarily in flowering plants [2] [3].
The evolutionary dynamics of NLR genes in flowering plants exhibit remarkable variation without clear correlation to phylogenetic relationships, suggesting species-specific mechanisms of expansion and contraction. For example, within the Brassicaceae family, Arabidopsis thaliana, Arabidopsis lyrata, and Brassica rapa possess 151, 138, and 80 full-length NLRs respectively, demonstrating significant variation even among closely related species [2].
Recent research on Apiaceae species reveals further evidence of dynamic NLR evolution, with gene numbers ranging from 95 in Angelica sinensis to 183 in Coriandrum sativum [5]. Phylogenetic analysis of these species indicates they descended from approximately 183 ancestral NLR lineages, with different species experiencing distinct patterns of gene loss and gain events during evolution [5].
The expansion of NLR genes in flowering plants has been primarily driven by tandem duplication events, which facilitate rapid generation of new resistance specificities. For example, in pepper (Capsicum annuum), tandem duplication accounts for 18.4% of NLR genes (53 of 288), with particularly high density on chromosomes 08 and 09 [6]. This clustering of NLR genes, especially near telomeric regions, enables efficient recombination and sequence diversification, allowing plants to keep pace with rapidly evolving pathogens [6].
Research Methods and Experimental Approaches
The identification and characterization of NLR genes and their evolutionary history relies on sophisticated bioinformatic and experimental methodologies. Below are the key protocols used in this field of research.
Genome-Wide Identification of NLR Genes
The standard workflow for comprehensive NLR identification involves both homology-based and domain-based approaches [6] [4] [5]:
- Sequence Retrieval: Obtain reference NLR protein sequences from databases such as TAIR for model organisms.
- Homology Search: Perform BLASTp searches against the target proteome using known NLR sequences.
- Domain Analysis: Conduct HMMER searches using the NB-ARC domain profile (PF00931) from the Pfam database with an E-value cutoff of 1×10⁻⁵.
- Domain Validation: Verify candidate sequences using NCBI's Conserved Domain Database (cd00204 for NB-ARC) and Pfam batch search.
- Architecture Classification: Classify NLRs into subclasses (TNL, CNL, RNL) based on N-terminal domains and identify partial-length NLRs.
- Manual Curation: Remove redundant sequences and verify domain organization.
Evolutionary and Phylogenetic Analysis
Reconstructing the evolutionary history of NLR genes involves several computational steps [6] [4] [5]:
- Domain Extraction: Parse NB-ARC domain sequences from full-length proteins.
- Multiple Sequence Alignment: Use tools such as Clustal Omega or Muscle with default parameters.
- Phylogenetic Reconstruction: Construct maximum likelihood trees using IQ-TREE with 1000 bootstrap replicates.
- Tree Visualization: Annotate and display phylogenetic trees using ggtree in R.
- Gene Duplication Analysis: Identify duplication events using MCScanX and determine duplication types (tandem, segmental, transposed).
- Synteny Analysis: Examine genomic contexts using Dual Synteny Plotter in TBtools.
Expression and Functional Analysis
Linking NLR genes to biological function involves integrated molecular approaches [6]:
- Transcriptome Profiling: Map RNA-seq reads to reference genomes using HISAT2.
- Differential Expression: Identify significantly differentially expressed NLR genes using DESeq2 with thresholds of |log2 Fold Change| ≥ 1 and FDR < 0.05.
- Promoter Analysis: Extract 2kb upstream sequences and identify cis-regulatory elements using PlantCARE.
- Protein Interaction Prediction: Construct PPI networks using STRING database with confidence >0.4.
- Experimental Validation: Conduct RT-qPCR under controlled conditions to verify expression patterns.
Figure 2: Workflow for NLR Identification and Analysis. The diagram outlines the bioinformatics pipeline for comprehensive NLR gene identification and characterization.
Research Reagent Solutions
The following table provides essential reagents and resources for conducting evolutionary and functional studies of NLR genes.
Table 3: Essential Research Reagents for NLR Evolutionary Studies
| Reagent/Resource | Primary Function | Application Examples | Key Features |
|---|---|---|---|
| PlantNLRatlas Database | Comprehensive NLR dataset | Comparative studies across 100 plant species | 68,452 full and partial-length NLRs from diverse taxa [4] |
| RefPlantNLR Database | Experimentally validated NLRs | Reference for functional annotation | 415 experimentally confirmed NLR proteins from 73 plants [4] |
| Pfam NB-ARC Domain (PF00931) | Domain identification | HMMER-based NLR identification | Curated hidden Markov model for NB-ARC domain [6] [5] |
| InterProScan | Protein domain annotation | Comprehensive domain architecture analysis | Integrates multiple databases including Pfam, SUPERFAMILY [4] |
| MCScanX | Gene duplication analysis | Identifying tandem and segmental duplications | Synteny-based evolutionary analysis [6] [5] |
| STRING Database | Protein-protein interactions | Predicting NLR immune networks | Interaction predictions with confidence scores [6] |
The evolutionary history of NLR proteins reveals a remarkable journey from individual domains in prokaryotic organisms to sophisticated immune receptors in plants. The building blocks of NLR proteins—NB-ARC, LRR, and TIR domains—were present in prokaryotes and assembled into functional immune receptors through independent fusion events in plants and animals. In plants, this assembly created a versatile immune platform that subsequently expanded dramatically, particularly in flowering plants, through mechanisms such as tandem duplication. This expansion has resulted in diverse, species-specific NLR repertoires that enable plants to detect rapidly evolving pathogens. Understanding these evolutionary processes provides crucial insights for harnessing NLR diversity to improve crop resistance through both traditional breeding and biotechnological approaches.
Within the intricate immune systems of plants, nucleotide-binding leucine-rich repeat (NLR) proteins function as critical intracellular sentinels, orchestrating defense responses against diverse pathogens. For decades, the evolutionary origin of these sophisticated immune receptors has been a subject of intense scientific inquiry. Prevailing theories suggested that NLRs emerged concurrently with plants' colonization of land, coinciding with the need to cope with more complex terrestrial pathogen environments. However, recent genomic investigations have fundamentally challenged this paradigm, tracing the ancestry of NLR genes back to early green plants. This whitepaper synthesizes cutting-edge research that identifies functional NLR genes in algal lineages, revealing the ancient origin of plant intracellular immunity and providing unprecedented insights into the evolutionary trajectory of immune receptor families. These findings not only reshape our understanding of plant immunity evolution but also open new avenues for engineering disease resistance in crops by harnessing ancient immune mechanisms.
Evolutionary Genomic Evidence for Ancient NLR Origins
Phylogenomic Distribution Across Green Plant Lineages
Comprehensive genome-wide analyses across diverse algal species have revealed the presence of NLR genes in early-diverging green plant lineages. A systematic investigation of 44 chlorophyte species across seven classes and seven charophyte species across five classes identified a variable number of NLR genes, ranging from one to twenty, in five chlorophytes and three charophytes [7]. Notably, several algal genomes contained no detectable NLR genes, suggesting either gene loss or the presence of alternative immune recognition systems in these lineages [7].
Table 1: Distribution of NLR Genes in Green Plant Lineages
| Plant Group | Species Surveyed | Species with NLRs | NLR Count Range | TNLs Identified | nTNLs Identified |
|---|---|---|---|---|---|
| Chlorophytes | 44 species | 5 species | 1-20 | Yes | Yes |
| Charophytes | 7 species | 3 species | 1-20 | Yes | Yes |
| Land Plants | Multiple | All surveyed | 150-500 | Yes | Yes |
When compared to land plants, which typically possess expanded NLR repertoires ranging from approximately 150 in Arabidopsis thaliana to 500 in Oryza sativa, algal genomes contain significantly fewer NLR genes [7] [6]. This quantitative disparity supports the hypothesis that the substantial expansion of NLR genes in land plants represents an adaptive response to more complex pathogen environments encountered during terrestrial colonization [7].
Structural Conservation and Divergence in Algal NLRs
Detailed analysis of algal NLR protein architecture has revealed remarkable structural conservation with their land plant counterparts while highlighting lineage-specific innovations:
- Domain Organization: Algal NLRs exhibit the characteristic tripartite domain structure consisting of N-terminal signaling domains, central nucleotide-binding sites (NBS), and C-terminal leucine-rich repeats (LRRs) [7].
- N-terminal Diversity: Both TIR (Toll/Interleukin-1 Receptor) and non-TIR (including CC, RPW8) N-terminal domains have been identified in algal NLRs, demonstrating that the major NLR subclasses diversified before plant terrestrialization [7] [8].
- Conserved Motifs: Profiling of conserved motifs within the NBS domain revealed shared sequence features between algal and land plant NLRs, supporting their common evolutionary origin [7].
- Lineage-specific Variations: Certain algal lineages possess NLR variants with domain combinations not observed in land plants, suggesting both functional conservation and lineage-specific innovation during NLR evolution [7].
Phylogenetic analyses demonstrate that the diversity of land plant NLRs nests within the broader diversity of charophyte NLRs, indicating that NLRs not only originated but diversified into major classes before plant colonization of land [8].
Experimental Validation of Primordial NLR Function
Immune Activation Capacity of Algal NLRs
Functional characterization of algal NLRs through immune-activation assays has provided compelling evidence for their capacity to initiate defense responses. Heterologous expression of both TNL and RNL proteins from green algae in Nicotiana benthamiana elicited hypersensitive responses, demonstrating that the molecular basis for immune activation had already emerged in the early evolutionary stage of different types of NLR proteins [7].
This conservation of function across billion years of evolutionary divergence indicates that the core signaling mechanisms underlying NLR-mediated immunity were established in the common ancestor of green plants and have been maintained under strong selective pressure [7]. The functional capacity of algal NLRs to trigger cell death responses in a distantly related land plant system underscores the deep conservation of immune signaling pathways.
Genomic Context and Evolutionary Dynamics
Examination of the genomic context of NLR genes in early green plants has revealed evolutionary patterns that presage the dynamic nature of NLRomes in land plants:
- Clustered Arrangement: Similar to land plants, algal NLR genes often display clustered genomic arrangements, potentially facilitating the rapid generation of diversity through unequal crossing over and gene conversion [6].
- Birth-and-Death Evolution: The variable presence/absence of NLR genes across algal lineages suggests a birth-and-death evolutionary process operating since the earliest stages of NLR evolution [7].
- Differential Expansion: The significant expansion of NLR genes in land plants compared to algae correlates with the increased complexity of terrestrial pathogen environments, supporting the hypothesis that NLR repertoire size scales with immunological demand [7] [9].
Methodological Framework for Identifying Primordial NLRs
Genomic Identification and Annotation Pipeline
The accurate identification of NLR genes in algal genomes requires specialized bioinformatic approaches tailored to overcome challenges posed by their high sequence diversity and complex domain architecture:
Table 2: Core Methodological Pipeline for NLR Identification
| Step | Method/Tool | Key Parameters | Purpose |
|---|---|---|---|
| Initial Identification | HMMER v3.3.2 | PF00931 (NB-ARC), E-value<1×10-5 | Detect core NLR domain |
| Domain Validation | NCBI CDD, Pfam | cd00204 (NB-ARC) | Confirm domain integrity |
| Architecture Classification | Custom scripts | TIR, CC, RPW8, LRR domains | Classify NLR subtypes |
| Phylogenetic Analysis | IQ-TREE | 1000 bootstrap replicates | Evolutionary relationships |
| Functional Prediction | MEME | Motif enrichment | Identify conserved motifs |
This pipeline begins with homology-based searches using known NLR sequences as queries against algal proteomes, followed by hidden Markov model (HMM) scans using representative NLR domain profiles (PF00931 for NB-ARC domains) [6]. Candidate sequences containing NB-ARC domains are retained and subjected to comprehensive domain architecture analysis using NCBI's Conserved Domain Database (CDD) and Pfam to verify the presence and completeness of N-terminal (TIR, CC, RPW8) and C-terminal (LRR) domains [7] [6].
Phylogenetic Reconstruction and Evolutionary Analysis
Robust phylogenetic analysis is essential for tracing the deep evolutionary history of NLR genes:
- Sequence Alignment: NB-ARC domain sequences or full-length protein sequences are aligned using tools such as Muscle v5 with default parameters [6].
- Tree Construction: Maximum likelihood trees are generated using IQ-TREE with 1000 bootstrap replicates to assess branch support [6].
- Evolutionary Timescale Estimation: Molecular dating approaches, incorporating fossil calibrations where available, help pinpoint the timing of key duplication events and lineage diversification [9].
Experimental Validation of Immune Function
Functional characterization of algal NLRs employs heterologous expression systems to overcome challenges associated with working directly with algal systems:
- Heterologous Expression: Candidate algal NLR genes are cloned into appropriate binary vectors and transiently expressed in model systems such as Nicotiana benthamiana [7].
- Cell Death Assays: Hypersensitive response (HR) cell death is quantified through visual scoring, ion leakage measurements, and vital staining [7].
- Protein Interaction Studies: Co-immunoprecipitation and bimolecular fluorescence complementation assays determine interaction partners and oligomerization capacity [10].
Figure 1: Experimental workflow for identification and functional validation of algal NLRs
Table 3: Key Research Reagents for Algal NLR Studies
| Reagent Category | Specific Examples | Application | Technical Notes |
|---|---|---|---|
| Genomic Resources | Chara braunii, Klebsormidium nitens, Chlamydomonas reinhardtii genomes | Phylogenomic analysis | Phytozome, NCBI Genome portals |
| Bioinformatics Tools | NLRtracker, NLR-Annotator | Automated NLR annotation | Domain-based classification |
| Expression Vectors | pEAQ-HT, pGWB2 | Heterologous expression | Gateway-compatible systems |
| Model Systems | Nicotiana benthamiana | Transient expression assays | 3-5 week old plants optimal |
| Detection Antibodies | Anti-GFP, Anti-MYC | Protein localization | Confocal microscopy |
| Cell Death Markers | Evans blue, electrolyte leakage kits | HR quantification | Multiple timepoints recommended |
Evolutionary Trajectory and Diversification of NLR Genes
From Algal Simplicity to Land Plant Complexity
The evolutionary journey of NLR genes from algae to land plants reveals a pattern of progressive complexity and adaptation:
- Ancient Origin: The presence of functional NLRs in both chlorophytes and charophytes indicates that the common ancestor of green plants possessed a basic NLR toolkit [7].
- Differential Expansion: While algal genomes typically contain zero to twenty NLR genes, land plants exhibit substantial expansions, with numbers ranging from approximately 150 in Arabidopsis to 500 in rice [7] [6].
- Life History Strategy Influence: Recent research in the genus Glycine reveals remarkable distinctions between annual and perennial species, with annuals exhibiting expanded NLRomes compared to perennials, highlighting how ecological strategies shape NLR evolution [9].
- Timing of Expansion: Evolutionary timescale analysis pinpoints recent accelerated gene duplication events in annual Glycine species between 0.1 and 0.5 million years ago, driven predominantly by lineage-specific and terminal duplications [9].
Mechanisms of NLR Diversification
Multiple molecular mechanisms have driven the diversification of NLR genes throughout plant evolution:
- Tandem Duplication: This represents the primary driver of NLR family expansion, accounting for 18.4% of NLR genes in pepper and resulting in significant clustering, particularly near telomeric regions [6].
- Whole Genome Duplication: Polyploidy events provide raw genetic material for neofunctionalization, as evidenced by the unbalanced expansion of the NLRome in the Dt subgenome compared with the At subgenome in the allopolyploid G. dolichocarpa [9].
- Birth of Novel Genes: Perennial Glycine lineages exhibit unique and highly diversified NLR repertoires with limited interspecies synteny, resulting from the birth of novel genes following individual speciation events [9].
- Domain Shuffling and Fusion: Frequent domain loss and fusion events have generated diverse NLR architectures, including TIR-only proteins that can function as independent immune sensors [8].
Figure 2: Evolutionary mechanisms driving NLR gene diversification
Implications for Crop Improvement and Biotechnology
The discovery of functional NLRs in algal species opens new possibilities for engineering disease resistance in crop plants:
- Minimalist Immune Receptors: The relatively simple domain architecture of algal NLRs may provide streamlined templates for engineering synthetic immune receptors with customized specificities [7].
- Conserved Signaling Modules: The demonstration that algal NLRs can activate immune responses in land plants suggests that core signaling pathways are deeply conserved, enabling interfamily transfer of immune components [11].
- Exploitation of Wild Relatives: The identification of highly diversified NLR repertoires in perennial Glycine species highlights the value of wild relatives as reservoirs of novel resistance genes for crop improvement [9].
- Engineering Broader Resistance: Understanding the primordial structural features of algal NLRs may facilitate the design of receptors with extended recognition capabilities, potentially providing resistance to multiple pathogen classes [11].
The identification of functional NLR genes in early green algae represents a paradigm shift in our understanding of plant immunity evolution. These findings demonstrate that the molecular foundations of intracellular immunity originated not with land colonization, but in aquatic ancestors that predated terrestrial plants by hundreds of millions of years. The conserved capacity of algal NLRs to activate immune responses in distantly related land plants underscores the remarkable conservation of core immune signaling mechanisms across a billion years of plant evolution. Future research characterizing the specific pathogen triggers and signaling partners of algal NLRs will provide deeper insights into the primordial immune networks from which the complex plant immune system evolved. These ancient immune receptors offer valuable genetic resources for engineering sustainable disease resistance in crop plants, potentially providing novel solutions to emerging agricultural challenges.
The intracellular immune system of plants is orchestrated by Nucleotide-binding domain and Leucine-rich Repeat (NLR) proteins, which function as sophisticated surveillance mechanisms detecting pathogen effector molecules and activating robust defense responses termed effector-triggered immunity (ETI) [3]. NLRs exhibit a conserved tripartite architecture consisting of a central nucleotide-binding (NB-ARC) domain, C-terminal leucine-rich repeats (LRRs), and variable N-terminal domains that directly execute immune signaling [12]. In flowering plants (angiosperms), the majority of NLR N-terminal domains belong to the coiled-coil (CC), Resistance to Powdery Mildew 8 (RPW8), or Toll/interleukin-1 receptor (TIR) subfamilies [12].
A defining characteristic of the NLR gene family is its extraordinary expansion throughout plant evolutionary history, particularly within flowering plant lineages [13]. This massive diversification has created one of the largest and most variable protein families in plant genomes, enabling recognition of rapidly evolving pathogen effectors [3] [6]. This whitepaper examines the patterns, mechanisms, and functional consequences of NLR repertoire diversification across land plants, from early-diverging bryophytes to modern angiosperms, within the broader context of plant immune system evolution.
Quantitative Expansion of NLR Repertoires Across Plant Lineages
Comparative Genomic Surveys Reveal Differential Expansion
Table 1: NLR Gene Repertoire Size Across Representative Plant Species
| Species | Common Name | Plant Group | Total NLRs | TNLs | CNLs | Other NLRs | Reference |
|---|---|---|---|---|---|---|---|
| Physcomitrella patens | Moss | Bryophyte | ~25 | 8 | 9 | 8 | [3] |
| Selaginella moellendorffii | Spike moss | Lycophyte | ~2 | 0 | NA | NA | [3] |
| Arabidopsis thaliana | Thale cress | Eudicot | 151 | 94 | 55 | 0 | [3] |
| Oryza sativa | Rice | Monocot | 458 | 0 | 274 | 182 | [3] |
| Vitis vinifera | Wine grape | Eudicot | 459 | 97 | 215 | 147 | [3] |
| Capsicum annuum | Pepper | Eudicot | 288 | Not specified | Not specified | Not specified | [6] |
| Arachis hypogaea | Peanut | Eudicot (tetraploid) | 654 | Not specified | Not specified | Not specified | [14] |
| Glycine max | Soybean | Eudicot | 319 | 116 | 20 | NA | [3] |
Genome-wide comparative analyses reveal that early land plant lineages possess relatively modest NLR repertoires. The bryophyte Physcomitrella patens (moss) contains approximately 25 NLR genes, while the lycophyte Selaginella moellendorffii (spike moss) possesses merely 2 NLR genes [3]. This stands in stark contrast to the massive expansions observed in flowering plants, where NLR repertoires typically range from approximately 150 to over 650 genes [3] [14].
This expansion trend is further exemplified in recent studies of crop species. A comprehensive analysis of 34 plant species identified 12,820 NBS-domain-containing genes, classifying them into 168 distinct architectural classes [13]. In the Arachis (peanut) genus, diploid species contained 284-521 NLR genes, while tetraploid cultivated peanut (A. hypogaea) harbored 654 NLR genes [14]. Similarly, pepper (Capsicum annuum) possesses 288 high-confidence canonical NLR genes, with notable clustering on specific chromosomes [6].
Evolutionary Patterns of NLR Subtypes
The distribution of NLR subfamilies across plant phylogeny reveals distinct evolutionary trajectories. While TIR-NLRs (TNLs) and CC-NLRs (CNLs) are widely distributed across land plants [12], some lineages exhibit notable specializations or losses. Monocot species, including rice, brachypodium, sorghum, and maize, have completely lost TNL genes [3], suggesting divergent evolutionary paths in immune receptor utilization between monocot and eudicot lineages.
In non-flowering plants, bioinformatic surveys have identified both common (CC, RPW8, TIR) and atypical N-terminal NLR domains, including αβ-hydrolases and protein kinases, which first appear in bryophytes [12]. These unusual configurations demonstrate the evolutionary innovation in NLR architecture that occurred during early land plant evolution.
Molecular Mechanisms Driving NLR Diversification
Gene Duplication as the Primary Driver of Expansion
Tandem duplication represents the predominant mechanism for NLR family expansion in flowering plants [6]. Chromosomal distribution analyses consistently reveal significant NLR clustering, particularly near telomeric regions known for high recombination rates. In pepper, 18.4% of NLR genes (53/288) arose through tandem duplication events, predominantly on chromosomes 08 and 09 [6]. Similarly, in peanut genomes, asymmetric expansion of NLRomes between subgenomes of wild and domesticated tetraploids indicates lineage-specific duplication pressures [14].
Whole-genome duplication (WGD) events have also contributed substantially to NLR repertoire growth, particularly in polyploid species. The cultivated peanut (A. hypogaea), an allotetraploid, possesses approximately twice the number of NLR genes compared to its diploid progenitors [14]. However, following polyploidization, NLR repertoires often undergo differential gene loss between subgenomes, leading to asymmetric NLR distribution [14].
Evolutionary Arms Race and Selection Pressures
The "arms race" model between plants and their pathogens imposes strong diversifying selection on NLR genes, particularly in the LRR domain responsible for effector recognition [6]. This selective pressure drives rapid sequence diversification to recognize evolving pathogen effectors. Studies in Arachis species reveal that wild relatives subjected to natural pathogens maintain larger and more diverse NLR repertoires compared to domesticated varieties, highlighting the impact of differential selection pressures on NLR evolution [14].
MicroRNA-mediated regulation represents an additional evolutionary adaptation for managing expanded NLR repertoires. Numerous microRNAs target conserved NLR motifs (e.g., the P-loop) in flowering plants, potentially providing a mechanism to mitigate the fitness costs associated with maintaining large NLR inventories through transcriptional suppression [3].
Experimental Methodologies for NLR Gene Identification and Analysis
Genomic Identification Pipeline
Protocol 1: Genome-Wide NLR Identification and Classification
A standardized methodology for comprehensive NLR annotation incorporates both homology-based and domain-based approaches [13] [6] [14]:
Sequence Retrieval: Obtain complete proteome and genome assemblies from relevant databases (NCBI, Phytozome, Plaza, or species-specific resources).
Domain Identification: Employ HMMER searches against the Pfam database using the NB-ARC domain model (PF00931) with a stringent E-value cutoff (1 × 10⁻⁵ to 1.1 × 10⁻⁵⁰) [13] [6]. Alternatively, use NLRtracker, a specialized pipeline that integrates InterProScan and predefined NLR motifs [14].
Validation and Filtering: Confirm NB-ARC domain presence using NCBI Conserved Domain Database (cd00204) and remove redundant sequences [6].
Architecture Classification: Annotate N-terminal (TIR, CC, RPW8) and C-terminal (LRR) domains via InterProScan or manual curation. Classify sequences based on domain combinations [13].
Orthogroup Analysis: Perform clustering using OrthoFinder with DIAMOND for sequence similarity and MCL for clustering. Identify core and lineage-specific orthogroups [13].
Evolutionary and Expression Analyses
Protocol 2: Evolutionary Dynamics and Functional Validation
Phylogenetic Reconstruction: Extract NB-ARC domains and align with reference sequences using MUSCLE or MAFFT. Construct maximum likelihood trees with IQ-TREE using best-fit models and 1000 bootstrap replicates [13] [14].
Selection Pressure Analysis: Calculate non-synonymous to synonymous substitution rates (Ka/Ks) using codon-aligned sequences. Apply Fisher's test to identify significant positive selection (P < 0.01) [14].
Gene Duplication Assessment: Utilize MCScanX for synteny analysis to distinguish tandem from segmental duplications. Calculate Ks values for dating duplication events [6].
Expression Profiling: Analyze RNA-seq data under biotic stress conditions. Calculate FPKM values and identify differentially expressed NLRs (|log₂FC| ≥ 1, FDR < 0.05) using DESeq2 [13] [6].
Functional Validation: Implement Virus-Induced Gene Silencing (VIGS) to assess gene function. Quantify pathogen titers and defense marker expression in silenced plants [13].
Figure 1: NLR identification and functional analysis workflow.
NLR Signaling Mechanisms and Functional Diversification
Biochemical Execution of Immune Responses
Activated NLRs undergo conformational changes that facilitate ADP-to-ATP exchange within the NB-ARC domain, functioning as a molecular "on-off switch" [12]. This triggers the formation of higher-order oligomeric complexes (resistosomes) that enable N-terminal domains to perform immune-related biochemical functions:
CC-NLR resistosomes from Arabidopsis ZAR1 and wheat Sr35 form calcium-permeable cation channels targeting the plasma membrane. Their first alpha helix contains conserved "MADA" or "MADA-like" motifs essential for cell death induction [12].
TIR-NLR resistosomes (e.g., Arabidopsis RPP1 and Nicotiana ROQ1) assemble into tetrameric complexes with reconstituted NADase activity, generating immunogenic nucleotides (pRib-AMP/ADP, diADPR/ADPr-ATP) that activate EDS1 signaling pathways [12].
RPW8-type helper NLRs similarly associate with membranes, alter calcium flux, and require N-terminal motifs for cell death induction [12].
Evolutionary Conservation of Function
Despite extensive sequence diversification, the core immune functions of NLR domains appear conserved across land plants. Functional studies demonstrate that CC, RPW8, and TIR domains from streptophyte algae and nonflowering plants can activate cell death when expressed in the angiosperm Nicotiana benthamiana [12]. Nonflowering plant CC domains encode a distinct N-terminal "MAEPL" motif functionally analogous to the angiosperm "MADA" motif, suggesting conservation of pore-forming capability across 500 million years of plant evolution [12].
Figure 2: NLR immune signaling pathways and downstream effects.
Table 2: Key Research Reagents for NLR Studies
| Reagent/Resource | Function/Application | Examples/Specifications |
|---|---|---|
| Genome Assemblies | Reference sequences for NLR identification | Quality varies; chromosome-level preferred for duplication analyses [6] [14] |
| Pfam HMM Models | Domain identification and annotation | NB-ARC (PF00931), TIR (PF01582), CC/RPW8 detection [13] |
| NLRtracker Pipeline | Automated NLR annotation | Integrates InterProScan and predefined NLR motifs [14] |
| OrthoFinder | Orthogroup clustering and analysis | Identifies core and lineage-specific NLR groups [13] |
| PlantCARE Database | Cis-regulatory element prediction | Identifies defense-related promoter motifs [6] |
| STRING Database | Protein-protein interaction prediction | Models NLR signaling networks [6] |
| VIGS Vectors | Functional validation through gene silencing | TRV-based systems for rapid gene function assessment [13] |
| RNA-seq Datasets | Expression profiling under stress conditions | Biotic/abiotic stress time courses; differential expression [13] [6] |
The massive expansion and diversification of NLR repertoires in flowering plants represents a cornerstone of plant immune system evolution. From modest beginnings in early land plants, NLR genes have proliferated through tandem duplication, polyploidization, and diversifying selection, creating extensive pathogen recognition capacities that underlie species-specific resistance. The evolutionary arms race with pathogens continues to drive NLR diversification, while conserved signaling mechanisms and biochemical functions are maintained across deeply divergent plant lineages. Understanding these patterns of NLR evolution provides fundamental insights into plant-pathogen coevolution and enables strategic identification of resistance genes for crop improvement. Future research leveraging increasingly sophisticated genomic tools and functional characterization across diverse plant taxa will further illuminate the dynamic evolutionary processes that have shaped the plant immune repertoire.
The evolutionary trajectories of plant immune systems have diverged significantly between monocot and dicot lineages, resulting in distinct genetic and molecular strategies for pathogen defense. This divergence is particularly evident in the evolution of Nucleotide-binding Leucine-Rich Repeat (NLR) genes, which constitute one of the largest and most dynamic gene families in plants [15]. NLR genes encode intracellular immune receptors that recognize pathogen effectors and initiate effector-triggered immunity (ETI), providing specific resistance against diverse pathogens [16]. The investigation of lineage-specific patterns in NLR evolution is not merely an academic exercise but provides fundamental insights into the evolutionary arms race between plants and pathogens, with significant implications for crop improvement and sustainable agriculture [13].
This technical review synthesizes current understanding of the contrasting evolutionary trajectories in monocots and dicots, focusing on genomic architecture, gene family expansion/contraction, molecular mechanisms, and experimental approaches for investigating these lineage-specific patterns. By framing this discussion within the broader context of land plant evolution, we aim to provide researchers with a comprehensive resource for understanding how these two major angiosperm lineages have arrived at distinct solutions to the common challenge of pathogen defense.
Fundamental Divergences in NLR Repertoires
Genomic Distribution and Organization
The genomic organization of NLR genes reveals striking differences between monocots and dicots. NLR genes are typically distributed unevenly across chromosomes, with a strong tendency to cluster in specific genomic regions [15]. In monocots such as barley (Hordeum vulgare), chromosome 7 contains 112 NLR genes, approximately seven times the number found on chromosome 4 [17]. This irregular distribution is observed in both lineages but manifests differently due to variations in genome architecture and evolutionary history.
A key organizational difference lies in the physical arrangement of NLR genes. Across angiosperms, 68% of NLR genes are located in multigene clusters, facilitating rapid evolution through unequal crossing over and gene conversion [17]. However, the specific genomic contexts of these clusters differ between lineages. In monocots, NLR genes often reside in subtelomeric regions characterized by higher recombination frequencies, as observed in species such as wheat, barley, and Setaria italica [15]. This location promotes increased genetic diversity through enhanced recombination rates, potentially enabling more rapid adaptation to evolving pathogens.
Lineage-Specific Gene Family Dynamics
NLR gene families have experienced dramatically different evolutionary trajectories in monocots and dicots, reflected in both gene numbers and subclass composition (Table 1).
Table 1: Comparative Analysis of NLR Gene Repertoires in Representative Monocot and Dicot Species
| Species | Lineage | Total NLR Genes | TNL Genes | CNL Genes | RNL Genes | Reference |
|---|---|---|---|---|---|---|
| Hordeum vulgare (barley) | Monocot | 468 | 0 | 467 | 1 | [17] |
| Triticum aestivum (bread wheat) | Monocot | >2,000 | 0 | >2,000 | Not reported | [15] |
| Arabidopsis thaliana | Dicot | ~200 | ~100 | ~100 | Not reported | [15] |
| Malus domestica (apple) | Dicot | ~1,000 | ~330 | ~670 | Not reported | [15] [16] |
| Carica papaya | Dicot | 50-100 | ~25-50 | ~25-50 | Not reported | [15] |
| Vitis vinifera (grapevine) | Dicot | ~500 | ~100 | ~400 | Not reported | [15] |
The most striking difference between monocot and dicot NLR repertoires concerns the TIR-NLR (TNL) subclass. TNL genes are conspicuously absent from most monocot genomes, with few exceptions [17] [16]. In contrast, dicot genomes typically contain a substantial complement of TNL genes, with the ratio of CNL to TNL genes varying considerably among dicot families [15]. For example, Brassicaceae species exhibit a TNL to CNL ratio of approximately 2:1, while in potato and grapevine, the ratio is reversed to 1:4 [15]. Apple maintains a more balanced 1:1 ratio [15].
The remarkable expansion of NLR genes in monocot cereals is another key distinction. Bread wheat (Triticum aestivum) possesses over 2,000 NLR genes – the largest number reported in any plant species to date [15]. This expansion is partially attributable to polyploidy, but also to extensive lineage-specific duplications. Even diploid monocots like barley maintain substantial NLR repertoires (468 genes), comparable to other diploid cereals [17]. This pattern contrasts with most dicots, which generally possess more moderate NLR repertoires, though exceptions exist such as apple with nearly 1,000 NLR genes [16].
Molecular Mechanisms and Functional Innovations
Distinct Immune Receptor Architectures
Beyond differences in gene numbers and subclass distribution, monocots and dicots have evolved distinct structural innovations in their immune systems. A significant discovery in cereal immunity is the emergence of tandem kinase proteins (TKPs) and kinase fusion proteins (KFPs) as novel immune receptors [18]. These proteins typically feature two functional kinase domains fused in tandem and represent a major class of resistance genes in cereals.
Agronomically important TKPs include Pm24 (WTK3) for broad-spectrum powdery mildew resistance and Sr62 for stem rust resistance [18]. These TKPs often function in partnership with non-canonical NLRs, forming integrated immune hubs. For example, WTK3 partners with WTN1, an NLR with two tandem NB-ARC domains, creating a "sensor-executor" module where the TKP acts as the effector sensor and the NLR functions as the executioner [18]. Similarly, Sr62TK requires cooperation with Sr62NLR for resistance to stem rust [18]. These TKP-NLR pairs represent a distinct evolutionary pathway largely specific to monocots, particularly cereals.
Evolutionary Drivers and Constraints
Several evolutionary forces have shaped the divergent trajectories of monocot and dicot NLR genes. Birth-and-death evolution characterizes NLR gene families across angiosperms, with frequent gene duplications generating new specificities and pseudogenization eliminating obsolete genes [16]. However, the balance of these processes differs between lineages.
Phylogenetic analyses reveal that at least 18 ancestral CNL lineages were present in the common ancestor of barley, Triticum urartu, and Arabidopsis thaliana [17]. Following divergence, these lineages expanded differentially in monocot and dicot lineages. Fifteen ancestral lineages expanded to 533 sub-lineages prior to the divergence of barley and T. urartu, with the barley genome inheriting 356 of these sub-lineages that subsequently duplicated to the 467 CNL genes observed today [17].
The absence of TNL genes in most monocots represents a significant evolutionary puzzle. This absence may reflect lineage-specific constraints or the evolution of alternative mechanisms that fulfill TNL functions. Interestingly, some monocots possess RNL genes (RPW8-NLR), as evidenced by the identification of one RNL subclass gene in barley [17]. RNLs function in signaling rather than direct pathogen recognition and may represent a conserved backbone of NLR immune signaling across angiosperms.
Experimental Approaches for Comparative Analysis
Genomic Identification and Annotation
Comprehensive identification of NLR genes across species requires integrated bioinformatic approaches. The following workflow represents a standard methodology for NLR annotation:
Table 2: Experimental Protocol for Genome-Wide NLR Identification and Analysis
| Step | Method | Key Parameters | Purpose |
|---|---|---|---|
| 1. Sequence Identification | BLASTp and HMMER search | E-value = 1.0; Pfam NBS domain (PF00931) | Initial identification of candidate NLR genes |
| 2. Domain Validation | HMMscan against Pfam-A | E-value = 0.0001 | Confirm presence of NBS domain |
| 3. Domain Architecture Analysis | NCBI CDD, Motif Analysis (MEME) | 20 motifs default settings | Identify integrated domains and conserved motifs |
| 4. Chromosomal Distribution | Sliding window analysis | Window size: 250 kb | Identify NLR clusters and genomic organization |
| 5. Phylogenetic Analysis | Sequence alignment (ClustalW), Maximum likelihood (IQ-TREE) | Model selection by ModelFinder; SH-aLRT/UFBoot2 tests | Reconstruct evolutionary relationships |
| 6. Orthogroup Analysis | OrthoFinder, DIAMOND, MCL clustering | 30% identity, 70% overlap cutoffs | Identify conserved and lineage-specific NLR groups |
This methodology has been applied successfully in multiple studies investigating NLR diversity across land plants [13] [17]. Recent resources such as NLRscape provide curated collections of over 80,000 plant NLR sequences with advanced annotations, offering powerful platforms for comparative analyses [19]. Similarly, the ANNA (Angiosperm NLR Atlas) database contains over 90,000 NLR genes from 304 angiosperm genomes, enabling large-scale comparative studies [13].
Expression and Functional Validation
Following genomic identification, expression profiling and functional validation are essential for understanding NLR function. Methodologies include:
- Transcriptomic analysis: RNA-seq of different tissues under biotic and abiotic stresses to identify differentially expressed NLR genes [13]
- Virus-Induced Gene Silencing (VIGS): Functional validation through silencing candidate NLR genes in resistant genotypes [13]
- Co-expression assays: Transient expression in systems like Nicotiana benthamiana to test NLR activation upon effector recognition [18]
- Protein interaction studies: Yeast two-hybrid, co-immunoprecipitation, and structural predictions (AlphaFold) to characterize protein-protein interactions [18]
These approaches have revealed that NLR genes show distinctive expression patterns in response to pathogens, with some orthogroups (e.g., OG2, OG6, OG15) showing consistent upregulation under biotic stress across species [13].
Visualization of Evolutionary Relationships and Immune Mechanisms
NLR Lineage Divergence and Domain Architecture
Monocot-Specific TKP-NLR Immune Activation
Table 3: Research Reagent Solutions for Comparative NLR Genomics
| Resource Category | Specific Tools/Reagents | Function/Application | Example Use Cases |
|---|---|---|---|
| Genomic Databases | NLRscape, ANNA, PRGdb, RefPlantNLR | Curated NLR collections with annotations | Evolutionary analysis, orthogroup identification [19] [13] |
| Identification Tools | HMMER (Pfam domains), BLAST, MEME | NLR identification and motif discovery | Genome-wide NLR annotation [13] [17] |
| Phylogenetic Analysis | OrthoFinder, IQ-TREE, MEGA-X | Evolutionary relationship reconstruction | Phylogeny of NLR subclasses, orthogroup analysis [13] [17] |
| Expression Resources | RNA-seq databases, CottonFGD, IPF database | Expression profiling across tissues/stresses | Differential expression analysis of NLRs [13] |
| Functional Validation | VIGS, Co-expression assays, AlphaFold | Functional characterization of NLR genes | Validation of immune function [18] [13] |
| Structural Analysis | AlphaFold, Molecular modeling | Protein structure prediction | Interaction interface mapping [18] |
The contrasting evolutionary trajectories of NLR genes in monocots and dicots illustrate how fundamental developmental and genetic differences have shaped distinct pathogen defense strategies in these two major angiosperm lineages. Monocots have largely eliminated TNL genes while expanding CNL repertoires and evolving novel immune receptors such as TKPs. Dicots have maintained both major NLR subclasses while developing diverse integrated domains that expand pathogen recognition capabilities.
These lineage-specific patterns reflect deep evolutionary divergences that began with the separation of monocot and dicot lineages approximately 140-150 million years ago. The differential retention and expansion of NLR subclasses, coupled with the emergence of lineage-specific immune innovations, demonstrates how conserved molecular frameworks can be adapted to create distinct defensive strategies.
Future research directions should include comprehensive pan-NLRome studies across diverse species to fully capture intraspecific NLR diversity, structural characterization of novel immune receptors like TKPs, and investigation of how developmental differences between monocots and dicots constrain or facilitate immune system evolution. Such studies will not only advance fundamental understanding of plant immunity but also provide new resources for crop improvement through informed manipulation of NLR genes and their signaling networks.
The Role of Whole Genome Duplication and Tandem Repeats in NLR Proliferation
Nucleotide-binding leucine-rich repeat receptors (NLRs) constitute the largest family of plant disease-resistance (R) genes and serve as crucial intracellular immune receptors that mediate effector-triggered immunity (ETI) [6] [16]. These proteins typically feature a characteristic modular structure: a variable N-terminal domain (often TIR, CC, or RPW8), a central conserved nucleotide-binding adapter (NB-ARC or NBS) domain, and a C-terminal leucine-rich repeat (LRR) domain [20] [2]. The NLR gene family has undergone massive expansion in land plants, with numbers ranging from fewer than a dozen in green algae to over a thousand in some cultivated species like wheat and apple [20] [16]. This proliferation is primarily driven by two fundamental genetic mechanisms: whole genome duplication (WGD) and tandem duplication, which provide the raw genetic material for the evolution of novel disease resistance specificities [20] [21]. Understanding these duplication mechanisms is essential for harnessing NLR diversity to improve crop disease resistance.
Table 1: NLR Gene Repertoire Variation Across Plant Species
| Species | Genome Type | NLR Count | Key Duplication Mechanisms | References |
|---|---|---|---|---|
| Bread wheat (Triticum aestivum) | Hexaploid | >2000 | WGD, Tandem Duplication | [20] |
| Pepper (Capsicum annuum) | Diploid | 288 | Tandem Duplication (18.4% of NLRs) | [6] |
| Arabidopsis thaliana | Diploid | ~150 | Tandem Duplication, Segmental Duplication | [16] |
| Apple (Malus domestica) | Diploid | ~1000 | WGD, Tandem Duplication | [16] |
| Bladderwort (Utricularia gibba) | Diploid | Very low (~0.003% of genes) | Gene Loss | [16] |
| Cucurbita Species | Diploid | ~1850 (across 12 species) | WGD, Tandem Duplication | [21] |
Mechanisms of NLR Proliferation
Whole Genome Duplication (WGD)
Whole genome duplication represents the most extensive mechanism for NLR expansion, providing sudden increases in genetic material that can be shaped by evolutionary forces [20] [22]. WGD occurs through polyploidization events, where an organism acquires complete additional sets of chromosomes. In plants, WGD has played a fundamental role in the evolution of many crop species and their NLR repertoires.
The evolutionary history of hexaploid bread wheat (Triticum aestivum) exemplifies the impact of WGD on NLR proliferation. Wheat underwent two hybridization and polyploidization events, forming a new species with a huge genome and abundant gene set [20]. Approximately 55% of bread wheat homologous genes exhibit 1:1:1 correspondence across the three homologous subgenomes, while another 15% possess at least one gene copy in at least one of the subgenomes [20]. This complex evolutionary history has contributed to wheat possessing one of the largest and most diverse NLR repertoires among cultivated plants, with over 1500 NLRs detected in some studies and more than 2000 identified using fully annotated reference genomes [20].
Similar patterns are observed in the Fabaceae family, where ancestors underwent whole genome duplication approximately 58.5 million years ago [22]. Subsequent analysis of the Vicioid clade (including chickpea, clover, alfalfa, and pea) revealed that the initial WGD was followed by differential evolutionary trajectories in different tribes. While Cicereae and Fabeae tribes experienced overall contraction of their NLRomes (complete sets of NLR genes), the Trifolieae tribe showed large-scale expansion regardless of genome size [22]. This expansion in Trifolieae occurred relatively recently (during the past 1-6 million years), likely driven by higher substitution rates that accelerated gene duplications after speciation [22].
Tandem Duplication
Tandem duplication occurs when two or more genes become positioned adjacent to each other on the same chromosome following duplication events [20] [6]. This mechanism is particularly significant for NLR gene expansion and the formation of NLR gene clusters in plant genomes [20].
Research in pepper (Capsicum annuum) demonstrates that tandem duplication serves as the primary driver of NLR family expansion, accounting for 18.4% of NLR genes (53 out of 288) [6]. These tandem duplications predominantly occur on chromosomes 08 and 09, with Chr09 harboring the highest density of NLRs (63 genes), often clustered near telomeric regions [6]. Similar patterns of clustering are observed in rice, where numerous NLRs cluster near chromosomal telomeres, facilitating rapid generation of new resistance alleles through local amplification [6].
The proliferation of NLRs through tandem duplication creates genomic environments conducive to further evolution. These cluster arrangements enable mechanisms such as gene conversion and asymmetric recombination, which contribute to subgroup diversification and the generation of novel resistance specificities [22]. This dynamic process results in NLR genes that are highly variable between ecotypes and cultivars, with cluster size and composition differing drastically even among closely related varieties [20] [23].
Complementary Evolutionary Forces
While WGD and tandem duplication are primary mechanisms for NLR expansion, several complementary forces shape the evolutionary trajectory of duplicated genes:
Birth-and-Death Evolution: NLR genes undergo rapid turnover, with frequent gene births (through duplication) and deaths (through pseudogenization or deletion) [21] [16]. This process constantly remodels the NLR repertoire.
Diploidization: Following WGD, genomes undergo diploidization, a process of gene loss and reorganization that returns the genome to a more diploid-like state [22]. This process explains why some lineages experience NLR contraction after WGD.
Transposable Element Activity: Replicative transposition by transposable elements forms dispersed duplicates that contribute to NLR diversity [20]. Research in Triticeae tribe species revealed a recent burst of gene duplications potentially linked to transposable element activity [20].
Diagram 1: NLR proliferation mechanisms and outcomes. Whole Genome Duplication (WGD) and Tandem Duplication drive NLR expansion, leading to functional divergence and enhanced pathogen recognition.
Structural and Functional Consequences of NLR Proliferation
Impact on NLR Architecture and Diversity
The duplication-mediated expansion of NLR genes has profound implications for their structural diversity and functional capabilities. The modular architecture of NLR proteins enables extensive functional diversification through domain-specific evolutionary pressures [16].
The N-terminal domain, which can be TIR, CC, or RPW8, serves as the primary structural element for signal transduction [20] [16]. The central NB-ARC domain functions as a molecular switch, cycling between ADP- (inactive) and ATP-bound (active) states [20] [2]. The C-terminal LRR domain, with its hypervariable tandem repeats, is primarily responsible for effector recognition and demonstrates the most rapid evolution [6] [16]. This domain organization creates multiple axes along which duplication-derived variations can generate novel functions.
Recent research has revealed that NLR diversity arises from multiple uncorrelated mutational and genomic processes [23]. Pangenomic studies in Arabidopsis thaliana have identified 3,789 NLRs across 17 diverse accessions, distributed across 121 pangenomic NLR neighborhoods that vary substantially in size, content, and complexity [23]. This diversity across multiple axes suggests that "diversity in diversity generation" is fundamental to maintaining a functionally adaptive immune system in plants [23].
Functional Specialization and Resistance Specificity
The proliferation of NLR genes through duplication enables several pathways to novel resistance specificities:
Neofunctionalization: Duplicated NLR genes accumulate mutations that confer recognition of new pathogen effectors [6] [21].
Subfunctionalization: Duplicated copies partition ancestral functions, potentially leading to specialization against different pathogen strains [21].
Helper/Sensor Systems: Some NLR subclasses, such as CCR-NLR and CCG10-NLR, have diversified into helper and sensor roles that function in coordinated networks [22].
Research in Cucurbita species revealed an unusual diversification of CNL/TNL genes alongside strong RNL conservation, indicating that different NLR subclasses experience distinct evolutionary pressures [21]. This differential evolution creates species-specific NLR compositions that reflect each species' unique pathogen exposure history.
Table 2: Evolutionary Patterns of NLR Subclasses
| NLR Subclass | N-terminal Domain | Evolutionary Pattern | Functional Role | Examples |
|---|---|---|---|---|
| TNL (TIR-NLR) | Toll/Interleukin-1 Receptor | Lineage-specific losses (e.g., in monocots); Rapid diversification | Effector recognition; Cell death signaling | Arabidopsis RPP1, Tobacco N [20] [16] |
| CNL (CC-NLR) | Coiled-Coil | Widespread conservation with expansion | Effector recognition; Resistosome formation | Arabidopsis ZAR1, Potato Rx [20] [16] |
| RNL (RPW8-NLR) | RPW8-like CC | Strong conservation across species | Helper function; Signal transduction | NRG1, ADR1 [21] [22] |
Research Methodologies for Studying NLR Proliferation
Genome-Wide Identification of NLR Genes
Comprehensive characterization of NLR proliferation requires precise identification and annotation of NLR genes across plant genomes. The following integrated methodology has been successfully applied in multiple studies [6] [21]:
Step 1: Initial Sequence Identification
- Retrieve reference NLR protein sequences from databases (e.g., TAIR for Arabidopsis)
- Perform BLASTp searches against target species proteomes
- Conduct HMMER searches (v3.3.2) using core NLR domain models (PF00931 for NB-ARC) with E-value cutoff of 1×10⁻⁵
Step 2: Domain Validation and Classification
- Validate candidate sequences containing NB-ARC domains using NCBI CDD (cd00204) and Pfam batch search
- Check for presence/completeness of N-terminal (TIR, CC, RPW8) and C-terminal (LRR) domains
- Classify NLRs into subclasses (TNL, CNL, RNL) based on domain architecture
Step 3: Phylogenetic and Evolutionary Analysis
- Align NB-ARC domain or full-length sequences using Muscle v5 or similar tools
- Construct Maximum Likelihood trees (IQ-TREE with 1000 bootstrap replicates)
- Use related species NLRs as outgroups for phylogenetic placement
Step 4: Gene Duplication Analysis
- Perform synteny analysis using MCScanX implemented in TBtools
- Identify tandem duplicates as adjacent NLR genes on the same chromosome
- Detect segmental duplicates through interchromosomal synteny analysis
- Generate visualization using Advanced Circos (TBtools v2.360)
Diagram 2: Experimental workflow for studying NLR proliferation. The integrated methodology progresses from gene identification to functional validation through bioinformatic and experimental approaches.
The Scientist's Toolkit: Essential Research Reagents and Solutions
Table 3: Key Research Reagents and Solutions for NLR Proliferation Studies
| Research Tool | Specific Examples | Function/Application | References |
|---|---|---|---|
| Genome Databases | CuGenDB, TAIR, PRGdb | Source of reference sequences and annotated genomes | [6] [21] |
| Domain Analysis Tools | NCBI CDD (cd00204), Pfam (PF00931), MEME/MAST | Identification and validation of NLR domains and motifs | [6] [21] |
| Synteny Analysis Software | MCScanX, TBtools v2.360, Dual Synteny Plotter | Identification of duplicated genes and evolutionary relationships | [6] [22] |
| Phylogenetic Tools | IQ-TREE, Muscle v5 | Reconstruction of evolutionary history and classification | [6] |
| Expression Analysis | RNA-seq (e.g., SRR9883231, SRR9883230), RT-qPCR, DESeq2 | Differential expression analysis under pathogen challenge | [6] |
| Interaction Networks | STRING database, PPI prediction | Protein-protein interaction network analysis | [6] |
Evolutionary Patterns and Ecological Adaptation
The proliferation of NLR genes through duplication is not uniform across plant lineages but demonstrates striking patterns of expansion and contraction correlated with ecological adaptation. The angiosperm NLR Atlas (ANNA), which includes NLR genes from over 300 angiosperm genomes, reveals that NLR copy numbers differ by up to 66-fold among closely related species due to rapid gene loss and gain [24].
A particularly revealing pattern emerges in plants with specialized ecological strategies. Convergent NLR reduction is associated with adaptations to aquatic, parasitic, and carnivorous lifestyles [24]. The NLR contraction observed in aquatic plants resembles the lack of NLR expansion during the long-term evolution of green algae before the colonization of land, suggesting that pathogen pressure may be reduced in aquatic environments [24]. This pattern highlights how ecological context shapes the evolutionary trajectory of the plant immune system.
Co-evolutionary patterns between NLR subclasses and components of plant immune pathways have also been identified. For instance, deficiencies in the EDS1–SAG101–NRG1 module, which is required for TNL signaling, may drive TNL loss in certain lineages [24]. Conversely, researchers have identified a conserved TNL lineage that may function independently of this module, illustrating how genetic compensation can enable divergent evolutionary paths [24].
These evolutionary patterns demonstrate that NLR proliferation is not merely a consequence of random duplication events but reflects complex interactions between genomic dynamics, immune system requirements, and ecological specialization.
Whole genome duplication and tandem duplication have played complementary and crucial roles in the proliferation of NLR genes throughout plant evolution. WGD provides dramatic increases in genetic material through polyploidization events, while tandem duplication enables rapid, localized expansion that generates diverse NLR clusters, particularly in genomic regions such as telomeres [20] [6]. These mechanisms collectively provide the raw material for the birth-death evolution that characterizes NLR gene families, enabling plants to continuously adapt to evolving pathogen pressures [21] [16].
The functional consequences of NLR proliferation extend beyond simple increases in gene numbers to encompass substantial structural and functional diversification. Through processes such as neofunctionalization and subfunctionalization, duplicated NLR genes evolve novel recognition specificities and specialized functions [6] [21]. This diversification occurs across multiple axes, creating complex pangenomic NLR neighborhoods that vary substantially between accessions and species [23]. The resulting NLR repertoires represent dynamic balances between expansion through duplication and contraction through gene loss, with the equilibrium shaped by both evolutionary history and ecological context [24] [22].
For researchers and crop improvement programs, understanding these duplication mechanisms provides valuable insights for harnessing NLR diversity. The identification of rapidly diversifying NLR clusters can guide mining of novel resistance specificities from wild relatives and landraces [6] [21]. Furthermore, elucidating the patterns of NLR proliferation informs strategies for deploying resistance genes in breeding programs, potentially enabling more durable disease control through pyramiding of effective NLR combinations. As genomic technologies continue to advance, the ability to precisely track and engineer NLR proliferation will become increasingly powerful for developing crops with enhanced resistance to evolving pathogen threats.
Genomic Mining and Analytical Frameworks for Deciphering NLR Evolution and Function
Nucleotide-binding leucine-rich repeat receptors (NLRs) constitute one of the most diverse and critical gene families in plant innate immunity, serving as intracellular sensors that detect pathogen effectors and trigger robust defense responses such as the hypersensitive response [25]. These immune receptors follow a modular tri-partite structure typically consisting of an N-terminal coiled-coil (CC) or Toll/interleukin-1 receptor (TIR) domain, a central nucleotide-binding adaptor (NB-ARC) domain, and C-terminal leucine-rich repeats (LRRs) [25] [16]. The evolutionary dynamics of NLR genes are characterized by remarkable sequence diversification and rapid evolution, reflecting the continuous arms race between plants and their pathogens [25]. This diversification enables plants to recognize a wide spectrum of fast-evolving pathogen-derived molecules, making NLRs a fascinating subject for evolutionary genomics studies in land plants.
The development of advanced bioinformatics pipelines has revolutionized our ability to identify, classify, and analyze NLR genes across plant species. Traditional multiple sequence alignment methods often encounter technical challenges with large NLR datasets due to extensive sequence diversity, gaps, and deletions [25]. Modern computational pipelines now integrate diverse bioinformatics tools including HMMER for domain identification, OrthoFinder for phylogenetic orthology inference, and pangenome graphs for capturing species-wide structural variations. These approaches have enabled researchers to move beyond single-reference genomics to pangenome perspectives that capture the full repertoire of NLR diversity within species [26]. This technical guide provides an in-depth overview of these advanced bioinformatics methodologies within the context of NLR gene evolution in land plants, offering detailed protocols and resources for researchers investigating plant immune receptor evolution.
Core Bioinformatics Pipeline for NLR Identification
Integrated Workflow for NLR Annotation and Analysis
A comprehensive NLR identification pipeline integrates multiple bioinformatics tools to overcome challenges posed by the sequence diversity and complex evolutionary history of this gene family. The pipeline progresses through distinct phases: initial sequence identification, domain annotation, phylogenetic analysis, orthology inference, and pangenome construction [25] [27] [26]. Each phase employs specialized tools optimized for specific aspects of NLR characterization, working synergistically to provide a complete picture of NLRome composition and evolution.
Figure 1: Comprehensive bioinformatics workflow for NLR identification and evolutionary analysis, showing the integration of domain annotation, phylogenetic reconstruction, orthology inference, and pangenome construction.
Domain Annotation and NLR Identification Tools
HMMER and InterProScan for Domain Characterization The initial identification of NLR genes relies heavily on domain annotation using hidden Markov models (HMMs). The HMMER software package provides hmmscan and hmmsearch utilities for identifying NB-ARC domains (Pfam: PF00931) in proteome datasets with an E-value cutoff of 10⁻⁴ [5]. This step is often complemented by BLASTp searches against reference NLR sequences (E-value = 1.0) to ensure comprehensive candidate identification [5]. Subsequently, InterProScan provides additional functional characterization by integrating multiple domain databases, confirming NLR identity through cross-referenced domain architecture analysis [25] [27]. This combined approach ensures high sensitivity in detecting canonical NLR domains while minimizing false positives.
Specialized NLR Annotation Tools Specialized tools have been developed specifically for plant NLR annotation. NLRtracker utilizes a protein sequence file as input and integrates InterProScan for comprehensive domain characterization [25]. It demonstrates higher sensitivity and accuracy compared to previous tools, successfully detecting functionally validated NLRs that may be missed by other methods. Alternatively, NLR-Annotator operates on nucleotide sequence files, making it suitable for users without access to Linux systems [25]. For downstream analysis, NLR-parser can be employed to identify genes containing NB-ARC domains for gene family-specific pangenome analysis [26]. These tools collectively enable researchers to extract NLR sequences from given plant proteomes or genomes with high confidence, providing the foundation for subsequent evolutionary analyses.
Table 1: Software Tools for NLR Identification and Analysis
| Tool | Function | Input | Key Features | Citation |
|---|---|---|---|---|
| HMMER | Domain identification | Protein sequences | Identifies NB-ARC domains using HMM profiles | [5] |
| InterProScan | Protein function characterization | Protein sequences | Integrates multiple domain databases | [25] |
| NLRtracker | NLR annotation | Protein sequences | High sensitivity, detects functionally validated NLRs | [25] [27] |
| NLR-Annotator | NLR annotation | Nucleotide sequences | Suitable for non-Linux users | [25] |
| NLR-parser | NLR gene family identification | Protein sequences | Creates gene family-specific pangenomes | [26] |
Phylogenetic Analysis and Motif Discovery
Multiple Sequence Alignment and Tree Building Phylogenetic analysis forms the cornerstone of NLR evolutionary studies, enabling classification into subfamilies and identification of evolutionary relationships. MAFFT performs multiple sequence alignment of identified NLR sequences, handling the challenges posed by their diversity through sophisticated algorithms [25]. For phylogenetic tree construction, RAxML implements maximum likelihood-based inference of large phylogenetic trees, while IQ-TREE provides an alternative with model selection capabilities [25] [27]. These tools typically use the NB-ARC domain sequences for phylogenetic reconstruction due to their relative conservation compared to other NLR domains, providing a stable framework for classifying NLRs into subgroups such as TIR-NLRs, CC-NLRs, CCR-NLRs, and the G10 subclade [27] [14].
Motif Discovery with MEME Suite The MEME Suite enables discovery of conserved sequence motifs that may not be apparent through standard domain annotation. This tool identifies evolutionarily conserved patterns such as the MADA and EDVID motifs within the CC-NLR subfamily [25]. MEME analysis can be performed either through the web interface or by installing the software locally, with parameters typically set to identify 10 motifs using default settings [25] [28]. This approach has been instrumental in characterizing novel conserved sequence patterns crucial for NLR function, particularly for understanding molecular features that have remained conserved across evolutionary time despite overall sequence diversification [25].
Orthology Inference with OrthoFinder
OrthoFinder implements a sophisticated phylogenetic orthology inference algorithm that extends beyond simple similarity scores to provide gene trees, rooted species trees, and gene duplication events [29]. The method addresses key challenges in orthology inference through five major steps: (1) orthogroup inference from sequence similarity scores, (2) inference of gene trees for each orthogroup, (3) analysis of gene trees to infer the rooted species tree, (4) rooting of gene trees using the species tree, and (5) duplication-loss-coalescence analysis of rooted gene trees to identify orthologs and gene duplication events [29]. This comprehensive approach is particularly valuable for NLR genes, as it can distinguish recent duplications from ancient diversification events and clarify orthology relationships despite variable evolutionary rates.
The default implementation uses DIAMOND for accelerated sequence similarity searches, followed by DendroBLAST for gene tree inference, balancing accuracy with computational efficiency [29]. However, OrthoFinder's modular design allows customization with alternative multiple sequence alignment (e.g., MUSCLE, MAFFT) and tree inference methods (e.g., RAxML, IQ-TREE) to suit specific research needs and computational resources [29]. For NLR analyses, OrthoFinder has demonstrated superior performance in ortholog inference accuracy, outperforming other methods by 3-24% on standardized benchmarks [29], making it particularly valuable for comparative analyses of NLR genes across multiple plant species.
Protocol for NLR Orthology Analysis
A typical OrthoFinder analysis for NLR genes follows these steps:
Input Preparation: Compile protein sequences from species of interest in FASTA format. For comprehensive NLR analysis, include reference sequences from known functional NLRs.
Orthogroup Inference: Run OrthoFinder with default parameters:
orthofinder -f [input_directory] -t [number_of_threads]. This performs all-vs-all sequence similarity searches and identifies orthogroups.Gene Tree and Species Tree Inference: OrthoFinder automatically infers gene trees for each orthogroup and reconstructs the rooted species tree from these gene trees.
Ortholog Identification: The software identifies orthologs between all species pairs using the phylogenetic relationships from the gene trees.
Gene Duplication Events: OrthoFinder maps gene duplication events to both the species tree and gene trees, providing crucial information for understanding NLR expansion mechanisms.
Output Analysis: Key outputs include: (a) orthogroups and their statistical summary, (b) orthologs between species pairs, (c) gene trees for all orthogroups, (d) rooted species tree, (e) gene duplication events, and (f) comparative genomics statistics [29].
For NLR-specific analyses, researchers often supplement this pipeline with additional clustering using tools like MCL with an identity threshold of 50% to further resolve relationships within NLR subgroups [27] [14].
Pangenome Graph Construction for NLRomes
Conceptual Framework and Implementation
Pangenome graphs represent genetic variation within a species by combining genomes of multiple individuals to identify genomic variations from single nucleotide polymorphisms to major structural variations [26]. In the context of NLR genes, pangenome graphs enable researchers to capture the full diversity of NLR sequences (the "NLRome") within a species, including presence-absence variations (PAVs), copy number variations (CNVs), and novel NLR alleles not present in reference genomes [26]. The pangenome is conceptually divided into the "core" genome (genes shared by all individuals) and "dispensable" genome (genes present in only a subset of individuals), with NLR genes frequently enriched in the dispensable component due to their rapid evolution [26].
The construction of pangenome graphs for NLRomes can be approached through linear pangenomes, which concatenate consensus sequences from multiple genomes, or graph-based pangenomes that explicitly represent variation as alternative paths [26]. Graph-based approaches are particularly powerful for NLR analysis as they naturally capture structural variations and presence-absence polymorphisms that characterize the evolution of this gene family. These approaches have revealed substantial NLR variation even between closely related cultivars, as demonstrated in sorghum where resistant and susceptible cultivars showed significant differences in NLR gene content (302 vs 239 NLR genes) [30].
Pangenome Analysis Workflow for NLR Genes
Figure 2: Pangenome graph construction workflow for NLRome analysis, showing the process from multiple genome assemblies through variant identification to evolutionary analysis.
The implementation of NLR pangenome analysis involves several key steps:
Genome Assembly Collection: Obtain high-quality genome assemblies for multiple individuals representing the genetic diversity of the species. Third-generation sequencing technologies (PacBio, Oxford Nanopore) have significantly improved assembly continuity, particularly for complex NLR regions [26].
NLR Identification and Annotation: Identify NLR genes in each genome using the pipeline described in Section 2, ensuring consistent annotation across all assemblies.
Pangenome Construction: Use pangenome construction tools to build either linear or graph-based pangenomes. For initial explorations, linear pangenomes provide simpler visualization, while graph-based pangenomes more accurately capture structural variation.
Variant Calling: Identify presence-absence variations (PAVs), copy number variations (CNVs), and other structural variations affecting NLR genes across the pangenome.
Classification: Categorize NLR genes into core (present in all individuals), shell (present in 5-94%), and cloud (present in 1-5%) components based on their distribution frequency [26].
Evolutionary Analysis: Analyze patterns of gene gain and loss, positive selection, and evolutionary dynamics across the NLRome.
A critical consideration in pangenome analysis is determining whether the NLRome is "open" or "closed" using Heaps' Law, which describes the relationship between newly sequenced individuals and the discovery of novel NLR genes [26]. This has practical implications for breeding programs, as species with open pangenomes may offer greater potential for discovering novel resistance genes from wild relatives or diverse landraces.
Evolutionary Insights from Integrated Bioinformatics Approaches
NLR Evolution Patterns Across Plant Species
The application of integrated bioinformatics pipelines has revealed remarkable diversity in NLR gene content across land plants, ranging from fewer than a dozen in green algae to many hundreds in angiosperms [16]. This expansion represents an evolutionary response to pathogen pressures as plants diversified and colonized new environments. Comparative genomic analyses have identified distinctive evolutionary patterns in different plant lineages, including contraction in Poaceae species, consistent expansion in Fabaceae species, and initial expansion followed by contraction in Brassicaceae species [5]. These patterns reflect both phylogenetic constraints and ecological adaptations shaping NLR repertoire evolution.
Table 2: NLR Gene Distribution Across Plant Species
| Plant Species | NLR Count | Genome Size | Key Evolutionary Features | Citation |
|---|---|---|---|---|
| Arabidopsis thaliana | ~200 | ~135 Mb | Model for NLR function and evolution | [25] |
| Oryza sativa (rice) | ~500 | ~430 Mb | NLR contraction in Poaceae | [5] |
| Trifolium pratense | 350 | ~300 Mb | NLR expansion in Fabaceae | [27] |
| Arachis cardenasii | 521 | ~1.2 Gb | Wild relative with extensive NLR diversity | [14] |
| Asparagus officinalis | 27 | ~690 Mb | Domesticated species with NLR contraction | [28] |
| Sorghum bicolor (BTx623) | 302 | ~730 Mb | Disease-resistant cultivar with expanded NLRome | [30] |
Polyploidization events have played a particularly important role in NLR evolution, as demonstrated in allopolyploid species like white clover (Trifolium repens) and cultivated peanut (Arachis hypogaea). In these species, NLRomes often evolve asymmetrically between subgenomes, with one subgenome showing expansion while the other undergoes contraction [27] [14]. This asymmetric evolution may result from distinct natural and artificial selection pressures acting on different subgenomes following polyploidization. Domesticated species frequently show NLR contraction compared to their wild relatives, as observed in asparagus where cultivated A. officinalis contains only 27 NLR genes compared to 63 in wild A. setaceus [28]. This pattern likely reflects artificial selection for yield and quality traits during domestication, potentially at the expense of defensive capabilities.
Case Studies in NLR Evolution
Legume NLR Evolution The Fabaceae family demonstrates particularly interesting patterns of NLR evolution. Studies in genus Arachis revealed that wild and domesticated tetraploid species show asymmetric expansion of NLRomes in both subgenomes, with the A-subgenome of wild A. monticola exhibiting contraction while the B-subgenome shows expansion, and the opposite pattern in domesticated A. hypogaea [14]. This suggests distinct evolutionary pressures acting on wild and cultivated species. Similarly, in genus Trifolium, specific NLR subgroups (G4-CNL, CCG10-CNL, TIR-CNL) show distinct duplication patterns in specific species, indicating subgroup-specific duplications that are hallmarks of divergent evolution [27]. The overall expansion of NLR repertoire in T. subterraneum appears driven by gene duplication events and birth of new gene families after speciation [27].
Apiaceae NLR Dynamics Comparative analysis of four Apiaceae species (Angelica sinensis, Coriandrum sativum, Apium graveolens, and Daucus carota) revealed dynamic evolutionary patterns of NLR genes, with counts ranging from 95 in A. sinensis to 183 in C. sativum [5]. Phylogenetic analysis demonstrated that NLR genes in these species were derived from 183 ancestral NLR lineages and experienced different levels of gene-loss and gain events during speciation [5]. While D. carota showed contraction of ancestral NLR lineages, the other three species exhibited a pattern of contraction after initial expansion of NLR genes [5]. These findings illustrate how rapid and dynamic gene content variation has shaped the evolutionary history of NLR genes even within a single plant family.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Bioinformatics Tools and Resources for NLR Analysis
| Tool/Resource | Category | Function in NLR Research | Access |
|---|---|---|---|
| NLRtracker | NLR Annotation | Annotates NLRs from protein sequences with high sensitivity | https://github.com/slt666666/NLRtracker [25] |
| OrthoFinder | Orthology Inference | Infers orthogroups, gene trees, and species trees from proteomes | https://github.com/davidemms/OrthoFinder [29] |
| MEME Suite | Motif Discovery | Identifies conserved sequence motifs in NLR proteins | https://meme-suite.org [25] |
| InterProScan | Domain Annotation | Characterizes protein domains and functional sites | https://www.ebi.ac.uk/interpro/download/ [25] |
| MAFFT | Sequence Alignment | Multiple sequence alignment for diverse NLR sequences | https://mafft.cbrc.jp/alignment/software/ [25] |
| IQ-TREE | Phylogenetics | Maximum likelihood tree inference with model selection | http://www.iqtree.org [27] |
| PlantCARE | cis-Element Analysis | Identifies regulatory elements in NLR gene promoters | http://bioinformatics.psb.ugent.be/webtools/plantcare/ [28] |
| PRGdb | NLR Database | Curated database of plant resistance genes | http://prgdb.org [28] |
Advanced bioinformatics pipelines integrating HMMER, OrthoFinder, and pangenome graphs have fundamentally transformed our understanding of NLR gene evolution in land plants. These approaches have revealed the remarkable diversity, rapid evolution, and complex evolutionary dynamics that characterize this crucial gene family. The integration of these tools enables researchers to move beyond single-reference genomics to capture the full spectrum of NLR diversity within and between species, providing insights into how plants adapt to evolving pathogen pressures through genomic innovation.
As sequencing technologies continue to advance and computational methods become more sophisticated, these pipelines will further refine our ability to connect NLR sequence diversity with functional capabilities. Future developments in graph-based pangenomes, machine learning approaches for predicting NLR function, and integration with expression and epigenetic data will provide even deeper insights into the evolutionary ecology of plant immunity. These advances will support crop improvement efforts by enabling more precise identification and deployment of NLR genes for durable disease resistance, ultimately contributing to global food security in the face of evolving pathogen threats.
Nucleotide-binding leucine-rich repeat (NLR) genes constitute the largest and most critical family of plant disease resistance (R) genes, encoding intracellular immune receptors that recognize pathogen-derived effectors and activate effector-triggered immunity (ETI) [5]. These genes are characterized by a conserved nucleotide-binding arc (NB-ARC) domain and C-terminal leucine-rich repeats (LRRs), with variable N-terminal domains classifying them into major subclasses: CNL (coiled-coil), TNL (Toll/interleukin-1 receptor), and RNL (RPW8) proteins [5]. NLR genes are now recognized as one of the most dynamic and rapidly evolving gene families in plant genomes, exhibiting remarkable variation in copy number, structural diversity, and evolutionary patterns across species [24].
The comparative genomic analysis of NLR contraction and expansion across plant species provides crucial insights into the evolutionary arms race between plants and their pathogens. Recent studies have revealed that NLR genes can vary up to 66-fold among closely related species due to rapid gene loss and gain events [24]. Understanding these dynamic evolutionary patterns is essential for uncovering the genetic basis of disease resistance and for developing sustainable crop protection strategies. This technical guide examines the mechanisms, methodologies, and evolutionary implications of NLR gene family dynamics within the broader context of land plant evolution.
Methodological Framework for Comparative NLR Genomics
Genome-Wide Identification and Classification of NLR Genes
The accurate identification and classification of NLR genes across multiple genomes form the foundation for comparative analysis. A standardized pipeline has emerged across recent studies, combining multiple complementary approaches to ensure comprehensive NLR detection [28] [5] [9].
Core Identification Protocol:
- Hidden Markov Model (HMM) Searches: Perform HMM searches using the conserved NB-ARC domain (Pfam: PF00931) as query against target proteomes with an E-value cutoff of 1e-10 [28] [5]. The HMM profile is downloaded from the Pfam database and used with HMMER software.
- BLASTp Analyses: Conduct local BLASTp searches against reference NLR protein sequences from well-annotated species (e.g., Arabidopsis thaliana, Oryza sativa) using stringent E-value cutoffs (1e-10) [28].
- Domain Architecture Validation: Validate candidate sequences through domain architecture analysis using InterProScan and NCBI's Batch CD-Search, retaining only sequences containing the NB-ARC domain (E-value ≤ 1e-5) as bona fide NLR genes [28].
- Subclassification: Classify NLR genes into subfamilies (CNL, TNL, RNL) based on N-terminal domains by querying Pfam and PRGdb 4.0 databases [28].
Recent studies have increasingly utilized specialized tools like NLRtracker, which employs canonical features of functionally characterized plant NLR genes for high-throughput annotation [27] [9] [14]. This tool uses InterProScan and predefined NLR motifs to extract NLRs and provide domain architecture analyses, though manual curation remains necessary for certain subclasses like CCR-NLR [14].
Phylogenetic Reconstruction and Evolutionary Analysis
Phylogenetic analysis of NLR genes provides insights into evolutionary relationships and duplication events:
Standardized Phylogenetic Workflow:
- Domain Extraction: Extract amino acid sequences of the NBS domain from identified NLR genes [5] [31].
- Multiple Sequence Alignment: Perform alignment using Clustal Omega, ClustalW, or MUSCLE with default parameters [28] [27].
- Model Selection: Identify best-fit substitution models using ModelFinder [5] [27].
- Tree Construction: Construct maximum likelihood trees using IQ-TREE or MEGA with branch support assessed through SH-aLRT, UFBoot2 tests, or bootstrap analysis (typically 1000 replicates) [28] [5] [31].
- Reconciliation Analysis: Reconcile gene trees with species trees using Notung software to identify gene duplication and loss events [5] [31].
Synteny and Cluster Analysis
NLR genes are frequently organized in clusters, and their genomic arrangement provides insights into evolutionary mechanisms:
Cluster Identification Protocol:
- Define NLR clusters using a distance-based criterion: genes located within 250 kb on a chromosome are considered cluster members [31].
- Determine gene orientations (head-to-head, head-to-tail, tail-to-tail) using BEDTools [28].
- Perform sliding-window analysis with 250 kb windows to identify singleton versus clustered loci [5].
- Analyze syntenic relationships between species using MCScanX or similar tools to identify conserved and species-specific NLR regions [31] [9].
Table 1: Standard Bioinformatics Tools for NLR Comparative Genomics
| Tool Category | Specific Tools | Primary Function | Key Parameters |
|---|---|---|---|
| NLR Identification | HMMER, NLRtracker, BLAST+ | Identify NLR candidates from genomic data | E-value ≤ 1e-10, NB-ARC domain (PF00931) |
| Domain Analysis | InterProScan, NCBI CD-Search | Validate domain architecture | E-value ≤ 1e-5 |
| Phylogenetic Analysis | IQ-TREE, MEGA, Clustal | Reconstruct evolutionary relationships | Bootstrap ≥ 1000, best-fit model selection |
| Synteny & Cluster Analysis | MCScanX, BEDTools, OrthoFinder | Identify gene clusters and orthologs | Window size: 250 kb for clusters |
| Orthology Analysis | OrthoFinder, OrthoVenn2 | Determine orthologous groups | E-value 1e-2, inflation parameter 1.5 |
Evolutionary Rate and Selection Analysis
To understand selection pressures acting on NLR genes:
Selection Analysis Protocol:
- Sequence Alignment: Align protein sequences of paralog groups using ClustalW.
- Codon Alignment: Convert protein alignment to codon alignment using pal2nal.
- Evolutionary Rate Calculation: Calculate Ka/Ks ratios using the Ka/Ks calculator with the MA method.
- Statistical Testing: Apply Fisher's test (p > 0.01) to identify significant duplication events.
- Data Filtering: Exclude Ks values >2 due to potential substitution saturation [27] [14].
The following workflow diagram illustrates the comprehensive pipeline for NLR comparative genomics:
Evolutionary Patterns of NLR Genes Across Plant Lineages
Comparative genomic analyses across diverse plant taxa have revealed distinct evolutionary patterns of NLR genes, influenced by life history, ecological adaptation, and domestication.
NLR Contraction in Domesticated and Specialized Species
Asparagus Genus Studies: A comprehensive analysis of NLR genes in garden asparagus (Asparagus officinalis) and its wild relatives (A. setaceus and A. kiusianus) revealed significant NLR contraction during domestication [28]. The study identified 63, 47, and 27 NLR genes in A. setaceus, A. kiusianus, and A. officinalis, respectively, demonstrating a marked contraction from wild species to domesticated asparagus [28]. Orthologous analysis identified only 16 conserved NLR gene pairs between A. setaceus and A. officinalis, representing the NLR repertoire preserved during domestication [28]. Pathogen inoculation assays demonstrated that domesticated A. officinalis was susceptible to Phomopsis asparagi, while A. setaceus remained asymptomatic, with retained NLR genes in the domesticated species showing unchanged or downregulated expression after fungal challenge [28].
Convergent NLR Reduction in Aquatic and Carnivorous Plants: Research utilizing the Angiosperm NLR Atlas (ANNA) revealed that NLR contraction is associated with adaptations to aquatic, parasitic, and carnivorous lifestyles [24]. This convergent NLR reduction in aquatic plants resembles the lack of NLR expansion during the long-term evolution of green algae before land colonization, suggesting that specific ecological niches may reduce reliance on diverse NLR repertoires [24].
NLR Expansion in Wild Relatives and Specific Lineages
Glycine Genus (Soybean): Divergent evolution of NLR genes between annual and perennial Glycine species reveals a remarkable expansion in annuals (G. max and G. soja) compared to perennial relatives [9]. Evolutionary timescale analysis pinpoints recent accelerated gene duplication events for this expansion between 0.1 and 0.5 million years ago, driven predominantly by lineage-specific and terminal duplications [9]. In contrast, perennial species experienced significant contraction during diploidization following the Glycine-specific whole-genome duplication event (~10 million years ago) [9]. Despite overall reduction, perennial lineages developed a unique and highly diversified NLR repertoire with limited interspecies synteny, resulting from birth of novel genes following individual speciation events [9].
Arachis Genus (Peanut): Wild and domesticated tetraploid peanut species show asymmetric expansion of NLRome in both subgenomes [14]. In wild tetraploid A. monticola, the A-subgenome exhibited significant contraction while the B-subgenome expanded, whereas the domesticated A. hypogaea showed the opposite pattern, likely due to distinct natural and artificial selection pressures [14]. Among diploid species, A. cardenasii revealed the largest NLR repertoire (521 genes) due to higher frequency of gene duplication and selection pressure [14].
Oleaceae Family: The genus Olea (olives) has undergone extensive NLR expansion driven by recent duplications and significant birth of novel NLR gene families [32]. In contrast, Fraxinus (ash trees) predominantly exhibits gene conservation, with Old World species showing dynamic gene expansion and contraction within the last 50 million years [32]. Genes acquired from an ancient whole genome duplication event (~35 Mya) have been retained across Fraxinus lineages [32].
Table 2: Evolutionary Patterns of NLR Genes Across Plant Taxa
| Plant Taxon | Representative Species | NLR Count Range | Dominant Evolutionary Pattern | Key Influencing Factors |
|---|---|---|---|---|
| Asparagus | A. officinalis (27), A. setaceus (63) | 27-63 | Contraction in domesticated species | Artificial selection, domestication |
| Apiaceae | A. sinensis (95), C. sativum (183) | 95-183 | Variable (contraction/expansion) | Lineage-specific adaptations |
| Glycine | Annuals vs. perennials | Highly variable | Expansion in annuals, contraction in perennials | Life history strategy, polyploidy |
| Arachis | A. cardenasii (521), A. stenosperma (354) | 284-794 | Asymmetric expansion in tetraploids | Domestication, subgenome dominance |
| Arecaceae | D. jenkinsiana (536), P. dactylifera (85) | 85-536 | "Consistent expansion" or "expansion then contraction" | Species-specific dynamics |
| Oleaceae | Olea (expansion), Fraxinus (conservation) | Variable | Genus-specific patterns | Geographical adaptation, WGD history |
Mechanistic Drivers of NLR Evolution
Whole Genome Duplication (WGD): Polyploidization events provide raw genetic material for NLR diversification. In Glycine species, a genus-specific WGD (~10 Mya) initially expanded NLR content, followed by differential retention in annuals versus perennials [9]. Similarly, ancient WGD in Fraxinus (~35 Mya) contributed NLR genes retained across lineages [32].
Tandem Duplications: Localized gene duplications represent a primary mechanism for rapid NLR expansion. In Trifolium species, the overall expansion of NLR repertoire in T. subterraneum is attributed to gene duplication events and birth of gene families after speciation [27].
Domain Integration and Loss: Frequent domain loss and alien domain integration shape NLR protein structures across lineages. Studies in Arecaceae species identified high variability in NLR domain architecture, contributing to functional diversification [31].
Birth-and-Death Evolution: NLR genes evolve through a birth-and-death process where new genes are created by duplication, and some duplicates are maintained while others are deleted or pseudogenized. This process is particularly evident in perennial Glycine species, where novel NLR genes emerged after speciation events despite overall contraction [9].
Table 3: Essential Research Reagents and Resources for NLR Comparative Genomics
| Resource Category | Specific Resource | Application in NLR Research | Key Features |
|---|---|---|---|
| Genomic Databases | Gramene, Ensembl Plants, NCBI Genome | Access to annotated plant genomes | Comparative genomics, orthology analysis |
| Specialized Databases | ANNA, PRGdb, PlantCARE | NLR-specific data, cis-element prediction | Curated NLR information, promoter analysis |
| Bioinformatics Tools | NLRtracker, OrthoFinder, MCScanX | Automated NLR identification, synteny analysis | High-throughput capability, user-friendly output |
| Domain Databases | Pfam, InterPro, CDD | Domain architecture analysis | Comprehensive domain models, validation |
| Phylogenetic Software | IQ-TREE, MEGA, Notung | Evolutionary relationship reconstruction | Model selection, duplication/loss inference |
| Visualization Tools | TBtools, Circos, iTOL | Data visualization and presentation | Customizable graphics, publication-ready figures |
The comparative genomic analysis of NLR genes across plant species reveals astonishing dynamism in their evolutionary patterns, driven by diverse selective pressures including pathogen coevolution, domestication, life history strategies, and ecological adaptations. The methodological framework presented here provides researchers with standardized protocols for identifying, classifying, and analyzing NLR genes across species, enabling consistent comparisons and deeper insights into plant immunity evolution.
Future research directions should include more comprehensive sampling across plant lineages, integration of pan-genome approaches to capture intra-species NLR diversity, and functional validation of evolutionary patterns through molecular studies. The increasing availability of high-quality genome assemblies and advanced bioinformatics tools will continue to enhance our understanding of how NLR gene contraction and expansion shapes plant-pathogen interactions and contributes to immune system evolution in land plants.
Understanding these dynamic evolutionary processes has profound implications for crop improvement, as wild relatives with expanded or diversified NLR repertoires represent valuable resources for introducing broad-spectrum disease resistance into cultivated varieties. The asymmetric evolution of NLR genes in polyploids and the impact of domestication on NLR contraction highlight both challenges and opportunities for sustainable crop protection through harnessing natural NLR diversity.
The Nucleotide-binding domain and Leucine-rich Repeat (NLR) gene family constitutes the primary intracellular immune receptor repertoire in land plants, responsible for detecting pathogen effector proteins and initiating robust defense responses through effector-triggered immunity (ETI) [33] [5]. Phylogenetic reconstruction of NLR genes has become an indispensable tool for unraveling the complex evolutionary relationships within this rapidly diversifying gene family. These analyses trace their origins to unicellular green algae [33], through major evolutionary transitions including the colonization of land, adaptations to diverse pathogen pressures, and whole-genome duplication events [34] [35] [32]. The dynamic evolutionary history of NLR genes—characterized by frequent gene duplications, domain rearrangements, functional diversification, and occasional gene losses—presents both challenges and opportunities for phylogenetic analysis. Within the context of land plant evolution, NLR phylogenetics provides crucial insights into how immune systems adapt to changing pathogenic threats over geological timescales, revealing patterns of lineage-specific expansion and contraction that correlate with ecological adaptations and life history strategies [34] [5] [35].
Core Principles: NLR Gene Architecture and Classification
NLR proteins exhibit a characteristic modular structure consisting of three core domains: a variable N-terminal domain, a central nucleotide-binding (NB-ARC) domain, and C-terminal leucine-rich repeats (LRRs) [33] [36]. Phylogenetic classification primarily recognizes three major NLR subfamilies based on N-terminal domain architecture:
- TNLs: Contain a Toll/Interleukin-1 Receptor (TIR) domain
- CNLs: Feature a Coiled-Coil (CC) domain
- RNLs: Possess a Resistance to Powdery Mildew 8 (RPW8) domain [5] [35] [36]
The RNL subfamily further divides into two functionally distinct clades: NRG1 (N-required gene 1) and ADR1 (activated disease resistance gene 1), which often function as "helper" NLRs in downstream signaling cascades [36]. This classification framework provides the foundation for phylogenetic reconstruction and comparative genomic analyses across land plants.
Table 1: Major NLR Subfamilies and Their Characteristics
| Subfamily | N-terminal Domain | Representative Motifs | Distribution in Land Plants |
|---|---|---|---|
| TNL | TIR (Toll/Interleukin-1 Receptor) | TIR-1 to TIR-5 | Absent in most monocots; multiple independent losses in magnoliids and Lamiales [35] |
| CNL | CC (Coiled-Coil) | RNBS-A, RNBS-D, MHD | Ubiquitous across all land plants; dramatic expansions in magnoliids [35] |
| RNL | RPW8 (Resistance to Powdery Mildew 8) | RNBS-D (CFLDLGxFP), MHD (QHD) | Highly diversified in conifers; two major clades (NRG1, ADR1) [36] |
Methodological Framework: Phylogenetic Reconstruction of NLR Genes
Identification and Annotation of NLR Genes
The initial and critical step in NLR phylogenetic analysis involves comprehensive identification and annotation of NLR genes from genomic or transcriptomic sequences. The NLRtracker pipeline has emerged as a standardized tool for this purpose, employing InterProScan and predefined NLR motif patterns to identify and characterize NLR genes in a high-throughput manner [34] [32] [14]. The workflow typically involves:
Sequence Acquisition: Obtain genomic sequences, annotated protein-coding sequences, and gene transfer format (GTF) files from relevant databases (NCBI, Phytozome, organism-specific databases) [33] [34] [14].
Domain Identification: Perform Hidden Markov Model (HMM) searches using Pfam domain profiles (NB-ARC: PF00931, TIR: PF01582, RPW8: PF05659) with HMMER software (E-value cutoff typically 10⁻⁴) [33] [5]. Complement with BLASTp searches (E-value cutoff 10) to identify divergent homologs [33].
Architecture Annotation: Validate domain organization using InterProScan and conserved domain search tools [33] [34]. Extract NB-ARC domains for phylogenetic analysis due to their high conservation and phylogenetic signal [14].
Motif Validation: Identify conserved motifs within the NB-ARC domain (P-loop, kinase 2, RNBS-A, RNBS-D, GLPL, MHD) using MEME suite or similar tools [5] [36]. These motifs provide additional validation of NLR identity and help distinguish subfamilies.
Sequence Alignment and Phylogenetic Analysis
Reconstruction of evolutionary relationships relies on accurate sequence alignment and appropriate phylogenetic inference methods:
Multiple Sequence Alignment: Extract NB-ARC domain sequences and align using MAFFT (L-INS-i algorithm) or ClustalW with default parameters [33] [5]. For large datasets, consider using tools like MUSCLE [14].
Model Selection: Identify best-fit substitution models using ModelFinder [5] or similar tools integrated in phylogenetic software. Complex mixture models (e.g., VT+F+R9) often perform well for NLR gene families [14].
Tree Inference: Implement maximum likelihood analysis using IQ-TREE with 1000 bootstrap replicates (SH-aLRT and UFBoot2) to assess branch support [5] [14]. Alternative approaches may include Bayesian inference using MrBayes for smaller datasets.
Tree Reconciliation: Compare gene trees with species trees using Notung software to infer duplication and loss events [5]. This step is particularly important for understanding the birth-death dynamics of NLR gene families.
Evolutionary Rate Analysis and Selection Pressure Assessment
Understanding selective constraints acting on NLR genes provides insights into their functional evolution:
Ortholog Identification: Identify orthologous NLR gene pairs across species using OrthoFinder [14] or similar tools.
Sequence Alignment: Perform codon-aware alignments of coding sequences using pal2nal [14].
Evolutionary Rate Calculation: Calculate non-synonymous (Ka) and synonymous (Ks) substitution rates using the MA method in Ka/Ks calculators [14]. Filter results using Fisher's test (P-value < 0.01) and exclude Ks values >2 to avoid saturation effects [14].
Table 2: Key Bioinformatics Tools for NLR Phylogenetic Analysis
| Tool Category | Software/Pipeline | Primary Function | Key Parameters |
|---|---|---|---|
| Gene Identification | NLRtracker [34] [32] [14] | Genome-wide NLR identification | InterProScan domains, predefined NLR motifs |
| Domain Analysis | HMMER [33] [5] | Domain identification | E-value = 10⁻⁴, Pfam profiles |
| Multiple Alignment | MAFFT/ClustalW/MUSCLE [33] [5] [14] | Sequence alignment | L-INS-i algorithm (MAFFT) |
| Phylogenetic Inference | IQ-TREE [5] [14] | Maximum likelihood tree building | ModelFinder, 1000 bootstraps |
| Evolutionary Rates | Ka/Ks calculator [14] | Selection pressure analysis | MA method, P-value < 0.01 |
| Gene Family Evolution | CAFE5 [14] | Gene gain/loss analysis | Stochastic birth-death model |
Case Studies in Land Plants
NLR Evolution in Angiosperms: Contrasting Patterns in Annual and Perennial Lineages
Comparative analysis of NLR genes in the genus Glycine reveals how life history strategy influences immune gene evolution. Annual species (G. max, G. soja) exhibit expanded NLRomes compared to perennial relatives, driven by recent duplication events between 0.1-0.5 million years ago [34]. In contrast, perennial lineages experienced significant NLR contraction following the Glycine-specific whole-genome duplication (~10 million years ago) but maintained a highly diversified NLR repertoire with limited interspecies synteny [34]. This suggests distinct evolutionary strategies: annuals employ quantitative expansion through recent duplications, while perennials rely on functional diversification of a core NLR set.
In magnoliids, phylogenetic analyses of seven species reveal dramatic expansions of CNLs and multiple independent losses of TNLs [35]. Reconstruction of ancestral NLR genes identified 74 ancestral R genes (70 CNLs, 3 TNLs, and 1 RNL) in the common magnoliid ancestor [35]. Tandem duplication served as the major driver of NLR expansion, with most species showing evolutionary patterns of "expansion followed by contraction" [35].
Evolutionary Patterns in Response to Polyploidization
Allopolyploidization events present natural experiments for studying NLR evolution. In tetraploid peanuts (Arachis hypogaea and A. monticola), asymmetric expansion of NLRomes occurred between A and B subgenomes [14]. Wild tetraploid A. monticola exhibited contraction in the A-subgenome and expansion in the B-subgenome, while the domesticated A. hypogaea showed the opposite pattern, suggesting distinct evolutionary pressures under natural and artificial selection [14]. Similarly, analysis of the tetraploid G. dolichocarpa revealed unbalanced NLR expansion favoring the Dt subgenome over the At subgenome [34].
NLR Phylogenetics in Gymnosperms: Conservation and Diversification
Conifers possess remarkably diverse and numerous RNL genes compared to angiosperms, with four distinct RNL groups, two of which are conifer-specific [36]. Phylogenetic analysis of 3,816 expressed NLR sequences from seven conifer species identified unique RNL signatures in the RNBS-D (CFLDLGxFP) and MHD (QHD) motifs [36]. This RNL diversification may represent an important adaptation in long-lived conifers, with specific RNL groups showing responsiveness to drought stress [36].
Table 3: Essential Research Reagents and Resources for NLR Phylogenetics
| Category | Resource | Specification/Function | Application Examples |
|---|---|---|---|
| Genomic Resources | Reference Genomes (NCBI, Phytozome, organism-specific databases) | Chromosome-scale assemblies preferred | Comparative genomics, synteny analysis [34] [32] [14] |
| Software Tools | NLRtracker Pipeline | Integrates InterProScan and predefined NLR motifs | High-throughput NLR identification and annotation [34] [32] [14] |
| Domain Databases | Pfam Database | Curated HMM profiles (NB-ARC: PF00931) | Domain identification and verification [33] [5] [36] |
| Sequence Alignment | MAFFT v6.814b | L-INS-i algorithm for accurate alignment | Multiple sequence alignment of NB-ARC domains [33] |
| Phylogenetic Inference | IQ-TREE v2.0 | ModelFinder, ultrafast bootstrap approximation | Maximum likelihood tree building [5] [14] |
| Evolutionary Analysis | CAFE5 | Stochastic birth-death model for gene families | Gene gain/loss analysis across phylogeny [14] |
Interpretation and Synthesis: Integrating Phylogenetic Data with Biological Insights
Effective interpretation of NLR phylogenetic analyses requires integration of multiple lines of evidence:
Contextualizing Gene Trees: Map gene duplication events onto species phylogenies to distinguish lineage-specific expansions from ancestral NLR diversity [33] [35]. For example, the identification of the first NB-LRR arrangement in Chlorophyta indicates the ancient origin of NLR genes in green algae, possibly through horizontal gene transfer [33].
Correlating Evolutionary Patterns with Phenotypes: Associate NLR subfamily expansions with documented pathogen resistance. For instance, the expansion of specific CNL clades in magnoliids may reflect adaptations to particular pathogen pressures [35].
Assessing Functional Evolution: Integrate expression data (e.g., RNA-seq from multiple tissues or stress conditions) with phylogenetic positions to identify conserved regulatory patterns or neofunctionalization events [35] [32]. Studies in Saururus chinensis reveal low expression of most NLR genes except in roots and fruits, suggesting tissue-specific functions [35].
Evaluating Selection Pressures: Calculate Ka/Ks ratios to identify NLR clades under positive selection, potentially indicating arms-race coevolution with pathogens [14].
The phylogenetic reconstruction of NLR genes across land plants reveals a dynamic evolutionary history shaped by repeated cycles of expansion and contraction, with lineage-specific adaptations reflecting distinct life history strategies and pathogen pressures. These analyses not only illuminate the deep evolutionary history of plant immunity but also provide practical insights for identifying functional resistance genes for crop improvement.
Nucleotide-binding leucine-rich repeat receptors (NLRs) represent a major class of intracellular immune receptors that function as critical components of the plant immune system, conferring protection against diverse pathogens through effector-triggered immunity (ETI). Recent advances in transcriptomic profiling have revealed that NLR-mediated signaling extends beyond traditional biotic stress responses to include significant roles in abiotic stress adaptation, highlighting their dual functionality in plant stress perception. The evolution of NLR genes in land plants reflects a complex history of functional diversification, with gene family expansion and contraction dynamics shaped by continuous adaptive pressures from both pathogens and environmental challenges. Transcriptomic approaches have been instrumental in uncovering the sophisticated regulatory networks controlling NLR expression, demonstrating that these genes exhibit precise temporal and spatial expression patterns in response to stress stimuli. This technical review integrates current understanding of NLR gene expression dynamics under biotic and abiotic stress conditions, providing a comprehensive framework for researchers investigating the evolutionary plasticity of plant immune systems.
NLR Gene Family Architecture and Evolutionary Dynamics
The NLR gene family exhibits remarkable structural diversity and evolutionary dynamics across plant species, characterized by rapid expansion and contraction events driven primarily by tandem duplication and positive selection. Comparative genomic analyses reveal significant variation in NLR repertoire size and composition, reflecting species-specific adaptation to environmental pressures.
Table 1: Comparative Analysis of NLR Gene Family Size Across Plant Species
| Species | Total NLR Genes | CNL Subfamily | TNL Subfamily | RNL Subfamily | Reference |
|---|---|---|---|---|---|
| Arabidopsis thaliana | ~150 | 56 | 94 | 4 | [23] |
| Oryza sativa (rice) | ~500 | 378 | 7 | 15 | [37] |
| Capsicum annuum (pepper) | 288 | 199 | 67 | 22 | [38] |
| Asparagus officinalis (garden asparagus) | 27 | 15 | 9 | 3 | [28] |
| Asparagus setaceus (wild relative) | 63 | 32 | 25 | 6 | [28] |
| Vigna unguiculata (cowpea) | 2188 (R-genes) | Not specified | Not specified | Not specified | [39] |
The evolutionary trajectory of NLR genes is marked by significant genomic dynamics. Gene family contraction has been documented in domesticated species, as evidenced by the reduction from 63 NLR genes in wild Asparagus setaceus to just 27 in cultivated Asparagus officinalis, suggesting that artificial selection for agricultural traits may compromise immune repertoire diversity [28]. Conversely, tandem duplication serves as a primary mechanism for NLR family expansion, particularly in response to pathogen pressure, with 18.4% (53/288) of pepper NLR genes arising through this mechanism, predominantly clustered on chromosomes 08 and 09 [38]. Promoter cis-regulatory element analysis reveals that NLR genes are enriched in defense-related motifs, with 82.6% of pepper NLR promoters containing binding sites for salicylic acid (SA) and/or jasmonic acid (JA) signaling pathways, indicating conserved transcriptional regulation mechanisms across plant species [38].
Transcriptomic Signatures of NLR Expression in Biotic Stress Responses
Transcriptomic profiling has elucidated sophisticated NLR expression patterns during plant-pathogen interactions, revealing both constitutive and induced expression dynamics that correlate with resistance phenotypes. Advanced RNA sequencing technologies have enabled researchers to capture these expression signatures with unprecedented temporal resolution and sensitivity.
Expression Dynamics in Fungal Pathogen Interactions
Comprehensive time-course transcriptomic analyses of soybean challenged with Fusarium oxysporum revealed 1,496 differentially expressed genes following pathogen challenge, with significant enrichment in MAPK signaling and plant-pathogen interaction pathways [40]. Among these, 13 key NLR genes demonstrated coordinated expression patterns, with the most dramatic transcriptional activation observed in resistant genotypes. Similarly, in asparagus, transcriptomic profiling following Phomopsis asparagi infection revealed that the majority of preserved NLR genes in susceptible cultivated A. officinalis exhibited either unchanged or downregulated expression, indicating potential functional impairment in disease resistance mechanisms during domestication [28].
Functional NLR Expression Signatures
A groundbreaking multi-species analysis demonstrated that functional NLRs consistently exhibit high steady-state expression levels in uninfected plants across both monocot and dicot species [41]. This expression signature challenges the traditional paradigm that NLRs require strict transcriptional repression to avoid autoimmunity. In Arabidopsis, known functional NLRs are significantly enriched in the top 15% of expressed NLR transcripts, with the most highly expressed NLR (ZAR1) exceeding median and mean expression levels for all genes in the Col-0 ecotype [41]. This pattern holds across diverse species, with barley Rps7/Mla7 and Rps7/Mla8, Aegilops tauschii-derived Sr46, SrTA1662, and Sr45, and tomato Mi-1 all appearing among highly expressed NLR transcripts in their respective species [41].
Pathway Diagram 1: NLR-Mediated Biotic Stress Signaling Network - This diagram illustrates the integrated signaling network activated following NLR recognition of biotic stress, highlighting key pathways identified through transcriptomic analyses.
NLR Roles in Abiotic Stress Tolerance: Emerging Transcriptomic Evidence
Recent transcriptomic and functional studies have revealed surprising connections between NLR genes and abiotic stress responses, particularly chilling tolerance, expanding their traditional roles beyond pathogen recognition. These findings suggest that certain NLR proteins have been co-opted during evolution to function in environmental stress adaptation.
Chilling Tolerance Mechanisms
In japonica rice, the NLR gene RGA4L has been identified as a major determinant of chilling tolerance throughout all growth stages, with overexpression enhancing tolerance at both vegetative and reproductive stages [37]. Transcriptomic and protein interaction analyses revealed that RGA4L physically interacts with both OsHSP90 and OsLEA5, facilitating proper assembly of a protein complex that senses and transduces chilling signals to downstream pathways [37]. Population genetic analysis demonstrates that RGA4L has been a major target of artificial selection during japonica rice domestication for low-temperature acclimation, explaining the subspecies' adaptation to high-altitude and temperate regions [37].
Regulatory Networks in Abiotic Stress
The involvement of NLRs in abiotic stress extends beyond direct protein interactions to include transcriptional reprogramming. Transcriptomic analyses of cold-stressed rice plants revealed that RGA4L modulates the expression of late embryogenesis abundant (LEA) proteins and heat shock proteins, connecting NLR function with established abiotic stress tolerance mechanisms [37]. This suggests that certain NLR proteins may have evolved to integrate biotic and abiotic stress signaling networks, potentially through shared components like HSP90 chaperones.
Table 2: Documented NLR Genes with Dual Roles in Biotic and Abiotic Stress
| NLR Gene | Species | Biotic Stress Function | Abiotic Stress Function | Mechanistic Insights |
|---|---|---|---|---|
| RGA4L | Oryza sativa | Not specified | Chilling tolerance throughout all growth stages | Interacts with OsHSP90 and OsLEA5 to sense and transduce chilling signals [37] |
| ACQOS/VICTR | Arabidopsis thaliana | Disease resistance | Osmotic stress tolerance | Involved in trade-off between abiotic and biotic stress adaptation [37] |
| CHS2 | Arabidopsis thaliana | Disease resistance | Chilling sensitivity | Activation mediated by SGT1b-RAR1-HSP90 complex [37] |
| ADR1 | Arabidopsis thaliana | Disease resistance | Drought tolerance | Positive regulator of drought resistance [37] |
Experimental Frameworks for NLR Transcriptomic Analysis
Comprehensive transcriptomic profiling of NLR genes requires carefully designed experimental approaches that capture both temporal dynamics and tissue-specific expression patterns. The following methodologies represent state-of-the-art protocols for investigating NLR expression in stress responses.
RNA Sequencing Workflow for NLR Expression Analysis
Workflow Diagram 2: Transcriptomic Analysis of NLR Genes - This experimental workflow outlines key steps for RNA sequencing-based analysis of NLR gene expression in stress responses, incorporating best practices from recent studies.
Table 3: Key Research Reagent Solutions for NLR Transcriptomic Studies
| Reagent/Resource | Specification | Application | Example Implementation | ||
|---|---|---|---|---|---|
| RNA Extraction Kit | Qiagen RNeasy Plant Mini Kit or equivalent | High-quality RNA isolation from plant tissues | Duplicate extraction from young leaves [39] | ||
| Quality Control Instruments | Nanodrop 2000 (A260/A280: 1.8-2.0), Qubit, agarose gel electrophoresis | RNA quantity/quality assessment | Samples with A260/A230 > 1.8, no degradation used for sequencing [39] | ||
| Library Preparation Kit | NEXTFLEX Rapid DNA-seq kit for Illumina | Sequencing library construction | 500ng DNA fragmented to 200-250bp [39] | ||
| Sequencing Platforms | Illumina HiSeq X Ten (150bp paired-end), Nanopore GridION X5 | High-throughput transcriptome sequencing | Hybrid assembly combining both platforms [39] | ||
| NLR Identification Tools | HMMER (PF00931), BLASTp against reference NLRs | Genome-wide NLR annotation | E-value cutoff 1e-10, domain validation [28] [38] | ||
| Differential Expression Analysis | DESeq2, HISAT2, FPKM quantification | Identification of stress-responsive NLRs | log2FC | ≥ 1, FDR < 0.05 [40] [38] | |
| Transgenic Validation | High-throughput transformation systems | Functional characterization of NLR candidates | Wheat transgenic array of 995 NLRs [41] |
Evolutionary Implications and Future Research Directions
The integration of transcriptomic data with evolutionary analysis reveals that NLR genes represent dynamic components of plant genomes, with expression patterns that have been shaped by competing pressures from both biotic and abiotic environments. Domestication-associated NLR contraction observed in species like asparagus, where cultivated varieties retain only 43% of the NLR genes found in wild relatives, demonstrates how artificial selection can reshape immune gene repertoires, potentially compromising stress resilience [28]. Conversely, the conservation of high expression for functional NLRs across diverse plant species suggests positive selection for maintained expression of certain NLR loci, challenging the historical view that NLRs require strict transcriptional repression [41].
Future research directions should prioritize multi-omics integration, combining transcriptomic data with genomic, epigenomic, and proteomic analyses to fully elucidate NLR regulatory networks. The development of pangenome-scale transcriptomic resources will be essential for capturing the full extent of NLR expression diversity across species and populations [23]. Additionally, tissue-specific and single-cell transcriptomic approaches will provide unprecedented resolution for understanding NLR expression dynamics in spatially restricted defense responses. These advanced methodologies will further illuminate the evolutionary mechanisms through which NLR genes have been co-opted for diverse stress adaptation functions in land plants, with significant implications for crop improvement strategies facing climate change and emerging pathogen threats.
In the study of plant innate immunity, the Nucleotide-binding Leucine-rich Repeat (NLR) gene family represents one of the most dynamic and rapidly evolving components of the plant immune system. These intracellular receptors recognize pathogen-derived effector molecules and initiate robust defense responses [3]. The extraordinary diversity of NLR genes, driven by constant evolutionary arms races with pathogens, presents a significant challenge for comparative genomics. Orthogroup analysis has emerged as an essential computational framework for deciphering these complex evolutionary relationships across multiple species.
This methodology allows researchers to cluster NLR genes into orthogroups—sets of genes descended from a single gene in the last common ancestor of the species being compared. Through this process, scientists can distinguish between core NLR clusters (conserved across species) and species-specific clusters (lineage-specific expansions), providing crucial insights into evolutionary conservation, functional specialization, and adaptive innovation in plant immune systems [28] [42]. When framed within the broader context of land plant evolution, orthogroup analysis reveals how different plant lineages have deployed distinct evolutionary strategies to maintain effective immune recognition systems against diverse pathogen threats.
Technical Foundations: Orthogroup Methodology and Workflow
Conceptual Framework and Key Definitions
Orthogroup analysis provides a phylogenetic framework for understanding gene family evolution across multiple species. The foundational concepts include:
- Orthogroup: A set of genes that all descended from a single gene in the last common ancestor of all species considered, including both orthologs and paralogs [28] [42].
- Orthologs: Genes in different species that evolved from a common ancestral gene by speciation, typically retaining the same function over evolutionary time.
- Paralogs: Genes related by duplication within a genome, which may evolve new functions or specializations.
- Core NLR Clusters: Orthogroups containing NLR genes conserved across multiple related species, representing essential immune components maintained through evolutionary time.
- Species-Specific NLR Clusters: Orthogroups restricted to a single species or lineage, often resulting from recent duplications and potentially conferring specialized resistance capabilities.
Experimental Workflow for NLR Orthogroup Analysis
The following workflow diagram illustrates the comprehensive pipeline for conducting orthogroup analysis of NLR genes:
Diagram: NLR Orthogroup Analysis Workflow. This pipeline outlines the key steps from initial data preparation through biological interpretation.
Table: Core Bioinformatics Tools for NLR Orthogroup Analysis
| Tool/Resource | Primary Function | Key Parameters | Application Context |
|---|---|---|---|
| OrthoFinder [28] [42] | Orthogroup inference and comparative genomics | -d (species tree inference), -M msa (multiple sequence alignment) | Core analysis pipeline for clustering NLR genes across species |
| NLRtracker [32] | Domain-based NLR identification | Default parameters with plant database | High-throughput mining of NLR genes from genomic data |
| InterProScan [28] [42] | Protein domain annotation | -appl Pfam, -iprlookup | Validation of NB-ARC, TIR, CC, and LRR domains |
| TBtools [28] [42] | Comparative genomics visualization | One-Step MCScanX, Gene Location Visualize | Synteny analysis and chromosomal mapping of NLR clusters |
| MEME Suite [28] [42] | Conserved motif discovery | -nmotifs 10, -mod anr | Identification of conserved NLR structural motifs |
Case Studies in Orthogroup Analysis of NLR Genes
NLR Conservation and Contraction in Asparagus Species
A compelling application of orthogroup analysis comes from comparative genomic studies of garden asparagus (Asparagus officinalis) and its wild relatives (A. setaceus and A. kiusianus). This research revealed a striking pattern of NLR gene contraction during the domestication process, with orthogroup analysis providing quantitative evidence of this evolutionary trajectory [28] [42].
Table: NLR Gene Distribution in Asparagus Species
| Species | Total NLR Genes | Core Orthogroups with A. setaceus | Species-Specific Expansions | Domestication Status |
|---|---|---|---|---|
| A. setaceus (wild) | 63 | 16 | 47 | Wild relative |
| A. kiusianus (wild) | 47 | Not analyzed | Not analyzed | Wild relative |
| A. officinalis (cultivated) | 27 | 16 | 11 | Domesticated |
The orthogroup analysis identified 16 conserved NLR gene pairs between A. setaceus and A. officinalis, representing the core NLR repertoire preserved during domestication [28] [42]. Functional characterization revealed that most of these conserved NLRs showed unchanged or downregulated expression following pathogen challenge in the cultivated species, suggesting potential functional impairment during domestication. This finding illustrates how orthogroup analysis can pinpoint specific genetic changes underlying agronomically important traits like disease susceptibility.
Contrasting Evolutionary Strategies in Oleaceae Family
Orthogroup analysis of NLR genes across the Oleaceae family reveals how different genera have employed distinct evolutionary strategies to adapt to their specific pathogen environments. A comprehensive study of 30 species across genera including Fraxinus (ash trees), Olea (olives), Jasminum (jasmine), Forsythia, and Syringa (lilac) demonstrated remarkable variation in NLR evolution [32].
In the genus Fraxinus, orthogroup analysis revealed a pattern dominated by gene conservation, with maintenance of NLR genes originating from an ancient whole genome duplication event approximately 35 million years ago. This conservation strategy maintains specialized immune responses potentially optimized for co-evolved pathogens. In contrast, the genus Olea exhibited extensive gene expansion driven by recent duplications and birth of novel NLR gene families, likely enhancing its ability to recognize diverse pathogens through rapid innovation [32].
These contrasting evolutionary strategies—conservation in Fraxinus versus expansion in Olea—demonstrate how orthogroup analysis can reveal fundamental differences in immunological adaptation across plant lineages. The analysis further revealed consistent patterns across Oleaceae, including enhanced pseudogenization of TIR-NLRs and expansion of CCG10-NLRs, suggesting family-wide evolutionary trends [32].
Research Reagent Solutions for NLR Orthogroup Studies
Table: Essential Research Reagents and Resources
| Category | Specific Resource | Specifications/Function | Application Example |
|---|---|---|---|
| Genomic Resources | A. officinalis genome assembly | BUSCO completeness: 97.5% (assembly), 98.1% (annotation) | Asparagus NLR contraction analysis [28] [42] |
| Genomic Resources | Fraxinus pennsylvanica genome | 757 Mb assembly, 35,470 gene models | Oleaceae comparative genomics [32] |
| Software Pipelines | NLRtracker | Automated NLR identification and classification | High-throughput NLR mining in Oleaceae [32] |
| Validation Tools | PlantCARE database | cis-element prediction in promoter regions | Identification of defense-responsive elements [28] [42] |
| Expression Data | RNA-seq datasets (Olea europaea) | SRA BioProject PRJNA638671 | Expression validation of NLR orthogroups [32] |
Evolutionary Interpretation of Orthogroup Data
Integration with Broader Evolutionary Patterns in Land Plants
Orthogroup analysis of NLR genes must be interpreted within the broader context of land plant evolution. Several key evolutionary patterns emerge from comparative studies:
- Differential Expansion Patterns: NLR gene families have undergone remarkable differential expansion across land plants. Early land plants like the moss Physcomitrella patens contain only approximately 25 NLRs, while angiosperms typically possess substantially expanded repertoires, with some species like wheat (Triticum aestivum) containing over 2,000 NLR genes [28] [3].
- Lineage-Specific Adaptations: Certain plant lineages have developed unique evolutionary strategies for NLR diversification. Cereal crops like rice, brachypodium, and sorghum have completely lost TNL genes while expanding CNL repertoires, representing a major lineage-specific adaptation [3].
- Pangenome Context: Recent pangenome studies in Arabidopsis thaliana have revealed that NLR neighborhoods vary tremendously among accessions, forming a "pangenomic NLR repertoire" that greatly expands the potential for pathogen recognition beyond what is present in any single individual [23].
Evolutionary Forces Shaping NLR Orthogroup Dynamics
The following diagram illustrates the key evolutionary processes that shape NLR orthogroup patterns across plant species:
Diagram: Evolutionary Forces Shaping NLR Orthogroups. Multiple mechanisms and selection pressures interact to generate observed patterns.
The diagram illustrates how multiple evolutionary mechanisms interact to shape NLR orthogroup patterns. Gene duplication, unequal crossing-over, and recombination facilitate the diversification of NLR genes, allowing plants to rapidly generate new recognition specificities [32]. These diversification mechanisms are driven by selection pressures including pathogen coevolution, host life history traits, and human selection during domestication. The balance between these forces determines whether NLR evolution in a particular lineage will favor conservation of core functions or expansion of species-specific recognition capabilities.
Orthogroup analysis has established itself as an indispensable methodology for deciphering the complex evolutionary dynamics of NLR genes in land plants. Through the identification of core and species-specific NLR clusters, this approach provides critical insights into how plant immune systems balance conservation of essential recognition functions with innovation in response to evolving pathogen threats. The case studies in asparagus and Oleaceae demonstrate how orthogroup analysis can reveal both conserved immune components and lineage-specific adaptations, linking evolutionary patterns to functional outcomes in plant-pathogen interactions.
Future developments in this field will likely include more sophisticated integration of pangenome references, enabling researchers to move beyond single-reference frameworks to capture the full extent of NLR diversity within species [23]. Additionally, the coupling of orthogroup analysis with high-throughput functional screening approaches—such as the transgenic array screening that identified 31 new functional NLRs against wheat rust pathogens [41]—will accelerate the translation of evolutionary insights into practical crop improvement. As genomic resources continue to expand across the plant kingdom, orthogroup analysis will remain a foundational approach for understanding the evolutionary ecology of plant immunity and harnessing this knowledge for sustainable agriculture.
Balancing Act: Navigating Autoimmunity, Fitness Costs, and Evolutionary Trade-offs in NLR Function
Hybrid necrosis is a post-zygotic reproductive barrier in plants where hybrids develop necrotic lesions and exhibit reduced fitness in the absence of pathogens [43] [44]. This phenomenon represents a classic "Dangerous Mix" scenario, where immune components from different parental lineages malfunction when combined in hybrids. The condition arises from deleterious epistatic interactions between genes that have diverged in isolated populations, and when reunited in hybrids, trigger autoimmunity [43] [45]. Most documented cases involve incompatible interactions between nucleotide-binding leucine-rich repeat (NLR) proteins or between NLRs and other host proteins [43] [44] [45]. As intracellular immune receptors, NLRs function similarly to NOD/CARD proteins in animals and play crucial roles in plant innate immunity [43]. The constant co-evolutionary arms race between plants and their pathogens drives diversification of immune components, including NLRs, making hybrid necrosis an inadvertent consequence of this evolutionary process [43]. This review synthesizes current understanding of hybrid necrosis within the broader context of NLR gene evolution in land plants, examining molecular mechanisms, experimental approaches, and evolutionary implications.
Molecular Mechanisms of NLR-Mediated Hybrid Necrosis
Genetic Incompatibility Models
The molecular basis of hybrid necrosis typically involves incompatible genetic interactions between divergent immune components. Several distinct models have been characterized across plant species, with most cases following an "incompatible gene pair" model where a sensor NLR from one parent interacts improperly with a helper NLR or other immune component from another parent [45]. In Arabidopsis thaliana, the DM10/DM11 interaction exemplifies this model, where a truncated singleton TIR-NLR (DM10) with a premature stop codon interacts with an unlinked locus (DM11) to trigger severe necrosis [43]. The DM10 risk allele has a truncated LRR–PL (leucine-rich repeat–post-LRR) region, indicating that substantial NLR truncations can lead to hybrid incompatibility [43].
In rice, the Pik NLR pair demonstrates allelic specialization, where matched pairs of Pik-1 (sensor) and Pik-2 (helper) NLRs mount effective immune responses, while mismatched pairs lead to autoimmune phenotypes [45]. This incompatibility is underpinned by a single amino acid polymorphism in Pik-2 that determines preferential association between matching pairs of Pik NLRs [45]. The functional specialization in these alleles reveals how co-adapted NLR pairs can become incompatible when mismatched in hybrids.
In Petunia, a unique case involves the interaction between a chitinase/lysozyme (ChiA1) on chromosome 2 and an unlinked locus on chromosome 7 [44]. Unlike typical NLR-NLR interactions, this case involves a bifunctional GH18 chitinase/lysozyme encoded by ChiA1, where the enzymatic activity is dispensable for necrosis development [44]. The ChiA1 protein is homologous to AtLYS1/ChiA in Arabidopsis, which has a central role in triggering immune responses [44].
Signaling Pathways and Immune Activation
The downstream signaling events in hybrid necrosis consistently involve activation of pathogen response pathways despite the absence of pathogens. Transcriptomic analyses of necrotic hybrids reveal massive transcriptional changes, with upregulation of most NLR genes and defense-related genes [43]. In Arabidopsis DM10/DM11 hybrids, approximately half of all detectable genes show differential expression, with defense response and salicylic acid biosynthesis being the most enriched categories [43].
Key signaling components consistently upregulated in hybrid necrosis include:
- Salicylic acid (SA) biosynthesis and signaling genes: EDS1, ICS1, EDS5, PAD4, PBS3, CBP60, and FMO1 [43]
- Pathogenesis-related (PR) genes: Multiple PR genes show induced expression [44]
- Endoplasmic reticulum (ER) stress markers: BiP4, BiP5, and bZIP60 in Petunia hybrids [44]
- Reactive oxygen species (ROS): Increased production observed in multiple systems [44]
The molecular architecture of these interactions reveals how hybrid necrosis emerges from conflicting immune components. The diagram below illustrates the core genetic and molecular pathways common to hybrid necrosis across systems:
Figure 1: Core signaling pathway in hybrid necrosis showing genetic incompatibility leading to immune activation and necrotic phenotype. SA: salicylic acid; ER: endoplasmic reticulum; NLR: nucleotide-binding leucine-rich repeat receptors.
Experimental Analysis of Hybrid Necrosis
Genetic Mapping and Validation Approaches
The identification and characterization of hybrid necrosis loci employs integrated genetic and genomic approaches. Bulked segregant RNA sequencing (BSR-seq) has proven effective for locating genomic regions associated with necrotic phenotypes, as demonstrated in Petunia, where strong signals were detected on chromosomes 2 and 7 (HNe2 and HNe7) [44]. This approach allows rapid mapping of causal loci by sequencing RNA from pooled individuals with similar phenotypes.
Fine-mapping strategies involve screening recombinant progenies to narrow candidate intervals. In Petunia, this approach reduced the HNe2 interval from 8.7 Mb to 1.74 Mb through whole-genome sequencing of recombinant lines [44]. The table below summarizes key experimental approaches used in hybrid necrosis research:
Table 1: Experimental Methods for Hybrid Necrosis Analysis
| Method | Application | Key Outcomes | References |
|---|---|---|---|
| BSR-seq | Genetic mapping of necrosis loci | Identified HNe2 and HNe7 in Petunia | [44] |
| RNA sequencing | Transcriptome profiling | Revealed upregulation of NLRs and defense genes in Arabidopsis | [43] |
| Virus-induced gene silencing (VIGS) | Functional validation | Confirmed ChiA1 as causal gene in Petunia HN | [44] |
| Quantitative trait locus (QTL) analysis | Mapping incompatibility loci | Identified DM10 and DM11 in Arabidopsis | [43] |
| Transient overexpression | Validation of gene function | Demonstrated ChiA1Ax triggers necrosis in Petunia | [44] |
| Allelic swap experiments | Testing functional specialization | Revealed Pik-1/Pik-2 specificity in rice | [45] |
Transcriptomic Profiling Protocols
Detailed transcriptomic analysis provides insights into the global gene expression changes underlying hybrid necrosis. The following protocol outlines the standard approach for RNA sequencing in hybrid necrosis studies:
Plant Material Collection: Sample leaf tissues from F1 hybrids and both parents at the developmental stage when early necrotic symptoms are visible but before severe tissue degradation [43]. For severe cases like Cdm-0×TueScha-9, sampling at 10 days after germination is appropriate [43].
RNA Extraction and Library Preparation: Extract total RNA using standardized kits (e.g., TRIzol method). Assess RNA quality using Bioanalyzer or similar systems. Prepare sequencing libraries using poly-A selection for mRNA enrichment or rRNA depletion protocols.
Sequencing and Data Analysis: Sequence libraries on an appropriate platform (Illumina recommended). Process raw reads through quality control (FastQC), alignment to reference genome (HISAT2, STAR), and quantification of gene expression (featureCounts, HTSeq).
Differential Expression Analysis: Identify differentially expressed genes using statistical packages (DESeq2, edgeR). Compare hybrids versus both parents and mid-parental values. Apply multiple testing correction (Benjamini-Hochberg FDR).
Functional Annotation: Conduct Gene Ontology (GO) enrichment analysis using specialized tools (TopGO, clusterProfiler). Focus on defense response, immune system process, and cell death categories.
The experimental workflow for comprehensive analysis of hybrid necrosis integrates multiple approaches from initial phenotype characterization to molecular validation:
Figure 2: Experimental workflow for hybrid necrosis research from phenotypic characterization to mechanistic understanding. BSR-seq: bulked segregant RNA sequencing; QTL: quantitative trait locus; WGS: whole-genome sequencing; VIGS: virus-induced gene silencing.
Comparative Analysis of Hybrid Necrosis Systems
Severity Spectrum and Phenotypic Manifestations
Hybrid necrosis displays a continuum of severity across different systems, from mild growth retardation to complete lethality. The table below compares representative cases of hybrid necrosis:
Table 2: Comparative Severity of Hybrid Necrosis Systems
| Plant System | Causal Genes | Severity Level | Key Phenotypic Features | Developmental Stage |
|---|---|---|---|---|
| Arabidopsis thaliana (Cdm-0×TueScha-9) | DM10 (TIR-NLR), DM11 | Severe | No development past cotyledon stage, death at 3 weeks | Early seedling [43] |
| Petunia (axillaris×exserta) | ChiA1 (chitinase), HNe7 | Moderate | Necrotic leaves, reduced growth, poor flower production | 38 days after sowing [44] |
| Rice (Pik mismatches) | Pik-1, Pik-2 | Mild-Moderate | Constitutive cell death, reduced growth | Vegetative stage [45] |
| Arabidopsis (Other DM cases) | Various NLR pairs | Variable | Range from mild chlorosis to severe necrosis | Various stages [43] |
The variation in severity reflects differences in the strength of autoimmune activation and the specific signaling pathways involved. Severe cases like DM10/DM11 in Arabidopsis involve massive transcriptional changes, with approximately one-third of the entire transcriptome being differentially expressed [43]. In contrast, milder cases may show more limited immune activation.
Evolutionary Genetics of Risk Alleles
The population distribution of risk alleles provides insights into the evolutionary dynamics of hybrid necrosis. In Arabidopsis, the DM10 risk allele (with premature stop codon) is geographically widespread but highly differentiated from non-risk alleles in the global population, suggesting recent expansion [43]. The DM11 risk allele is much rarer, found only in two accessions from southwestern Spain—a region where the DM10 risk haplotype is absent [43]. This non-overlapping distribution suggests that selection maintains the spatial separation of these incompatible alleles.
In rice, the functional specialization of Pik alleles demonstrates how paired NLRs co-evolve to maintain immune homeostasis while adapting to recognize rapidly evolving effectors [45]. The single amino acid polymorphism in Pik-2 that underpins both allelic specialization and immune homeostasis represents a key evolutionary checkpoint [45].
Research Toolkit for Hybrid Necrosis Studies
Table 3: Essential Research Reagents and Resources for Hybrid Necrosis Investigation
| Reagent/Resource | Function/Application | Example Use Cases |
|---|---|---|
| Arabidopsis accessions (Cdm-0, TueScha-9) | Parental lines for crosses | DM10/DM11 interaction studies [43] |
| Petunia introgression lines (IL5) | Genetic material for mapping | HNe2/HNe7 identification [44] |
| Rice Pik allelic variants | Paired NLR functional analysis | Sensor/helper specificity tests [45] |
| TRV-based VIGS vectors | Gene silencing in plants | Functional validation of ChiA1 [44] |
| RNA-seq libraries | Transcriptome profiling | Global expression analysis in hybrids [43] |
| Salicylic acid markers (EDS1, PAD4) | Defense signaling assessment | SA pathway activation confirmation [43] |
| ROS detection kits | Oxidative burst measurement | Detection of reactive oxygen species [44] |
| ER stress markers (BiP, bZIP60) | Endoplasmic reticulum stress monitoring | ER-stress-induced cell death [44] |
Discussion and Evolutionary Implications
Hybrid necrosis represents an evolutionary dilemma where the same genetic conflicts that drive immune receptor diversification also create genetic barriers to gene flow. The phenomenon illustrates how plant immune systems walk a tightrope between adaptive evolution to recognize rapidly evolving pathogens and maintaining functional homeostasis within the immune network [45]. The high diversification of NLRs, driven by co-evolutionary arms-races with pathogens, creates potential for incompatibility when divergent alleles meet in hybrids [43].
The tight genetic linkage of hybrid necrosis loci with other reproductive barriers strengthens isolation and potentially promotes speciation. In Petunia, the ChiA1 locus causing hybrid necrosis is tightly linked to major genes involved in pollination syndrome adaptation (MYB-FL, CNL1, EOBII), forming a supergene region on chromosome 2 [44]. This linkage between pre-zygotic (pollinator isolation) and post-zygotic (hybrid necrosis) barriers probably contributes to rapid diversification and speciation [44].
From a breeding perspective, understanding hybrid necrosis mechanisms has practical applications for crop improvement. Many crop species show similar autoimmune phenomena that limit the gene pool available for breeding [45]. Identifying risk alleles and incompatible combinations enables predictive approaches to avoid deleterious combinations in breeding programs. Furthermore, the structural insights from systems like the Pik pair in rice provide frameworks for engineering synthetic NLR combinations with desired specificities without triggering autoimmunity [45].
Future research directions should focus on structural characterization of incompatible protein complexes, population genomics of risk alleles across diverse species, and engineering approaches to mitigate hybrid necrosis while maintaining disease resistance. The integration of evolutionary genetics with molecular immunology continues to reveal how plants navigate the fundamental dilemma of maintaining a flexible, adaptive immune system without compromising internal harmony.
Nucleotide-binding leucine-rich repeat (NLR) genes constitute one of the largest and most dynamic gene families in plant genomes, encoding intracellular immune receptors essential for effector-triggered immunity. While crucial for pathogen recognition and defense activation, NLR expression and maintenance impose significant metabolic costs that create fundamental trade-offs between growth and defense in plants. This whitepaper synthesizes current research on the physiological and evolutionary mechanisms generating these costs, examining how plants mitigate fitness trade-offs through sophisticated regulatory networks, genomic architecture, and environmental sensing. We analyze quantitative data on NLR diversification across species, transcript dynamics under stress conditions, and resource allocation patterns that underlie growth-defense balance. Understanding these trade-offs provides crucial insights for developing crop varieties with enhanced disease resistance without compromising yield, addressing pressing challenges in agricultural sustainability and food security.
Plants have evolved a sophisticated innate immune system comprising both cell-surface and intracellular receptors to detect diverse pathogens. NLR (NOD-like receptor) proteins represent a major class of intracellular immune receptors that recognize pathogen effectors following the "gene-for-gene" model and activate robust defense responses [15]. These proteins exhibit a characteristic modular structure with a central nucleotide-binding domain (NB-ARC) that functions as a molecular switch, a C-terminal leucine-rich repeat (LRR) domain involved in effector recognition, and variable N-terminal domains (TIR, CC, or RPW8) that determine signaling specificity [15] [46]. The NB-ARC domain regulates NLR activation through ADP-ATP exchange, while the LRR domain provides structural stability in the inactive state and undergoes conformational changes upon pathogen perception [15].
The evolution of NLR genes reflects an ongoing arms race between plants and their pathogens, driving extraordinary sequence and structural diversification within this gene family [46]. This coevolutionary dynamic has produced one of the largest and most variable gene families in plant genomes, with NLR copy numbers ranging from approximately 50-100 in cucumber and watermelon to over 2000 in bread wheat [15]. This expansion is primarily driven by tandem duplication events, which account for approximately 18.4% of NLR genes in pepper genomes and facilitate rapid generation of novel resistance specificities [6]. NLR genes are frequently organized in complex clusters, particularly in subtelomeric regions with high recombination frequencies, creating genomic environments conducive to rapid evolution and functional diversification [15] [6].
Table 1: NLR Gene Family Size Across Plant Species
| Species | Genome Size | NLR Count | TIR-type | Non-TIR-type | Key Genomic Features |
|---|---|---|---|---|---|
| Arabidopsis thaliana | ~135 Mb | ~207 | Predominant | Minority | Balanced distribution |
| Capsicum annuum (pepper) | ~3.5 Gb | 288 | Not specified | Not specified | High density on Chr09 |
| Oryza sativa (rice) | ~389 Mb | ~500 | Absent | All | Telomeric clustering |
| Malus domestica (apple) | ~740 Mb | ~1000 | ~500 | ~500 | Recent duplication events |
| Triticum aestivum (wheat) | ~17 Gb | >2000 | Not specified | Not specified | Hexaploid genome |
Metabolic Costs of NLR Expression and Maintenance
Physiological Trade-offs Between Growth and Defense
The maintenance and activation of NLR-mediated immunity incur substantial metabolic costs that manifest as growth-defense trade-offs. These trade-offs arise from multiple physiological mechanisms, including direct resource competition, antagonistic hormone signaling, and allocation dilemmas. Both the constitutive maintenance of NLR proteins and their induced expression during defense responses divert energy and nutrients away from growth processes, creating fundamental fitness trade-offs that shape plant evolution and agriculture [47].
Approximately two-thirds of Arabidopsis NLR genes are induced by pathogens, immune elicitors, or salicylic acid, suggesting that transcriptional induction represents a significant metabolic investment during immune activation [48]. This investment extends beyond transcription to include protein synthesis, post-translational modifications, and signaling cascade activation. Research has demonstrated that resistant plant genotypes often exhibit reduced development of reproductive tissue under nutrient-poor conditions compared to susceptible genotypes, highlighting the resource-intensive nature of NLR-mediated defense [47].
Mechanisms Generating Growth-Defense Trade-offs
The metabolic costs of NLR immunity arise through several interconnected mechanisms. Nutrient limitation represents a primary source of trade-offs, as defense responses involving salicylic acid, auxin, glucosinolates, and methyl transferases all depend on sulfur availability and upregulate sulfur metabolism genes [47]. Similarly, access to nitrogen and phosphorus influences allocation to defenses, with nutrient-poor conditions exacerbating growth reductions in resistant genotypes [47].
Beyond direct resource allocation, antagonistic crosstalk among hormone signaling pathways creates physiological trade-offs. Gibberellins, which promote growth by destabilizing DELLA proteins, are suppressed during immune activation, leading to DELLA-mediated growth suppression [47]. Similarly, salicylic acid (SA)-mediated defense responses often inhibit growth-related processes, creating negative correlations between defense activation and biomass accumulation [47] [48]. This coregulation of growth and immunity reflects an evolutionary adaptation that allows plants to dynamically balance resource allocation based on environmental conditions.
Table 2: Metabolic Costs Associated with NLR-Mediated Immunity
| Cost Category | Specific Manifestations | Experimental Evidence |
|---|---|---|
| Maintenance Costs | Constitutive expression of NLR genes and proteins; ongoing immunological surveillance; infrastructure maintenance | Resistant genotypes show reduced reproductive tissue development under nutrient limitation [47] |
| Deployment Costs | Resource allocation during immune activation; synthesis of defense compounds; signaling cascade initiation | Physiological costs observed following immune challenge; fecundity reductions post-infection [49] |
| Autoimmunity Costs | Aberrant defense activation in absence of pathogens; spontaneous cell death; pleiotropic effects on development | Arabidopsis DM1 and DM2 NLR genes cause autoimmunity in specific hybrid combinations [15] |
| Ecological Costs | Reduced competitive ability; altered interactions with beneficial organisms; environmental sensitivity | Induced defenses cause greater growth reduction when plants face competition [47] |
Evolutionary Dynamics and Genomic Architecture of NLR Genes
Evolutionary Mechanisms Driving NLR Diversification
The NLR gene family exhibits extraordinary evolutionary dynamics characterized by rapid birth-death cycles and functional diversification. Several genetic mechanisms contribute to this evolutionary plasticity, including tandem duplication, segmental duplication, gene conversion, and domain shuffling [15] [46]. Tandem duplication serves as the primary driver of NLR family expansion, accounting for 18.4% of NLR genes in pepper and facilitating the rapid generation of novel resistance specificities through localized amplification [6]. These duplication events create genomic environments conducive to neo-functionalization and sub-functionalization, enabling plants to continually adapt to evolving pathogen effector repertoires.
The LRR domains of NLR genes experience particularly strong diversifying selection, especially at predicted solvent-exposed residues involved in protein-protein interactions [15]. This pattern reflects selective pressure to maintain binding specificity for rapidly evolving pathogen effectors. Comparative analyses reveal that NLR genes are frequently located in recombination-hotspots, which accelerates the generation of novel resistance alleles through sequence exchange between paralogous genes [15] [23]. This dynamic genomic organization creates a reservoir of genetic variation that can be rapidly mobilized in response to pathogen pressure.
Genomic Organization and Cluster Evolution
NLR genes display non-random distribution patterns across plant genomes, with pronounced clustering in specific genomic regions. Chromosomal distribution analyses in pepper reveal significant NLR clustering near telomeric regions, with chromosome 9 harboring the highest density (63 NLRs) [6]. Similarly, common bean features three 'super clusters' on the distal ends of chromosomes 4, 10, and 11, while related clustering patterns occur in potato, tomato, cotton, and Setaria italica [15].
These NLR clusters can be categorized as homogeneous (containing similar NLR types) or heterogeneous (containing diverse NLR classes), with some clusters additionally incorporating mixtures of NLR, RLP, and RLK genes [46]. The clustering of NLR genes into coregulatory modules may represent an evolutionary adaptation to reduce the metabolic costs of defense by enabling coordinated expression and functional specialization [47]. Pangenomic studies in Arabidopsis thaliana have identified 121 NLR neighborhoods that vary substantially in size, content, and complexity, highlighting the extensive intraspecific variation in NLR genomic architecture [23].
Figure 1: Evolutionary Dynamics of NLR Genes. The diagram illustrates key mechanisms, selection pressures, and outcomes driving NLR diversification in plant genomes.
Regulatory Mechanisms Mitigating NLR Costs
Transcriptional and Post-transcriptional Regulation
Plants employ sophisticated regulatory mechanisms to minimize the metabolic costs of NLR immunity while maintaining effective pathogen defense. Tight control of NLR gene expression occurs at multiple levels, including transcriptional regulation, post-transcriptional processing, and protein modification [15]. Approximately 82.6% of NLR promoters in pepper contain binding sites for salicylic acid (SA) and/or jasmonic acid (JA) signaling, enabling precise pathogen-responsive regulation [6]. This inducible expression strategy allows plants to maintain low basal NLR expression in the absence of pathogen challenge, reducing constitutive maintenance costs.
Meta-analyses of Arabidopsis NLR genes reveal complex transcript dynamics under different stress conditions, with most NLR genes induced by pathogens but repressed by abscisic acid, high temperature, and drought [48]. This opposing regulation under biotic versus abiotic stress conditions suggests that transcriptional reprogramming represents an important mechanism for balancing defense priorities under changing environmental conditions. Additionally, some NLR genes display SA-dependent induction patterns, while others are SA-independent, indicating diversification of regulatory networks controlling NLR expression [48].
Genomic and Epigenetic Strategies
The genomic organization of NLR genes into coregulatory modules represents another strategy for cost mitigation. By clustering functionally related NLR genes, plants can achieve coordinated expression through shared regulatory elements, potentially reducing the regulatory machinery required for individual gene control [47]. This organizational principle may explain the prevalence of NLR clusters across diverse plant species despite the potential evolutionary risks of linked inheritance.
Epigenetic mechanisms also contribute to NLR regulation, with histone modifications influencing expression dynamics and potentially facilitating transgenerational resistance priming [47]. Mutants defective in histone deacetylation, such as hos15-4, exhibit hyperactivated immune responses accompanied by upregulated expression of approximately one-third of NLR genes [48]. This connection between chromatin remodeling and NLR expression provides an additional layer of regulatory control that may fine-tune defense responses to minimize fitness costs.
Experimental Approaches and Research Methodologies
Genome-Wide NLR Identification and Characterization
Comprehensive profiling of NLR gene families relies on integrated bioinformatic and experimental approaches. The NLGenomeSweeper pipeline provides a specialized tool for genome-wide NLR identification with high specificity for complete functional genes, based on detection of the conserved NB-ARC domain using BLAST suite algorithms [50]. This approach typically combines homology searches using known NLR sequences with hidden Markov model (HMM) profiling of core NLR domains (PF00931) against whole proteomes [6]. Candidate sequences containing NB-ARC domains are subsequently validated through NCBI Conserved Domain Database (cd00204) and Pfam batch searches, with manual curation to remove redundancies and pseudogenes [6].
Additional computational tools have been developed to address specific challenges in NLR annotation. LRRpredictor utilizes an ensemble of classifiers to detect irregular LRR motifs in plant NLR proteins, overcoming limitations of standard motif detection methods faced with highly variable LRR domains [50]. For phylogenetic analysis, NB-ARC domain sequences or full-length NLR proteins are aligned using tools like Muscle v5, with maximum likelihood trees constructed in IQ-TREE with bootstrap validation [6]. These computational approaches enable researchers to reconstruct evolutionary relationships and identify lineage-specific innovations in NLR gene families.
Expression Profiling and Functional Validation
Transcriptional dynamics of NLR genes under various conditions are typically analyzed using RNA sequencing approaches. Experimental designs often compare resistant and susceptible cultivars under pathogen challenge conditions, with differential expression analysis conducted using tools like DESeq2 with thresholds of |log2 Fold Change| ≥ 1 and FDR < 0.05 [6]. Meta-analyses integrating multiple RNAseq datasets have proven valuable for identifying consistent expression patterns across diverse experimental conditions, as demonstrated by studies analyzing 88 datasets from 27 independent studies on Arabidopsis NLR genes [48].
Functional validation of candidate NLR genes employs multiple experimental approaches. Protein-protein interaction networks can be predicted using STRING database analyses, identifying potential hub genes with central regulatory roles [6]. Reverse genetics approaches, including T-DNA insertion lines and RNA interference, help establish gene-phenotype relationships, while transgenic complementation tests provide definitive functional validation. For NLR proteins with integrated domains (NLR-IDs), structural modeling using SWISS-MODEL can reveal potential effector binding interfaces and functional mechanisms [6] [50].
Figure 2: Experimental Workflow for NLR Gene Analysis. The diagram outlines integrated bioinformatic, transcriptomic, and functional approaches for comprehensive NLR characterization.
Table 3: Essential Research Reagents and Tools for NLR Studies
| Research Tool | Specific Application | Function and Utility |
|---|---|---|
| NLGenomeSweeper | Genome-wide NLR identification | Identifies NLR genes with high specificity for complete functional genes using BLAST-based NB-ARC detection [50] |
| LRRpredictor | Irregular LRR motif detection | Ensemble classifier method adapted for plant NLR irregularities; compensates for high sequence variability [50] |
| PlantCARE Database | Promoter cis-element analysis | Identifies defense-related regulatory motifs (SA/JA-responsive, WRKY-binding) in NLR promoters [6] |
| STRING Database | Protein-protein interaction prediction | Predicts interaction networks among NLR proteins; identifies potential hub genes with confidence scores [6] |
| SWISS-MODEL | Protein structure prediction | Generates homology models for NLR proteins; predicts functional domains and potential effector binding interfaces [6] |
| DESeq2 | Differential expression analysis | Statistical analysis of RNAseq data; identifies significantly regulated NLR genes under pathogen challenge [6] |
The metabolic costs associated with NLR maintenance and expression represent fundamental constraints on plant immunity that have shaped the evolutionary trajectory of this critical gene family. The trade-offs between growth and defense manifest across multiple biological scales, from cellular resource allocation to ecosystem-level fitness consequences. Plants have evolved sophisticated regulatory strategies to mitigate these costs, including inducible expression, genomic clustering, hormonal crosstalk, and epigenetic memory. Understanding these balancing mechanisms provides crucial insights for both evolutionary biology and crop improvement.
Future research directions should leverage emerging technologies to address key unanswered questions in NLR biology. Single-cell transcriptomics could reveal cell-type-specific expression patterns of NLR genes, potentially identifying specialized immune cells where defense costs are concentrated. Genome editing technologies like CRISPR-Cas9 enable precise manipulation of NLR regulatory elements to engineer optimal expression patterns that maximize resistance while minimizing fitness costs. Integrating NLR genomics with pangenome analyses across diverse accessions will further elucidate how evolutionary forces maintain functional diversity while managing metabolic constraints. These approaches will advance both fundamental understanding of plant immunity and practical applications in developing sustainable disease-resistant crops.
Plant domestication has dramatically altered the evolutionary trajectory of crop species, often resulting in a phenomenon known as the "domestication bottleneck"—a significant reduction in genetic diversity as humans selectively propagate plants with desirable traits. For nucleotide-binding leucine-rich repeat (NLR) genes, which encode crucial intracellular immune receptors in plants, this bottleneck may have profound consequences for disease resistance. NLR proteins recognize pathogen effector molecules and initiate robust immune responses, including programmed cell death at infection sites [51]. Their genomic repertoires are highly dynamic, evolving rapidly in response to pathogen pressure through duplication, recombination, and diversifying selection [15]. The central thesis of this review posits that artificial selection during domestication has consistently reduced NLR diversity, potentially compromising the immune resilience of cultivated species compared to their wild relatives. This NLR repertoire contraction represents an evolutionary trade-off, where selection for agronomic traits may have inadvertently relaxed pressure on immune gene maintenance. Understanding the extent, mechanisms, and consequences of this phenomenon is crucial for future crop improvement strategies aimed at enhancing disease resistance.
Comparative Genomics Evidence: Documenting NLR Loss Across Crop Families
Systematic Evidence of NLR Contraction
A comprehensive comparative genomics analysis of 15 domesticated crop species and their wild relatives across nine plant families has provided robust evidence for domestication-associated NLR repertoire contraction. The study revealed that five crops—grapes (Vitis vinifera subsp. vinifera), mandarins (Citrus reticulata), rice (Oryza sativa), barley (Hordeum vulgare), and yellow sarson (Brassica rapa var. yellow sarson)—exhibited significantly reduced immune receptor gene repertoires compared to their wild counterparts [52]. Notably, the overall rate of immune receptor gene loss generally reflected background rates of gene loss, suggesting a pattern of relaxed selection rather than strong selective sweeps against specific resistance genes. Furthermore, researchers identified a positive association between domestication duration and the extent of immune receptor gene loss, indicating that NLR repertoire contraction represents a subtle, cumulative pressure that intensifies over the domestication timeline [52].
Table 1: Documented Cases of NLR Repertoire Contraction in Domesticated Crops
| Crop Species | Plant Family | Wild Relative | Reduction Significance | Primary Drivers |
|---|---|---|---|---|
| Asparagus officinalis (garden asparagus) | Asparagaceae | A. setaceus, A. kiusianus | 57% reduction (27 vs 63 NLRs) | Artificial selection for yield/quality [28] |
| Vitis vinifera subsp. vinifera (grape) | Vitaceae | Wild grape relatives | P = 0.0018 | Domestication duration, relaxed selection [52] |
| Citrus reticulata (mandarin) | Rutaceae | Wild citrus relatives | P = 0.026 | Relaxed selection under cultivation [52] |
| Oryza sativa (rice) | Poaceae | Wild rice relatives | P = 0.046 | Domestication bottleneck, artificial selection [52] |
| Hordeum vulgare (barley) | Poaceae | Wild barley relatives | P = 0.0302 | Domestication duration, relaxed selection [52] |
| Brassica rapa var. yellow sarson | Brassicaceae | Wild Brassica relatives | P = 0.0222 | Relaxed selection under cultivation [52] |
Case Study: NLR Contraction in the Asparagus Genus
A compelling case study of NLR contraction comes from comparative analysis within the Asparagus genus. Research comparing garden asparagus (Asparagus officinalis) with its wild relatives (A. setaceus and A. kiusianus) revealed a dramatic 57% reduction in NLR gene count, from 63 NLRs in A. setaceus to just 27 in the domesticated A. officinalis [28]. Orthologous gene analysis identified only 16 conserved NLR gene pairs between A. setaceus and A. officinalis, representing the core NLR repertoire preserved during domestication. Pathogen inoculation experiments demonstrated functional consequences: domesticated asparagus was susceptible to Phomopsis asparagi infection, while the wild relative A. setaceus remained asymptomatic. Notably, most preserved NLR genes in the cultivated species showed either unchanged or downregulated expression following fungal challenge, indicating potential functional impairment in disease resistance mechanisms alongside the numerical reduction [28]. This case exemplifies how domestication can impact both the size and functionality of NLR repertoires.
Evolutionary Mechanisms Driving NLR Repertoire Contraction
Population Genetic Forces
The contraction of NLR repertoires during domestication primarily results from three interconnected evolutionary forces. First, relaxed selection occurs when human management practices reduce pathogen exposure, diminishing the selective pressure to maintain diverse NLR repertoires [52]. Second, the domestication bottleneck itself reduces genetic diversity genome-wide, with NLR repertoires being particularly affected due to their inherent variability [52]. Third, the cost of resistance hypothesis suggests that maintaining and expressing NLR genes carries metabolic burdens that may trade off with yield or growth traits favored during domestication [52] [15]. Evidence indicates that relaxed selection rather than strong cost-of-resistance effects predominates, as NLR gene loss typically occurs at background gene loss rates rather than showing patterns of strong selective sweeps [52].
Genomic Processes and Structural Variants
At the genomic level, NLR repertoire contraction occurs through several molecular mechanisms. Tandem gene loss through deletion events frequently impacts NLR clusters, particularly in pericentromeric and telomeric regions where NLRs often reside [53]. Presence-absence variations (PAVs) are common in NLR genes, with cultivated accessions showing increased frequency of absent genes compared to wild relatives [53]. Pseudogenization represents another pathway, where NLR genes accumulate disabling mutations without physical deletion from the genome [32]. Research on olive (Olea europaea) indicates that even partially pseudogenized NLRs may retain expression, though their immune function is likely compromised [32]. These genomic processes collectively reshape the NLR landscape during domestication, often resulting in streamlined but potentially vulnerable repertoires in cultivated varieties.
Table 2: Evolutionary Mechanisms and Genomic Processes in NLR Contraction
| Mechanism Category | Specific Process | Impact on NLR Repertoire | Evidence |
|---|---|---|---|
| Population Genetic Forces | Relaxed selection | Reduced maintenance of diverse NLRs | Association with domestication duration [52] |
| Domestication bottleneck | Stochastic loss of NLR alleles | Reduced diversity in crops vs. wild relatives [52] [28] | |
| Cost of resistance | Trade-offs with favored agronomic traits | Autoimmunity phenotypes in NLR-overexpressing lines [15] | |
| Genomic Processes | Tandem gene loss | Contraction of NLR clusters | Fewer NLR clusters in cultivated asparagus [28] |
| Presence-absence variation | Complete loss of specific NLR genes | PAVs distinguishing wild and cultivated barley [53] | |
| Pseudogenization | Non-functional but retained NLR sequences | Expressed pseudogenes in olive [32] |
Experimental Approaches for NLR Repertoire Analysis
Genome-Wide NLR Identification and Annotation
Accurate identification and annotation of NLR genes across plant genomes requires specialized bioinformatic pipelines. The following workflow represents state-of-the-art methodology for comprehensive NLR characterization:
The foundational step requires high-quality genome assemblies with accurate gene annotations, as incomplete assemblies significantly compromise NLR identification due to their clustered arrangement and sequence diversity [28]. The NLRtracker pipeline provides a specialized approach for mining NLR genes in a high-throughput manner, processing reference proteomes to identify canonical and divergent NLRs [32]. For individual species analysis, HMMER searches using the conserved NB-ARC domain (PF00931) as query, combined with BLASTp analyses against reference NLR protein sequences from related species, effectively identifies candidate NLR genes [28] [38]. Subsequent domain architecture validation using InterProScan and NCBI's Conserved Domain Database (CDD) confirms NLR identity and enables classification into subfamilies (TNL, CNL, RNL) based on N-terminal domains [28] [38]. This multilayered approach ensures comprehensive NLR annotation while minimizing false positives from truncated or pseudogenized sequences.
Expression and Functional Validation
Following identification, expression profiling and functional validation determine which NLR genes contribute to immune responses. RNA-seq transcriptomics of pathogen-infected and control tissues identifies differentially expressed NLR genes, with time-course experiments revealing early versus late responders [28] [38]. For example, pepper NLR transcriptome profiling after Phytophthora capsici infection identified 44 significantly differentially expressed NLR genes between resistant and susceptible cultivars [38]. Protein-protein interaction networks predicted through tools like STRING can reveal potential immune signaling complexes, with hub genes representing key regulatory nodes [38]. Orthologous gene analysis between cultivated and wild species identifies conserved NLR pairs that have been maintained during domestication and are therefore likely functionally important [28]. Finally, functional characterization through heterologous expression, gene silencing, or genome editing establishes causal relationships between specific NLR genes and disease resistance phenotypes [51].
Table 3: Research Reagent Solutions for NLR Studies
| Resource Category | Specific Tools | Application | Function |
|---|---|---|---|
| Bioinformatic Tools | NLRtracker [32] | Genome-wide NLR identification | Specialized pipeline for NLR mining in proteomes |
| HMMER (PF00931) [28] [38] | Domain-based NLR discovery | Identifies NB-ARC domain-containing proteins | |
| InterProScan/NCBI CDD [28] | Domain architecture validation | Confirms NLR identity and classifies subfamilies | |
| OrthoFinder [28] | Comparative genomics | Identifies orthologous NLR groups across species | |
| Genomic Resources | High-quality genome assemblies [28] | NLR repertoire characterization | Foundation for comprehensive NLR identification |
| Pangenome datasets [53] | Structural variant analysis | Captures NLR presence-absence variation across accessions | |
| Wild relative genomes [52] [28] | Domestication comparisons | Reference for quantifying NLR contraction | |
| Experimental Resources | RNA-seq datasets [32] [38] | Expression profiling | Identifies pathogen-responsive NLR genes |
| STRING database [38] | Protein interaction prediction | Maps potential NLR immune networks | |
| PlantCARE [28] [38] | Promoter analysis | Identifies defense-related cis-regulatory elements |
Implications for Crop Improvement and Future Directions
The systematic contraction of NLR repertoires during domestication represents both a challenge and opportunity for crop improvement programs. Understanding the specific NLR genes lost during domestication provides targets for precision breeding approaches aimed at reintroducing valuable resistance specificities from wild germplasm [28]. The discovery that NLR pseudogenes may retain expression suggests possible neofunctionalization opportunities, where compromised immune receptors might be reactivated through gene editing [32]. Pangenome approaches that capture the full spectrum of NLR diversity across wild and cultivated accessions will be essential for identifying rare resistance alleles lost during domestication bottlenecks [53]. Future research directions should focus on functionally characterizing conserved NLR orthologs that have been maintained across domestication history, as these likely represent core components of the plant immune system with non-redundant functions [28]. Additionally, exploring the potential trade-offs between NLR repertoire size and agronomic performance will inform breeding strategies that balance disease resistance with yield and quality traits.
The diagram below illustrates the integrated research pipeline for studying NLR contraction and its functional consequences:
In conclusion, understanding NLR repertoire contraction during domestication provides crucial insights for enhancing disease resistance in modern crops. By leveraging comparative genomics, evolutionary analysis, and functional validation, researchers can identify key NLR losses and develop strategies to reintroduce valuable resistance traits while maintaining agricultural productivity.
In plant immunity, nucleotide-binding leucine-rich repeat (NLR) proteins serve as critical intracellular sentinels, initiating robust defense responses upon pathogen detection. However, constitutive NLR activation triggers autoimmunity, resulting in pleiotropic effects that compromise growth and yield. MicroRNAs (miRNAs) have emerged as essential post-transcriptional regulators that fine-tune NLR expression, maintaining immune homeostasis while preserving defense readiness. This review synthesizes current understanding of miRNA-mediated control over NLR networks, detailing the mechanistic basis, evolutionary conservation, and experimental approaches for investigating this crucial regulatory layer. We highlight how this miRNA-NLR axis represents a sophisticated evolutionary adaptation enabling plants to balance the metabolic costs of immunity with effective pathogen defense, providing insights crucial for future crop improvement strategies.
NLR genes constitute one of the largest and most dynamic gene families in plant genomes, encoding intracellular immune receptors that recognize pathogen effectors and activate effector-triggered immunity (ETI) [6] [15]. This defense response often culminates in a hypersensitive response (HR), characterized by programmed cell death at the infection site, effectively restricting biotrophic pathogen growth [15]. However, maintaining a vast NLR repertoire and sustaining their signaling readiness carries significant metabolic costs, potentially impairing plant growth and development [15].
The constitutive activation of NLRs presents a fundamental dilemma in plant immunity. While plants require sufficient NLR diversity and expression to counter rapidly evolving pathogens, improper regulation can lead to autoimmunity—a state where defense responses activate in the absence of pathogens [15]. This autoimmunity manifests as stunted growth, reduced yield, and spontaneous lesion formation, significantly compromising plant fitness [15]. In Arabidopsis thaliana, for instance, specific allele combinations of two NLR genes (DM1 and DM2) in hybrids cause autoimmunity, illustrating the dangerous consequences of improper NLR regulation [15].
MicroRNAs (miRNAs) have emerged as pivotal regulators resolving this dilemma through precise, post-transcriptional control of NLR expression. These small (~21-24 nucleotide) non-coding RNAs fine-tune gene expression by guiding mRNA cleavage or translational repression, providing a rapid, reversible regulatory mechanism ideal for immune homeostasis [54] [55]. Recent research has revealed that many miRNAs target conserved nucleotide sequences encoding motifs within NLRs, including the P-loop in the NB-ARC domain, enabling broad regulation across extensive NLR repertoires [13]. This review examines the molecular mechanisms, evolutionary significance, and experimental investigation of miRNA-mediated control preventing constitutive NLR activation.
miRNA Biogenesis and Mechanism of Action
miRNA Biogenesis Pathway
MicroRNA biogenesis in plants involves a sophisticated, multi-step process that transforms primary transcripts into mature regulatory RNAs:
Transcription: miRNA genes are transcribed by RNA polymerase II into primary miRNAs (pri-miRNAs) containing 5' caps and 3' poly-A tails, which form characteristic hairpin structures [54] [56]. These pri-miRNAs can be located in intronic or exonic regions of coding sequences or exist as independent transcriptional units [54].
Nuclear Processing: The microprocessor complex, comprising DICER-LIKE1 (DCL1), HYPONASTIC LEAVES1 (HYL1), and SERRATE (SE), catalyzes the cleavage of pri-miRNAs into precursor miRNAs (pre-miRNAs) with hairpin structures approximately 70 nucleotides long [56]. DCL1 further processes pre-miRNAs into miRNA/miRNA* duplexes of 20-25 nucleotides with characteristic 2-nucleotide 3' overhangs [54].
Maturation and Loading: The miRNA/miRNA* duplex undergoes methylation by HUA ENHANCER1 (HEN1) for stabilization before export to the cytoplasm [56]. One strand (the guide miRNA) is selectively loaded into ARGONAUTE (AGO) proteins, forming the core of the miRNA-Induced Silencing Complex (miRISC), while the passenger strand (miRNA*) is typically degraded [54].
Table 1: Core Proteins in Plant miRNA Biogenesis and Function
| Protein Component | Function in miRNA Pathway | Domain/Characteristics |
|---|---|---|
| DCL1 | RNase III enzyme; processes pri-miRNA to pre-miRNA and pre-miRNA to miRNA/miRNA* duplex | Double-stranded RNA-binding domain, PAZ domain, two RNase III domains |
| HYL1 | dsRNA-binding protein; assists DCL1 in precise pri-miRNA processing | Double-stranded RNA-binding domain |
| SERRATE | Zinc finger protein; facilitates miRNA processing | Zinc finger protein, RNA-binding capability |
| HEN1 | Methyltransferase; adds methyl group to 3' ends of miRNA/miRNA* duplex | Small RNA methyltransferase domain |
| AGO1 | Core component of RISC; binds mature miRNA and slices/complements target mRNAs | PAZ and PIWI domains, RNA slicer activity |
Mechanisms of Gene Silencing
Mature miRISC complexes employ multiple mechanisms to regulate gene expression:
Post-Transcriptional Gene Silencing (PTGS): The canonical miRNA function involves guiding AGO proteins to complementary mRNA sequences, primarily in the 3' untranslated regions (UTRs) [54]. Plant miRNAs typically exhibit extensive complementarity to their targets, enabling AGO-catalyzed mRNA cleavage [55]. Additionally, miRNAs can repress translation without mRNA degradation through mechanisms that interfere with ribosomal scanning or protein synthesis initiation [54].
Transcriptional Gene Silencing (TGS): Nuclear-localized miRNAs can direct DNA methylation and histone modifications at genomic loci sharing complementarity, leading to epigenetic repression of transcription [54] [56]. This mechanism extends miRNA regulatory potential beyond post-transcriptional control.
Regulatory Network Integration: miRNAs function within complex regulatory networks, competing with RNA-binding proteins and interacting with long non-coding RNAs, which adds layers of regulation to miRNA accessibility and activity [54].
Diagram 1: Plant miRNA Biogenesis and Function
miRNA-Mediated Regulation of NLR Genes
Molecular Mechanisms of NLR Control
miRNAs employ several sophisticated molecular strategies to regulate NLR genes and prevent their constitutive activation:
Target Site Conservation: Multiple miRNAs target conserved nucleotide sequences encoding key functional motifs within NLR genes, particularly the P-loop within the NB-ARC domain [13]. This targeting strategy allows a limited number of miRNA families to regulate extensive NLR repertoires, providing an efficient mechanism for immune system homeostasis.
Transcriptional and Post-transcriptional Regulation: miRNAs can direct both the cleavage of NLR transcripts and the repression of their translation, enabling rapid adjustment of NLR protein levels without the energetic costs of continuous transcription [54] [13]. This dual mechanism allows plants to maintain NLR transcripts in a translationally repressed state that can be rapidly activated during genuine pathogen attack.
Feedback Integration: The miRNA-NLR regulatory network incorporates feedback mechanisms where NLR activation can influence miRNA expression, creating dynamic control circuits that fine-tune immune responses [15]. This reciprocal regulation enables precise temporal control over defense activation and termination.
Evolutionary Significance
The co-evolution of miRNAs and their NLR targets represents a crucial adaptation in land plants:
Lineage-Specific Expansion: As NLR gene families expanded dramatically in flowering plants, miRNA-based regulatory networks co-evolved to manage this increased complexity [13]. Bryophytes like Physcomitrella patens possess relatively small NLR repertoires (~25 NLRs), while angiosperms often contain hundreds to thousands, necessitating sophisticated control mechanisms [13].
Diversification and Specialization: miRNA families targeting NLRs have diversified alongside their targets, with some miRNAs showing lineage-specific emergence while others are deeply conserved across land plants [13]. This evolutionary pattern reflects the continuous arms race between plants and their pathogens, requiring constant innovation in immune regulation.
Fitness Cost Balancing: miRNA-mediated control of NLRs likely evolved to mitigate the fitness costs associated with maintaining large NLR repertoires and preventing autoimmunity [15] [13]. Plants with properly regulated NLR networks achieve an optimal balance between defense readiness and growth investment, maximizing evolutionary fitness in fluctuating pathogen environments.
Table 2: miRNA-Mediated NLR Regulation Across Plant Species
| Plant Species | Total NLR Genes | miRNA Families Targeting NLRs | Key Regulatory Features |
|---|---|---|---|
| Arabidopsis thaliana | ~150 [15] | Multiple families targeting P-loop | Balanced TNL and CNL regulation; telomeric clustering |
| Oryza sativa (Rice) | ~500 [15] | Conserved and lineage-specific miRNAs | Absence of TNLs; distinct regulatory needs |
| Malus domestica (Apple) | ~1000 [15] | Expanded miRNA families | NLR expansion correlated with perennial habit |
| Triticum aestivum (Wheat) | >2000 [15] [13] | Complex miRNA regulatory network | Polyploidy contributions to NLR repertoire |
| Capsicum annuum (Pepper) | 288 canonical [6] | Defense-responsive miRNAs | Promoter elements responsive to SA/JA signaling |
Experimental Approaches for Investigating miRNA-NLR Networks
Genome-Wide Identification and Annotation
Modern approaches for identifying miRNA-NLR regulatory networks combine computational predictions with high-throughput sequencing:
sRNA-seq Library Construction: Essential requirements include deep sequencing coverage (>10 million reads per library) and biological replication (minimum two independent replicates) to confidently detect miRNA* species and establish precise processing patterns [55]. Library preparation should capture the full size range of small RNAs (18-28 nucleotides) to distinguish miRNAs from other small RNA classes.
miRNA Annotation Criteria: Current standards require: (1) sequencing of both miRNA and miRNA* strands with characteristic 2-nucleotide 3' overhangs; (2) precursor hairpins ≤300 nucleotides without large internal loops or secondary stems; (3) predominant accumulation (>75%) of reads from exact miRNA or miRNA* sequences; and (4) exclusion of RNAs <20 or >24 nucleotides from miRNA annotation [55].
Target Prediction and Validation: Computational algorithms (e.g., psRNATarget, TargetFinder) identify potential miRNA targeting sites in NLR transcripts, followed by experimental validation through RLM-RACE to confirm cleavage sites, and transgenic approaches expressing miRNA-resistant NLR versions to assess functional consequences [13].
Functional Characterization Methods
Several established experimental protocols enable functional investigation of miRNA-mediated NLR regulation:
Protocol 1: High-Throughput miRNA-mRNA Interaction Validation
- Materials: Plant tissues under study, sRNA-seq library preparation kit, RNA extraction reagents, 5'-RLM RACE kit, computational resources for target prediction.
- Procedure:
- Extract total RNA from tissues representing different developmental stages and immune conditions.
- Construct sRNA-seq libraries with adapter ligation, reverse transcription, and PCR amplification.
- Sequence libraries with sufficient depth to detect low-abundance miRNAs and miRNA* species.
- Process reads through bioinformatic pipelines (adapter trimming, size selection, alignment).
- Identify miRNAs meeting annotation criteria and predict NLR targets.
- Validate interactions experimentally using 5'-RLM RACE to confirm cleavage at predicted sites.
Protocol 2: Functional Assessment Through Virus-Induced Gene Silencing (VIGS)
- Materials: VIGS vectors (e.g., TRV-based), Agrobacterium tumefaciens strains, syringe infiltration equipment, pathogen isolates for challenge assays.
- Procedure:
- Clone fragments of candidate miRNAs or their NLR targets into VIGS vectors.
- Transform constructs into Agrobacterium and infiltrate into plant leaves.
- Monitor plants for autoimmunity symptoms (stunting, lesion formation) indicating NLR dysregulation.
- Challenge with pathogens to assess immunity functionality.
- Quantify NLR transcript and protein levels to confirm regulatory relationships.
- Document growth-defense tradeoffs through biomass and yield measurements.
Diagram 2: Experimental Workflow for miRNA-NLR Investigation
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Investigating miRNA-NLR Networks
| Reagent/Category | Specific Examples | Research Application | Technical Considerations |
|---|---|---|---|
| Sequencing Kits | Illumina TruSeq Small RNA Kit; NEBNext Small RNA Library Prep | sRNA-seq library construction for miRNA discovery | Size selection critical for enriching authentic miRNAs |
| VIGS Vectors | Tobacco Rattle Virus (TRV)-based vectors; Barley Stripe Mosaic Virus (BSMV) vectors | Functional analysis through targeted gene silencing | Efficiency varies by plant species; requires optimization |
| AGO Antibodies | Anti-AGO1 immuno-precipitation grade antibodies | RIP-seq to identify endogenous miRNA targets | Cross-reactivity considerations across plant species |
| Target Prediction Tools | psRNATarget; TargetFinder; miRanda | Computational identification of miRNA-NLR pairs | Plant-specific parameters improve prediction accuracy |
| RACE Kits | 5'-RLM RACE Kit; GeneRacer Kit | Experimental validation of miRNA cleavage sites | Requires detection of cleaved mRNA fragments |
| Expression Vectors | 35S-driven miRNA overexpression; MIR gene genomic clones | Gain-of-function studies | Genomic context important for proper miRNA processing |
MicroRNA-mediated regulation of NLR genes represents a sophisticated evolutionary adaptation that enables plants to manage the inherent risks of maintaining a powerful immune system. By preventing constitutive NLR activation, miRNAs resolve the fundamental conflict between defense readiness and growth investment, allowing plants to optimize fitness in pathogen-rich environments. The mechanistic insights into this regulatory layer not only advance our fundamental understanding of plant immunity but also provide promising targets for future crop improvement.
Future research directions should focus on elucidating the dynamic regulation of miRNA-NLR networks across diverse environmental conditions, understanding how pathogen effectors might manipulate this regulatory layer, and exploring the potential of synthetic miRNA technologies for engineering disease resistance in crop plants. As we deepen our understanding of these sophisticated regulatory networks, we move closer to designing crops with optimally balanced immune systems—achieving durable disease resistance without compromising productivity.
Plant immunity is fundamentally shaped by a continuous coevolutionary struggle with pathogens, a dynamic process often described as an endless race for recognition specificity. This interaction is primarily governed by effector-triggered immunity (ETI), where intracellular immune receptors encoded by Nucleotide-binding leucine-rich repeat (NLR) genes recognize specific pathogen effectors, leading to a robust defense response often including programmed cell death [6]. The evolutionary dynamics between plants and their pathogens exist on a continuum between two classical models: "arms-race" dynamics, characterized by recurrent selective sweeps and rapid allele replacements, and "trench-warfare" dynamics (Red Queen dynamics), where multiple alleles are maintained over long periods through balancing selection [57]. The precise position on this spectrum is determined by complex interactions between negative frequency-dependent selection, genetic drift, and mutation, creating an extraordinarily diverse NLR repertoire across plant species [23] [57].
Table 1: Key Concepts in Plant-Pathogen Coevolution
| Concept | Description | Evolutionary Signature |
|---|---|---|
| Arms-Race Dynamics | Recurrent selective sweeps where new resistance/infectivity alleles rapidly fixate | Signatures of positive selection: reduced genetic diversity, increased linkage disequilibrium |
| Trench-Warfare Dynamics | Stable maintenance of multiple alleles over long time periods through balancing selection | Signatures of balancing selection: higher-than-average genetic diversity, stable polymorphisms |
| Gene-for-Gene (GFG) Interaction | Infection matrix where one universally infective parasite genotype interacts with specific host resistance genotypes | Characterized by specific fitness costs (infection, resistance, infectivity) determining equilibrium frequencies |
| Negative Frequency-Dependent Selection | Fitness advantage of alleles when they are rare, inversely proportional to their own frequency or allele frequencies in interacting partner | Maintains genetic diversity over time; necessary for trench-warfare dynamics |
Molecular Mechanisms of NLR Function and Evolution
NLR Protein Architecture and Signaling
NLR proteins function as sophisticated molecular switches with a characteristic modular structure that enables pathogen detection and immune signaling activation. The core architecture consists of: (1) an N-terminal signaling domain (typically Toll/Interleukin-1 Receptor homology [TIR], Coiled-Coil [CC], or RPW8-like domain) that initiates downstream signaling cascades; (2) a central nucleotide-binding domain (NBS) that serves as a molecular switch regulated by nucleotide binding and hydrolysis; and (3) a C-terminal leucine-rich repeat (LRR) domain responsible for effector recognition or mediating protein interactions [6]. This sophisticated architecture enables NLRs to detect direct or indirect effector interference through conformational changes, subsequently activating robust immune signaling pathways [6].
Genomic Drivers of NLR Diversity
The remarkable diversity of the NLR gene family is generated through specific genomic mechanisms that facilitate rapid adaptation to evolving pathogen effectors. Tandem duplication has been identified as the primary driver of NLR family expansion, accounting for approximately 18.4% of NLR genes in pepper (Capsicum annuum), with these duplicated genes predominantly clustered on specific chromosomes (Chr08 and Chr09) [6]. This clustering, particularly near telomeric regions, creates genomic environments conducive to rapid generation of new resistance alleles through local amplification and recombination [6]. Additional mechanisms including segmental duplication and retrotransposition further contribute to NLR repertoire expansion and diversification across plant genomes [6]. Recent pangenomic analyses in Arabidopsis thaliana have revealed that NLRs are diverse across many axes, with 3,789 NLRs identified across 17 diverse accessions organized into 121 pangenomic NLR neighborhoods that vary substantially in size, content, and complexity [23].
Empirical Evidence from Plant Systems
Genome-Wide NLR Analysis in Pepper
A comprehensive genome-wide identification of NLR genes in pepper (Capsicum annuum) using the high-quality 'Zhangshugang' reference genome revealed 288 high-confidence canonical NLR genes with non-random genomic distribution [6]. Chromosome 09 exhibited the highest NLR density, harboring 63 NLR genes, predominantly clustered near telomeric regions [6]. This strategic positioning facilitates rapid evolution of recognition specificities through localized recombination and duplication events. Promoter analysis of these NLR genes demonstrated significant enrichment in defense-related cis-regulatory elements, with 82.6% (238 genes) containing binding sites for salicylic acid (SA) and/or jasmonic acid (JA) signaling pathways, highlighting the intricate regulatory networks controlling NLR expression [6].
Table 2: Chromosomal Distribution of NLR Genes in Capsicum annuum
| Chromosome | Number of NLR Genes | Notable Features |
|---|---|---|
| Chr01 | 24 | - |
| Chr02 | 17 | - |
| Chr03 | 21 | Contains Caz03g40070 candidate gene |
| Chr04 | 14 | - |
| Chr05 | 19 | - |
| Chr06 | 16 | - |
| Chr07 | 18 | - |
| Chr08 | 35 | High tandem duplication activity |
| Chr09 | 63 | Highest NLR density; telomeric clustering |
| Chr10 | 25 | Contains Caz10g20900 and Caz10g21150 candidates |
| Chr11 | 20 | - |
| Chr12 | 16 | - |
Expression Dynamics During Pathogen Challenge
Transcriptome profiling of pepper cultivars with contrasting resistance to Phytophthora capsici revealed sophisticated temporal and genotypic expression patterns of NLR genes. Analysis of resistant (CM334) versus susceptible (NMCA10399) cultivars identified 44 significantly differentially expressed NLR genes following pathogen infection [6]. Protein-protein interaction network analysis predicted key interactions among these differentially expressed NLRs, with Caz01g22900 and Caz09g03820 emerging as potential network hubs, suggesting their central role in coordinating immune responses [6]. This study identified several strong candidate NLR genes for functional validation, including Caz03g40070, Caz09g03770, Caz10g20900, and Caz10g21150, which represent valuable targets for developing molecular markers for pepper resistance breeding programs [6].
Methodologies for Studying Coevolutionary Dynamics
Genomic Identification and Characterization of NLR Genes
Comprehensive NLR Identification Pipeline: The identification and annotation of NLR genes requires a multi-step computational approach that leverages both homology-based and domain-based search strategies [6]. Initially, known NLR protein sequences from reference species (e.g., Arabidopsis thaliana) are used as queries for BLASTp searches against the target species proteome [6]. Concurrently, HMMER searches should be performed against the entire proteome using the core NLR domain (PF00931, NB-ARC) with an E-value cutoff of 1×10⁻⁵ [6]. Candidate sequences containing NB-ARC domains are retained, and redundancy is manually removed. The remaining candidates must be validated using NCBI Conserved Domain Database (cd00204 for NB-ARC) and Pfam batch searches to confirm domain architecture and completeness [6]. Additional validation includes checking for presence/completeness of N-terminal (TIR, CC, RPW8) and C-terminal (LRR) domains.
Evolutionary and Phylogenetic Analysis: For phylogenetic reconstruction, NB-ARC domain sequences (or full-length sequences) of identified NLRs should be aligned using Muscle v5 with automatic settings [6]. Maximum Likelihood trees are constructed using IQ-TREE with 1000 bootstrap replicates to assess node support, using NLRs from related species (e.g., Arabidopsis and Solanum lycopersicum) as outgroups [6]. Gene duplication events and synteny relationships can be analyzed using MCScanX implemented in TBtools, with synteny plots generated using Advanced Circos visualization [6].
Inferring Coevolutionary Parameters from Genomic Data
Approximate Bayesian Computation Framework: A sophisticated method has been developed to infer coevolutionary dynamics and parameters from population genomic data of host and pathogen pairs [57]. This approach couples a gene-for-gene model with coalescent simulations and uses Approximate Bayesian Computation (ABC) to estimate key parameters of past coevolutionary history [57]. The method requires polymorphism data from both host and parasite populations at candidate coevolving loci, ideally with multiple replicates (10-30 repetitions) from controlled experiments or natural populations to control for the effects of genetic drift [57].
Key Parameters in Coevolutionary Inference: The ABC framework enables simultaneous estimation of three fundamental fitness costs that define coevolutionary dynamics: (1) Cost of infection (s): the fitness loss experienced by hosts upon successful infection; (2) Cost of resistance (cH): the fitness cost paid by resistant hosts in absence of the parasite, such as reduced competitive ability; and (3) Cost of infectivity (cP): the fitness cost incurred by highly infective pathogens, such as reduced spore production [57]. These parameters collectively determine equilibrium allele frequencies and the strength of coevolutionary signatures detectable in genomic data.
Network Visualization and Analysis of NLR Interactions
The analysis of NLR networks and their interactions requires specialized visualization and analysis tools capable of handling complex biological networks. Cytoscape provides a comprehensive platform for visualizing molecular interaction networks and integrating gene expression profiles and other molecular data [58] [59]. It supports various file formats including SIF, GML, XGMML, BioPAX, PSI-MI, and SBML, and offers extensive plugin ecosystems for specialized analyses [58]. For programming-based approaches, NetworkX (Python) and igraph (multiple languages) provide comprehensive libraries for network analysis, including algorithms for calculating shortest paths, centrality measures, and community detection [59]. These tools enable researchers to identify network hubs, cluster co-expressed NLR genes, and visualize complex interaction networks underlying immune signaling.
Table 3: Experimental Reagents and Computational Tools for NLR Research
| Category | Specific Tool/Reagent | Function/Application |
|---|---|---|
| Genome Analysis | HMMER v3.3.2 | Identification of NLR domains in proteome datasets |
| Genome Analysis | MCScanX (TBtools) | Synteny and gene duplication analysis |
| Phylogenetics | IQ-TREE | Maximum likelihood phylogenetic reconstruction |
| Expression Analysis | HISAT2 + DESeq2 | RNA-seq read alignment and differential expression |
| Network Visualization | Cytoscape | Biological network visualization and analysis |
| Network Analysis | NetworkX (Python) | Complex network analysis and algorithm implementation |
| Coevolution Inference | Approximate Bayesian Computation | Parameter estimation from host-parasite polymorphism data |
| Protein Interaction | STRING database | Prediction of protein-protein interaction networks |
The coevolutionary dynamics between plants and their pathogens represent a continuous molecular arms race driven by competing survival strategies. NLR genes stand at the forefront of this battle, evolving through complex genomic mechanisms including tandem duplication, segmental duplication, and positive selection to maintain recognition specificity against rapidly adapting pathogens. The integration of pangenomic approaches with functional studies and sophisticated computational frameworks like Approximate Bayesian Computation provides unprecedented insights into the parameters governing these coevolutionary dynamics. Future research directions should focus on integrating multi-omics data across broader phylogenetic scales, developing more sophisticated models of NLR network dynamics, and translating this fundamental knowledge into crop improvement strategies through marker-assisted selection and genome editing approaches. Understanding the endless race for recognition specificity not only reveals fundamental evolutionary principles but also provides critical tools for enhancing crop resilience in sustainable agricultural systems.
Functional Validation and Cross-Kingdom Insights: From Plant Immunity to Human Disease Paradigms
Within the sophisticated innate immune system of plants, nucleotide-binding leucine-rich repeat (NLR) proteins serve as critical intracellular immune receptors that mediate effector-triggered immunity (ETI) [51]. This robust defense response is typically characterized by a form of programmed cell death known as the hypersensitive response (HR), which acts to restrict pathogen colonization and proliferation [6]. The HR presents as rapid, localized cell death at the site of pathogen recognition, effectively creating a biological barrier that prevents pathogen spread [51]. From an evolutionary perspective, NLR genes represent some of the most diverse and rapidly evolving sequences in plant genomes, exhibiting extraordinary sequence, structural, and regulatory variability as a result of the constant arms race with rapidly evolving pathogens [51] [23]. This diversity arises through multiple uncorrelated mutational and genomic processes, including tandem duplications, segmental duplication, and retrotransposition, with NLRs often clustering in complex genomic neighborhoods [6] [23]. Functional assays that validate NLR immune activation through the hypersensitive response are therefore essential tools for dissecting plant-pathogen co-evolution and identifying key genetic components of disease resistance in land plants.
NLR Biology and Hypersensitive Response Mechanisms
NLR Architecture and Activation Dynamics
NLR proteins function as molecular switches within the plant immune system, featuring a conserved tripartite domain architecture [51] [60]. This architecture consists of: (1) an N-terminal signaling domain (typically coiled-coil (CC), Toll/interleukin-1 receptor (TIR), or RPW8-type domains) that initiates downstream immune signaling; (2) a central nucleotide-binding domain (NB-ARC) that serves as a molecular switch through ADP/ATP exchange; and (3) a C-terminal leucine-rich repeat (LRR) domain involved in effector recognition and autoinhibition [51]. In their resting state, NLRs maintain an autoinhibited conformation, often as monomers or homodimers, with intramolecular interactions preventing unintended activation [60]. Recent structural studies of the helper NLR NRC2 from Nicotiana benthamiana have revealed that it accumulates as a homodimer in its resting state, with three distinct intermolecular interfaces contributing to autoinhibition [60]. Upon pathogen perception, NLRs undergo significant conformational changes, exchanging ADP for ATP and transitioning to active oligomeric complexes known as resistosomes [60].
The following diagram illustrates the transition of an NLR from its autoinhibited state to an active resistosome, a process that culminates in the hypersensitive response:
Molecular Events in Hypersensitive Response Execution
The transition from NLR activation to hypersensitive response execution involves a meticulously coordinated cascade of molecular events. For CC-NLRs like ZAR1 and NRCs, resistosome formation enables direct insertion into the plasma membrane, where they function as calcium-permeable channels [60]. This calcium influx serves as a critical secondary messenger, triggering downstream immune signaling cascades that include: (1) burst of reactive oxygen species (ROS); (2) activation of defense-related genes; (3) callose deposition and cell wall fortification; (4) production of antimicrobial compounds; and (5) eventual programmed cell death [61]. The HR cell death program is characterized by cytoplasmic condensation, chromatin fragmentation, and organelle disintegration, ultimately creating a necrotic lesion that confines the pathogen [61] [51]. This strategic sacrifice of infected and surrounding cells effectively deprives biotrophic and hemibiotrophic pathogens of living tissue, halting disease progression. The entire process from pathogen recognition to visible HR can occur within a few hours, making it a valuable rapid readout for NLR functionality in experimental settings.
Experimental Framework for HR-Based NLR Validation
Core Assay Workflow and Design Considerations
Validating NLR immune activation through hypersensitive response requires a multidisciplinary approach that combines molecular biology, protein biochemistry, and plant pathology techniques. The following diagram outlines a comprehensive experimental workflow for HR-based NLR validation:
When designing HR validation assays, several critical considerations must be addressed. First, researchers must select appropriate expression systems based on their experimental goals—transient expression in Nicotiana benthamiana offers rapid screening capabilities, while stable transformation in target crops provides insights into physiological relevance [61] [60]. Second, the method of immune activation should be carefully chosen: natural pathogen infection, effector delivery, or engineered systems such as protease-activated NLRs that trigger upon detection of pathogen-derived proteases [61]. Third, proper controls are essential, including: (1) inactive NLR mutants with disrupted nucleotide-binding (Walker A or B mutations); (2) autoactive mutants as positive controls; and (3) vector-only controls to establish baseline responses [60]. Finally, experimental timing must be optimized, as HR readouts are time-sensitive and can vary from 12 to 72 hours post-induction depending on the NLR-pathogen system.
Quantitative Assessment of Hypersensitive Response
Robust quantification of hypersensitive response is essential for validating NLR immune activation. Multiple parameters should be assessed to comprehensively characterize the HR phenotype:
Table 1: Hypersensitive Response Quantification Metrics
| Parameter Category | Specific Metrics | Measurement Methods | Typical Timeframe |
|---|---|---|---|
| Cell Death Progression | Lesion diameter, Cell viability, Ion leakage | Evans blue staining, Trypan blue exclusion, Conductivity measurement | 24-72 hours post-induction |
| Immune Signaling Markers | ROS burst, Callose deposition, Defense gene expression | DAB staining, Aniline blue fluorescence, RT-qPCR | 2-24 hours post-induction |
| Pathogen Restriction | Pathogen biomass, Sporulation, Infection progression | CFU counting, qPCR for pathogen DNA, Microscopic assessment | 48-96 hours post-infection |
| Structural Changes | NLR oligomerization, Subcellular localization | BN-PAGE, Size-exclusion chromatography, Confocal microscopy | 4-24 hours post-induction |
Data from recent studies demonstrate the effectiveness of these quantification methods. For example, in pepper (Capsicum annuum) response to Phytophthora capsici infection, transcriptome profiling identified 44 significantly differentially expressed NLR genes, with functional validation through HR assays confirming their role in disease resistance [6]. Similarly, engineering of pathogen protease-activated autoactive NLRs resulted in rapid induction of hypersensitive response and elevated expression of defense-related genes, showcasing the potent immune activation achievable through NLR manipulation [61].
Advanced Methodologies and Technical Approaches
Experimental Protocols for Key Assays
Agrobacterium-mediated Transient Expression in N. benthamiana This widely adopted protocol enables rapid screening of NLR function and is particularly valuable for assessing HR induction. Fresh Agrobacterium tumefaciens strains (GV3101 or LBA4404) harboring NLR expression constructs are grown overnight in appropriate selective media. Bacterial cells are pelleted and resuspended in infiltration buffer (10 mM MES, 10 mM MgCl₂, 150 μM acetosyringone, pH 5.6) to an OD₆₀₀ of 0.2-0.5. For co-infiltration experiments with pathogen effectors, bacterial suspensions are mixed in 1:1 ratio prior to infiltration. The abaxial side of 4-6 week old N. benthamiana leaves is infiltrated using a needleless syringe. Plants are maintained under standard growth conditions (22-25°C, 16h light/8h dark) and monitored for HR development over 24-72 hours. This method was successfully employed in characterizing NRC2 activation, where co-expression with the upstream sensor NLR Rx triggered HR [60].
Ion Leakage Measurement for Quantifying Cell Death This quantitative approach provides objective assessment of HR-induced membrane integrity loss. Leaf discs (typically 6-8 mm diameter) are collected from infiltrated zones at specified timepoints and rinsed briefly in distilled water to remove surface ions. Discs are placed in tubes containing 10 mL of distilled water and vacuum-infiltrated for 15 minutes. Initial conductivity (C₁) is measured using a conductivity meter. Tubes are then incubated with shaking at room temperature for 3-6 hours, followed by second conductivity measurement (C₂). Finally, samples are autoclaved or frozen-thawed to release all ions, and total conductivity (C₃) is measured. Ion leakage is calculated as: [(C₂ - C₁) / C₃] × 100%. This method reliably detects significant differences in ion leakage between activated NLR expressions and controls, with autoactive NLR mutants typically showing 3-5 fold increases compared to wild-type receptors [60].
Blue Native PAGE for NLR Oligomerization Analysis This technique detects the formation of higher-order NLR complexes during activation. Plant tissue (0.5-1 g) is harvested and ground in liquid nitrogen, then homogenized in extraction buffer (50 mM Tris-HCl pH 7.5, 150 mM NaCl, 10% glycerol, 1% digitonin, 1 mM PMSF, and protease inhibitor cocktail). Extracts are centrifuged at 15,000 × g for 15 minutes at 4°C, and supernatants are mixed with NativePAGE sample buffer and G-250 additive. Samples are loaded onto 4-16% Bis-Tris NativePAGE gels and electrophoresed at 150 V for 1-2 hours with cathode buffer (containing 0.02% Coomassie G-250) and anode buffer. Proteins are transferred to PVDF membranes for immunoblotting with NLR-specific antibodies. This approach confirmed that helper NLR NRC2 transitions from homodimers to higher-order oligomers upon activation by upstream sensor NLRs [60].
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Research Reagents for NLR-HR Functional Assays
| Reagent Category | Specific Examples | Function & Application |
|---|---|---|
| Expression Systems | Gateway-compatible vectors (pEarleyGate, pGWB), 35S promoter, Ubiquitin promoter | High-level protein expression in plant systems, modular cloning |
| Plant Materials | N. benthamiana wild-type, NLR knockout lines (nrc2/3/4 KO), Crop cultivars with varying resistance | Transient expression, Genetic background control, Comparative studies |
| Pathogen Strains | Phytophthora capsici, Pseudomonas syringae, Xanthomonas species, Virus vectors (TRV, PVX) | Natural pathogen challenge, Effector delivery, Disease resistance assessment |
| Detection Reagents | α-NLR antibodies, HRP-conjugated secondary antibodies, Evans blue, DAB staining solution | Protein detection, Cell death visualization, ROS detection |
| Chemical Inhibitors | DPI (NADPH oxidase inhibitor), LaCl₃ (calcium channel blocker), Cycloheximide (protein synthesis inhibitor) | Dissecting signaling pathways, Identifying downstream components |
| Molecular Biology Tools | CRISPR/Cas9 systems for gene editing, RNAi constructs for silencing, Luciferase reporter genes | Generating mutant lines, Functional validation, Promoter activity analysis |
Recent technological advances have significantly expanded the research toolkit for NLR-HR studies. CRISPR/Cas-mediated genome editing enables precise insertion of protease cleavage motifs into NLRs without destabilizing native receptor architecture, creating engineered receptors activated by pathogen proteases [61]. Additionally, the development of modular NLR systems, such as the NRC immune receptor network in solanaceous plants, allows researchers to study how multiple sensor NLRs signal redundantly via arrays of downstream helper NLRs [60]. The integration of cryo-EM structural biology with functional assays has further enhanced our understanding, revealing how helper NLRs like NRC2 transition from autoinhibited homodimers to active oligomeric resistosomes [60].
Data Interpretation and Evolutionary Context
Analysis of NLR-HR Assay Results
Interpreting HR assay data requires careful consideration of both quantitative metrics and temporal patterns. Positive HR validation is characterized by: (1) statistically significant increase in cell death markers (ion leakage, dye uptake) compared to controls; (2) temporal correlation between NLR expression/activation and HR development; (3) spatial restriction of cell death to sites of NLR activation; and (4) concomitant induction of defense gene expression and pathogen restriction. However, false positives can arise from non-specific cytotoxicity, while false negatives may result from insufficient expression, improper timing, or suppression by pathogen effectors. Recent studies of pepper NLR responses to Phytophthora capsici demonstrate the importance of comprehensive analysis—while 44 NLRs showed differential expression during infection, only subset validation confirmed their direct role in HR-mediated resistance [6].
The table below summarizes quantitative data from recent NLR studies, highlighting the relationship between NLR activation metrics and HR outcomes:
Table 3: Quantitative NLR Immune Activation Parameters from Recent Studies
| NLR System | Activation Trigger | HR Onset | Ion Leakage Increase | Defense Gene Induction | Pathogen Restriction |
|---|---|---|---|---|---|
| Engineered Protease-Activated NLRs [61] | Pathogen protease cleavage | 18-24 hours | 3.5-4.8 fold | 5-12 fold (PR genes) | 85-95% reduction in pathogen biomass |
| Pepper NLRs vs Phytophthora [6] | P. capsici infection | 24-48 hours | 2.5-4.2 fold | 3-8 fold (SA/JA markers) | 70-90% reduction in sporulation |
| NRC2 Helper NLR [60] | Rx sensor NLR activation | 16-20 hours | 4.5-5.2 fold | 8-15 fold (ETI markers) | Not quantified |
| Autoactive Mutants [60] | Disrupted autoinhibition | 12-16 hours | 5.8-7.3 fold | 10-20 fold (HR-related genes) | Not applicable |
Evolutionary Insights from HR-Assayed NLRs
Functional HR assays provide critical insights into the evolutionary dynamics of NLR genes across land plants. Comparative analyses reveal that NLRs exhibit lineage-specific expansions and contractions, primarily driven by tandem duplication events in response to pathogen pressures [6] [23]. In pepper genomes, approximately 18.4% of NLR genes (53 of 288 identified) arose through tandem duplication, with notable clustering on chromosomes 08 and 09 [6]. Promoter analysis of these NLRs reveals enrichment in defense-related cis-regulatory elements, with 82.6% containing binding sites for salicylic acid (SA) and/or jasmonic acid (JA) signaling pathways [6], indicating coordinated regulatory evolution alongside coding sequence diversification.
The NRC helper NLR network exemplifies how functional specialization has evolved within NLR families. Phylogenomic analysis reveals that homodimerization interfaces have diverged among NRC paralogs, creating molecular insulation that prevents undesired cross-activation while maintaining genetic redundancy [60]. This evolutionary innovation enables solanaceous plants to deploy multiple parallel helper NLR pathways, enhancing both robustness and evolvability of the immune system. Similarly, engineering of pathogen protease-activated NLRs capitalizes on evolutionary constraints—since proteases are essential virulence factors that pathogens cannot easily mutate or dispense with, receptors targeting these enzymes demonstrate enhanced durability against pathogen escape [61]. These evolutionary principles inform strategic selection of NLR targets for crop improvement, emphasizing the importance of targeting conserved pathogen processes rather than highly variable effectors.
Plant nucleotide-binding domain and leucine-rich repeat receptors (NLRs) constitute a major line of defense against pathogen invasion, operating through effector-triggered immunity (ETI) that often culminates in a hypersensitive response (HR) to limit pathogen spread [62] [63]. These intracellular immune receptors recognize pathogen effector proteins either directly or indirectly, activating robust defense signaling cascades [64]. In land plants, NLR genes have undergone significant lineage-specific expansions, resulting in substantial diversity that reflects continuous evolutionary arms races with pathogens [65]. The study of NLR evolution provides crucial insights into how plants adapt to changing pathogenic threats over evolutionary timescales.
This whitepaper examines two compelling case studies of NLR-mediated resistance in agronomically important crops: resistance to blast fungus (Magnaporthe oryzae) in rice and viral defense mechanisms in cotton. Through these cases, we explore the molecular mechanisms, evolutionary dynamics, and potential biotechnological applications of NLR genes in crop protection.
Rice Blast Resistance: The NLR Defense System
Rice blast, caused by the fungal pathogen Magnaporthe oryzae, represents one of the most devastating diseases affecting global rice production, causing yield losses of up to 30% annually and threatening food security [66]. The interaction between rice and M. oryzae has become a model system for understanding plant-fungal pathogen interactions, particularly concerning NLR-mediated immunity.
Molecular Mechanisms of NLR Function in Rice Blast Resistance
Rice employs a sophisticated two-tiered innate immune system against blast fungus. The first layer, pathogen-associated molecular pattern (PAMP)-triggered immunity (PTI), occurs when cell surface pattern recognition receptors (PRRs) detect conserved microbial patterns [66]. However, M. oryzae secretes effector proteins to suppress PTI, leading to the activation of the second layer—effector-triggered immunity (ETI)—mediated predominantly by NLR proteins [66] [63].
NLR proteins in rice blast resistance typically function as paired receptors with specialized roles. The current model suggests a helper NLR (such as RGA4 or Pias-1) is responsible for initiating defense signaling and HR, while its partnered sensor NLR (such as RGA5 or Pias-2) carries integrated domains that directly or indirectly recognize pathogen effectors [65]. Upon effector recognition by the sensor, suppression of the helper is relieved, triggering defense activation [65].
Table 1: Major Cloned NLR Genes Conferring Blast Resistance in Rice
| NLR Gene | Chromosomal Location | Protein Type | Recognized Effector | Functional Characteristics |
|---|---|---|---|---|
| Pias (allelic to Pia) | Not specified | Paired NLR: Pias-1 (helper) and Pias-2 (sensor) | AVR-Pias | Sensor Pias-2 carries C-terminal DUF761 domain; allelic to Pia system |
| Pia | Not specified | Paired NLR: RGA4 (helper) and RGA5 (sensor) | AVR-Pia/AVR1-CO39 | Sensor RGA5 contains HMA domain that directly binds AVR-Pia |
| Pi9 | Not specified | NBS-LRR | Not specified | First cloned major broad-spectrum blast resistance gene |
| Pik | Not specified | Allelic series: Pik, Pik-m, Pik-p, etc. | AVR-Pik | NLR pairs with integrated heavy metal-associated (HMA) domains |
| Piz-t | Not specified | NLR | AVR-Pizt | Confers resistance against specific blast strains |
| Pib | Not specified | NLR | Not specified | Early cloned blast resistance gene |
| Pi54 | Not specified | NLR | Not specified | Cloned blast resistance gene |
Evolutionary Dynamics of Rice NLR Genes
The evolutionary history of NLR genes in rice reveals remarkable adaptive dynamics. Phylogenomic analyses indicate that sensor NLRs undergo highly dynamic evolution with recurrent genomic recombination, resulting in diverse integrated domains that enable recognition of sequence-divergent pathogen effectors [65]. This diversification is maintained by balancing selection across different Oryza lineages. In contrast, helper NLRs exhibit evolutionary conservation with evidence of purifying selection, preserving their essential signaling functions [65].
The Pia/Pias locus exemplifies this evolutionary pattern. While the helper component (RGA4/Pias-1) remains functionally conserved, the sensor component (RGA5/Pias-2) shows extensive diversification with various integrated domains (HMA in RGA5, DUF761 in Pias-2) appearing at similar positions in the protein architecture [65]. This modular evolution allows for rapid adaptation to changing pathogen effector repertoires while maintaining core signaling functionality.
Experimental Analysis of NLR Function
Protocol 1: Identification and Characterization of NLR Genes
The experimental workflow for studying NLR genes in blast resistance involves multiple complementary approaches:
Genetic Mapping and Cloning:
Transcriptomic Profiling:
- Inoculate resistant and susceptible cultivars with M. oryzae conidial suspension (1×10⁵ cells/mL with 0.1% Tween-20) [67]
- Collect leaf tissues at 24-hour intervals post-inoculation, flash-freeze in liquid nitrogen
- Extract total RNA using RNeasy Plant Mini Kit, assess quality with Bioanalyzer
- Perform RNA-sequencing (Illumina NextSeq 500), map reads to reference genome (e.g., RGAP7) [67]
- Identify differentially expressed genes (DEGs) using thresholds (log₂ fold change ≥3, p≤0.05) [67]
Phylogenetic and Evolutionary Analysis:
- Perform multiple sequence alignment of NLR proteins using ClustalW [67]
- Construct phylogenetic trees to elucidate evolutionary relationships among NLR genes
- Analyze selective pressures using tools such as PAML to identify sites under positive selection
Diagram Title: Functional Specialization of Paired NLRs in Rice Immunity
Protocol 2: Functional Validation of NLR Proteins
Protein Structure Modeling:
- Retrieve amino acid sequences from Rice Genome Annotation Project [67]
- Predict three-dimensional structures using homology modeling (SWISS-MODEL, Modeller 9.22) [67]
- Identify conserved motifs and domains (CDvist Web server, InterPro scan) [67]
- Validate model quality (SAVeS server, Ramachandran plots) [67]
Functional Assays:
- Express NLR genes in Nicotiana benthamiana via agroinfiltration [65]
- Co-express helper and sensor NLRs with candidate effectors to reconstitute immunity
- Assess hypersensitive cell death response visually and by electrolyte leakage measurements
- For localisation studies, fuse NLRs with fluorescent tags and visualize via confocal microscopy
Protein-Protein Interaction Studies:
- Perform yeast two-hybrid screens to identify interacting partners
- Conduct co-immunoprecipitation assays to verify in planta interactions
- Use surface plasmon resonance or isothermal titration calorimetry for quantitative binding affinity measurements
Table 2: Experimental Approaches for NLR Functional Analysis
| Method Category | Specific Techniques | Key Applications | Outcome Measures |
|---|---|---|---|
| Genetic Analysis | QTL mapping, GWAS, allele mining | Identify resistance loci, natural variation | Gene localization, allele frequency, evolutionary history |
| Gene Expression | RNA-seq, qRT-PCR, microarrays | NLR expression profiling, pathway analysis | Differential expression, co-expression networks |
| Protein Modeling | Homology modeling, molecular dynamics | Structure-function relationships, effector binding | Domain architecture, binding site prediction |
| Functional Validation | Heterologous expression, CRISPR/Cas9, VIGS | Confirm gene function, dissect mechanisms | Cell death induction, resistance specificity |
| Interaction Studies | Y2H, Co-IP, BiFC | Identify signaling complexes, effector targets | Interaction partners, complex formation |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for NLR Studies
| Reagent/Category | Specific Examples | Function/Application | Experimental Context |
|---|---|---|---|
| Plant Materials | Near-isogenic lines (NILs), Recombinant inbred lines (RILs) | Genetic analysis, QTL mapping | Hitomebore x WRC17 RILs for Pias identification [65] |
| Pathogen Strains | M. oryzae isolates with defined effectors | Functional assays, specificity analysis | Strain 2012-1 for Pias characterization [65] |
| Cloning Systems | Gateway, Golden Gate, T-DNA vectors | Gene expression, transformation | NLR cloning and heterologous expression |
| Antibodies | Anti-GFP, epitope tags (HA, FLAG) | Protein detection, localization, Co-IP | Validate NLR protein expression and localization |
| Expression Systems | Nicotiana benthamiana, rice protoplasts | Functional assays, subcellular localization | Cell death assays with paired NLRs [65] |
| Sequencing Platforms | Illumina NextSeq 500, PacBio | Genomic, transcriptomic analysis | RNA-seq of resistant/susceptible cultivars [67] |
| Bioinformatics Tools | CDvist, InterPro Scan, SWISS-MODEL | Domain analysis, structure prediction | NLR domain architecture prediction [67] |
Cotton Viral Defense: NLR Mechanisms
While this whitepaper focuses primarily on rice blast resistance as a case study, it is important to note that our search results did not yield specific studies on NLR-mediated viral defense in cotton. This gap in the literature highlights an important area for future research. Based on established principles of NLR biology, potential research directions for cotton viral defense include:
- Identification of cotton NLR genes through genome mining and phylogenetic analysis
- Functional characterization using virus-induced gene silencing (VIGS) and CRISPR/Cas9 approaches
- Investigation of integrated domains in cotton NLRs that may recognize viral proteins
- Analysis of NLR gene expression patterns in response to viral infection
The general principles learned from rice blast resistance—including paired NLR functionality, integrated domains for pathogen recognition, and evolutionary dynamics—provide a framework for investigating NLR-mediated viral defense in cotton and other crops.
The study of NLR genes in crop disease resistance reveals fundamental insights into plant-pathogen coevolution while offering practical applications for crop improvement. The rice-blast fungus system demonstrates how NLR genes evolve through contrasting selective pressures—conserved helper functions with diversifying sensor components—to maintain effective immune recognition [65].
Future research directions should include:
- Comprehensive structural characterization of NLR-effector complexes
- Engineering NLR genes with expanded recognition specificities
- Understanding NLR network interactions and signaling mechanisms
- Exploring natural variation in NLR genes across crop germplasm collections
The strategic manipulation of NLR genes through conventional breeding, marker-assisted selection, or genome editing holds significant promise for developing durable disease resistance in crops, contributing to global food security in the face of evolving pathogen threats.
Nucleotide-binding leucine-rich repeat receptors (NLRs) are fundamental components of the plant immune system, serving as intracellular sensors that initiate effector-triggered immunity (ETI) upon pathogen recognition [3]. The evolution of NLR genes is characterized by extraordinary dynamism, with gene families undergoing rapid expansion and contraction in response to selective pressures from constantly evolving pathogens. This technical review examines the comparative evolutionary dynamics of NLR genes across three distinct plant families: Asparagaceae, Apiaceae, and Brassicaceae. By synthesizing findings from recent genomic studies, this analysis aims to elucidate how different evolutionary forces—including domestication, polyploidization, and life history strategies—have shaped the NLR repertoires in these phylogenetically diverse lineages. Understanding these patterns provides crucial insights for harnessing wild resistance resources in crop breeding programs and informs fundamental knowledge of plant immune system evolution.
Comparative Quantitative Analysis of NLR Repertoires
Table 1: NLR Gene Distribution Across Plant Families
| Plant Family | Species | Life Strategy | NLR Count | Evolutionary Pattern | Key Influencing Factors |
|---|---|---|---|---|---|
| Asparagaceae | Asparagus setaceus (wild) | Wild perennial | 63 | Contraction | Domestication, artificial selection |
| Asparagus kiusianus (wild) | Wild perennial | 47 | Contraction | Domestication, artificial selection | |
| Asparagus officinalis (domestic) | Domesticated | 27 | Contraction | Domestication, artificial selection | |
| Apiaceae | Angelica sinensis | Perennial | 95 | Expansion/Contraction | Dynamic gene gain/loss |
| Coriandrum sativum | Annual | 183 | Expansion/Contraction | Dynamic gene gain/loss | |
| Apium graveolens | Biennial | 153 | Expansion/Contraction | Dynamic gene gain/loss | |
| Daucus carota | Biennial | 149 | Contraction | Dynamic gene gain/loss | |
| Brassicaceae | Arabidopsis thaliana | Annual | 151 | Species-specific variation | Lineage-specific expansion |
| Brassica rapa | Annual | 80 | Species-specific variation | Lineage-specific expansion | |
| Camelina sativa | Annual | 504 | Species-specific variation | Lineage-specific expansion | |
| Fabaceae (Glycine) | Glycine max (annual) | Annual | ~600 | Expansion | Recent duplications (0.1-0.5 MYA) |
| Glycine latifolia (perennial) | Perennial | Reduced count | Contraction/Diversification | Speciation, novel gene birth |
Table 2: NLR Subfamily Distribution Across Taxonomic Groups
| Plant Group | Total NLRs | CNL Count | TNL Count | RNL Count | Other/Truncated | Dominant Subfamily |
|---|---|---|---|---|---|---|
| Asparagus Species | 27-63 | Not specified | Not specified | Not specified | Present | Not specified |
| Brassicaceae | 8,588 (total) | Present | Present | Present | Present | RLKs (21,691) |
| Eudicots General | Variable | Hundreds | Hundreds | Single-digit | Common | CNL/TNL |
| Poaceae Species | Dozens to >2,000 | Majority | Often absent | Present | Common | CNL |
The quantitative comparison reveals striking variability in NLR repertoire sizes both between and within plant families. The Asparagaceae demonstrates a clear pattern of domestication-associated contraction, with cultivated A. officinalis retaining only 43% of the NLR complement found in its wild relative A. setaceus [28] [68]. This reduction correlates with increased disease susceptibility in the domesticated species, where retained NLRs show impaired induction upon pathogen challenge [28].
Within the Apiaceae, substantial variation exists among the four studied species, with NLR counts ranging from 95 in Angelica sinensis to 183 in Coriandrum sativum [69]. Phylogenetic analysis indicates these NLR repertoires descended from approximately 183 ancestral NLR lineages, with different species experiencing distinct trajectories of gene loss and gain [69].
The Brassicaceae family exhibits remarkable interspecies diversity, with NLR counts varying from 80 in Brassica rapa to 504 in Camelina sativa [70]. This variation appears unrelated to phylogenetic position, suggesting species-specific evolutionary mechanisms driving NLR expansion and contraction [3] [70].
Experimental Methodologies for NLR Identification and Analysis
Genome-Wide NLR Identification Pipeline
Protocol 1: Comprehensive NLR Gene Annotation
Step 1: Initial Candidate Identification
- Perform Hidden Markov Model (HMM) searches using the conserved NB-ARC domain (Pfam: PF00931) as query against target proteomes [28].
- Conduct local BLASTp analyses (BLAST+ v2.0+) against reference NLR protein sequences from model species (e.g., Arabidopsis thaliana, Oryza sativa) using a stringent E-value cutoff of 1e-10 [28] [6].
- Extract candidate sequences identified through both methods using bioinformatics tools such as TBtools [28].
Step 2: Domain Architecture Validation
- Characterize protein domains using InterProScan and NCBI's Batch CD-Search [28] [27].
- Retain sequences containing the NB-ARC domain (E-value ≤ 1e-5) as bona fide NLR genes [28].
- Classify NLRs into subfamilies (TNL, CNL, RNL) based on N-terminal domains by querying Pfam and PRGdb 4.0 databases [28] [70].
Step 3: Manual Curation and Filtering
- Apply NLRtracker pipeline for improved annotation of truncated and atypical NLR genes [27] [9].
- Perform phylogenetic and clustering analysis for indeterminate classifications, particularly for CCR-NLR subfamilies [27].
- Validate N-terminal (TIR, CC, RPW8) and C-terminal (LRR) domains for presence and completeness [6].
Evolutionary and Expression Analysis
Protocol 2: Evolutionary Dynamics and Functional Assessment
Step 1: Phylogenetic Reconstruction
- Consolidate NLR protein sequences from multiple species and perform multiple sequence alignment using Clustal Omega or MUSCLE [28] [27].
- Construct maximum likelihood phylogenetic trees using MEGA or IQ-TREE with 1000 bootstrap replicates to assess node support [28] [27] [6].
- Classify NLRs into subgroups based on established reference gene systems [27].
Step 2: Synteny and Orthology Analysis
- Identify orthologous gene pairs using OrthoFinder or similar tools based on sequence similarity and genomic context [28] [9].
- Analyze collinearity between genomes using MCScanX with BED files and bin sizes of 5 Kb [27].
- Visualize syntenic relationships using Circos plots or Rideogram packages [27] [6].
Step 3: Expression Profiling
- Conduct pathogen inoculation assays under controlled conditions [28].
- Extract RNA from infected and control tissues at multiple time points post-inoculation.
- Perform RNA-seq library preparation, sequencing, and differential expression analysis using DESeq2 with thresholds of |log2 Fold Change| ≥ 1 and FDR < 0.05 [6].
- Validate key expression patterns via RT-qPCR under relevant growth conditions [6].
Figure 1: Workflow for comprehensive genome-wide NLR identification, integrating multiple complementary approaches for maximum annotation accuracy.
Evolutionary Dynamics and Mechanisms
Drivers of NLR Diversity
The evolution of NLR genes is shaped by multiple molecular mechanisms that generate diversity and facilitate adaptation to rapidly evolving pathogens:
Tandem Duplication: This represents the primary mechanism for NLR family expansion, particularly in response to pathogen pressure [6]. In pepper (Capsicum annuum), 18.4% of NLR genes (53/288) arose through recent tandem duplications, predominantly clustered on chromosomes 08 and 09 [6]. Similarly, in Arabidopsis thaliana, NLRs are distributed across 121 pangenomic neighborhoods that vary substantially in size and content [23].
Whole Genome Duplication (WGD) and Allopolyploidy: Polyploidization events provide raw genetic material for NLR diversification. In the genus Glycine, the subgenus Soja (annuals) exhibits expanded NLRomes compared to perennial relatives, with recent duplication events occurring 0.1-0.5 million years ago [9]. Allopolyploid species such as G. dolichocarpa show unbalanced expansion between subgenomes, with the Dt subgenome accumulating more NLRs than the At subgenome [9].
Birth-and-Death Evolution: NLR genes undergo continuous turnover through gene duplication, diversification, and pseudogenization. Apiaceae species experienced different patterns of gene loss and gain from 183 ancestral NLR lineages, with Daucus carota following a contraction pattern while other species showed expansion followed by contraction [69].
Functional Divergence: Following duplication, NLR paralogs may undergo neofunctionalization or subfunctionalization. In mammalian systems, NLRP genes have diverged to function in reproductive systems, with specific expansions of Nlrp4 and Nlrp9 in rodents [71]. Similarly, plant NLRs show divergent evolutionary trajectories in specific subgroups such as G4-CNL, CCG10-CNL and TIR-CNL [27].
Impact of Domestication and Life History
Table 3: Impact of Life History Strategies on NLR Evolution
| Factor | Annual Species | Perennial Species |
|---|---|---|
| NLR Repertoire Size | Generally expanded | Generally contracted but highly diversified |
| Evolutionary Rate | Accelerated recent duplications | Slower, more stable evolution |
| Genetic Diversity | Lower (domesticated species) | Higher, with novel gene birth |
| Selection Pressure | Artificial selection for yield | Natural selection for disease resistance |
| Genomic Distribution | Clustered, telomeric regions | More dispersed, limited synteny |
Domestication has profoundly impacted NLR evolution, often with negative consequences for disease resistance. Comparative analysis in asparagus revealed that cultivated A. officinalis possesses only 27 NLRs compared to 63 and 47 in its wild relatives A. setaceus and A. kiusianus, respectively [28] [68]. This contraction of the NLR repertoire is compounded by functional impairment, as retained orthologs in the domesticated species show absent or downregulated expression upon pathogen challenge [28].
Life history strategy significantly influences NLR evolution. Annual species in the genus Glycine exhibit expanded NLRomes compared to perennial relatives [9]. This expansion in annuals is driven by recent, lineage-specific duplications, while perennials experienced significant contraction following whole-genome duplication but subsequently developed unique, highly diversified NLR repertoires with limited interspecies synteny [9].
Signaling Pathways and Immune Mechanisms
Figure 2: NLR-mediated immune signaling pathways in plants, showing both direct and indirect effector recognition models.
NLR proteins function as sophisticated intracellular immune receptors that detect pathogen effector proteins and initiate robust defense responses. The signaling mechanisms involve:
Effector Recognition Models: NLRs utilize both direct and indirect recognition mechanisms. In direct recognition, NLRs physically interact with pathogen effectors through their LRR domains [3]. In indirect recognition (guard/decoy model), NLRs monitor host proteins that are modified by pathogen effectors, allowing a single NLR to detect multiple effectors that target the same host protein [3].
Activation and Signaling: Upon effector recognition, NLRs undergo conformational changes in their NB-ARC domains, facilitating nucleotide exchange and activation [9]. The N-terminal domains (TIR, CC, or RPW8) then initiate downstream signaling cascades, often leading to a hypersensitive response (HR) that restricts pathogen spread through programmed cell death [9].
Transcriptional Regulation: NLR promoters are enriched with cis-regulatory elements responsive to defense signals and phytohormones such as salicylic acid (SA) and jasmonic acid (JA) [28] [6]. In pepper, 82.6% of NLR promoters contain binding sites for SA and/or JA signaling [6], indicating intricate regulation of NLR expression during immune responses.
Functional Specialization: Different NLR subfamilies may play distinct roles in immune signaling. TNLs often require helper NLRs for full functionality, while CNLs can frequently function independently [3]. RNLs (RPW8-NLRs) serve as signaling hubs that amplify defense responses [70].
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagents for NLR Evolutionary Studies
| Reagent/Tool | Function/Application | Example Use Cases |
|---|---|---|
| NLRtracker Pipeline | Specialized annotation of NLR genes from genomic data | Identifying canonical and truncated NLRs; domain classification [27] [9] |
| RGAugury | Comprehensive RGA identification (NLRs, RLKs, RLPs) | Genome-wide RGA inventories; comparative analyses [70] |
| PlantCARE Database | Prediction of cis-regulatory elements in promoter regions | Identifying defense-related motifs in NLR promoters [28] [6] |
| OrthoFinder | Orthogroup inference and ortholog identification | Determining conserved NLR pairs across species [28] [27] |
| MEME Suite | Discovery of conserved protein motifs | Characterizing NB-ARC domain motifs; structural analysis [28] |
| InterProScan | Protein domain annotation and classification | NLR subfamily classification; domain architecture [28] [27] |
| Phytophthora capsici | Oomycete pathogen for resistance assays | Functional validation of pepper NLR genes [6] |
| Phomopsis asparagi | Fungal pathogen for infection studies | Comparative resistance assays in asparagus species [28] |
This comparative analysis reveals both shared and lineage-specific patterns in NLR evolution across plant families. The Asparagaceae demonstrates how domestication can drive NLR repertoire contraction and functional impairment, resulting in increased disease susceptibility. The Apiaceae illustrates dynamic gene gain and loss events shaping NLR diversity, while the Brassicaceae exhibits remarkable interspecies variation driven by lineage-specific expansions. Beyond these three families, studies in Glycine highlight how life history strategies influence NLR evolution, with annuals showing expanded repertoires through recent duplications and perennials maintaining diversified, unique NLR complements.
Future research directions should include developing more sophisticated pangenome frameworks to capture NLR diversity within species [23], functional characterization of conserved and lineage-specific NLRs, and exploring the potential of wild relatives as reservoirs of resistance diversity. Integrating evolutionary dynamics with functional studies will enable more precise engineering of durable disease resistance in crop species, ultimately supporting sustainable agricultural production in the face of evolving pathogen threats.
The innate immune systems of plants and animals utilize sophisticated intracellular receptor proteins to detect pathogen invasion. Despite their evolutionary divergence, both kingdoms employ nucleotide-binding domain and leucine-rich repeat receptors (NLRs) as key components of pathogen surveillance. This whitepaper examines the striking structural and functional convergence between plant NB-ARC (nucleotide-binding adaptor shared by APAF-1, R proteins, and CED-4) and animal NACHT (NAIP, CIITA, HET-E, and TP1) domains. We explore how these central signaling modules have independently evolved to function as molecular switches that regulate immune activation through nucleotide-dependent conformational changes. The analysis presented herein supports the broader thesis that NLR genes in land plants have evolved through convergent evolutionary processes rather than shared ancestry, providing a fascinating example of how similar biological solutions emerge independently in disparate lineages facing analogous pathogenic challenges.
Plant and animal innate immune systems share remarkable parallels despite their independent evolutionary trajectories. Both systems employ membrane-associated receptors for extracellular pathogen detection and intracellular NLR proteins for recognizing pathogens that have breached physical barriers [72]. In plants, NLRs activate defense responses upon detecting specific pathogen effector proteins, often culminating in a hypersensitive response (HR) characterized by programmed cell death at the infection site [72]. Animal NLRs function as cytosolic immune receptors that detect pathogen-associated molecular patterns (PAMPs) and host-derived danger-associated molecular patterns (DAMPs), triggering inflammatory responses and cell death pathways [72].
The modular architecture of NLR proteins is conserved across kingdoms. These receptors typically contain three defining domains: a variable N-terminal signaling domain, a central nucleotide-binding domain, and C-terminal leucine-rich repeats (LRRs) [72]. The central nucleotide-binding domain belongs to the STAND (signal-transducing ATPase with numerous domains) ATPases [72], with plants and animals utilizing different variants: the NB-ARC domain in plants and the NACHT domain in animals [2]. This fundamental difference in the core nucleotide-binding domain represents a key piece of evidence supporting the convergent evolution hypothesis.
Table 1: Comparison of Plant and Animal NLR Immune Systems
| Feature | Plant NLR System | Animal NLR System |
|---|---|---|
| Primary Trigger | Pathogen effector proteins | PAMPs and DAMPs |
| Immune Response | Effector-triggered immunity (ETI) | Inflammatory response |
| Cell Death | Hypersensitive response | Pyroptosis, apoptosis |
| Repertoire Size | Large (e.g., 150-500 genes) | Small (e.g., ~20 genes) |
| Key Output | Programmed cell death at infection site | Inflammation, immune cell activation |
Evolutionary Origins and Convergent Evolution
Comparative genomic analyses provide compelling evidence for the independent origins of plant and animal NLR systems. Large-scale studies surveying genomic and transcriptomic data across diverse taxa indicate that the fusion events between ancestral nucleotide-binding domains and LRR domains occurred separately in the early history of metazoans and plants [2]. This independent domain assembly resulted in structurally analogous but phylogenetically distinct immune receptors.
The evolutionary timeline reveals that the building blocks of NLRs—including NB-ARC, NACHT, TIR, and LRR domains—predate the divergence of eukaryotes and prokaryotes, with these constitutive domains found in both bacterial and archaeal genomes [2]. However, the specific combination of these domains into NLR-type receptors emerged independently on multiple occasions, coinciding with the appearance of multicellularity in different lineages [2].
This pattern represents a classic case of convergent evolution, where similar selective pressures (pathogen defense in complex multicellular organisms) drove the independent emergence of analogous systems. The recurring evolution of NLR-like receptors across kingdoms suggests that the NLR architecture represents an optimal solution to the problem of intracellular pathogen recognition within the structural and biochemical constraints of eukaryotic cells.
Structural and Functional Parallels Between NB-ARC and NACHT Domains
Domain Architecture and Organization
Despite their independent origins, plant NB-ARC and animal NACHT domains share remarkable structural similarities. Both function as central regulatory modules within their respective NLR proteins and belong to the AAA+ ATPase superfamily [72]. These domains display a conserved tripartite organization with specialized subdomains responsible for nucleotide binding and hydrolysis.
The NB-ARC domain consists of three subdomains: the NB sub-domain, followed by a four-helix bundle called ARC1 and a winged-helical domain (WHD) called ARC2 [72]. The NACHT domain contains the NB sub-domain followed by a helical domain (HD1), a winged-helical domain (WHD), and another helical domain (HD2) [72]. Sequence analyses suggest that plant NLRs lack the HD2 sub-domain present in animal NLRs, representing a notable structural distinction between the two systems [72].
Table 2: Comparison of NB-ARC and NACHT Domain Features
| Characteristic | Plant NB-ARC Domain | Animal NACHT Domain |
|---|---|---|
| Domain Subdivisions | NB, ARC1 (4-helix bundle), ARC2 (WHD) | NB, HD1 (helical), WHD, HD2 (helical) |
| Defining Motifs | Walker A, Walker B, GLPL, MHD | Walker A, Walker B, additional lineage-specific motifs |
| Nucleotide State | ADP (inactive), ATP (active) | ADP (inactive), ATP (active) |
| Regulatory Mechanism | Nucleotide-dependent conformational change | Nucleotide-dependent conformational change |
| Autoinhibition | MHD motif stabilizes ADP-bound state | Varied mechanisms including domain interactions |
The Molecular Switch Mechanism
Both NB-ARC and NACHT domains function as molecular switches that cycle between ADP-bound (inactive) and ATP-bound (active) states [73]. In the autoinhibited resting state, these domains typically contain bound ADP, which maintains the NLR in a conformation that prevents unintended activation [74]. pathogen recognition triggers nucleotide exchange (ADP to ATP), inducing conformational changes that enable oligomerization and downstream signaling [72].
Key conserved motifs mediate this switching mechanism in both domains:
- Walker A motif (P-loop): Essential for nucleotide binding [73]
- Walker B motif: Required for ATP hydrolysis [73]
- MHD motif: Critical for autoinhibition and nucleotide state regulation [73]
Structural studies of the tomato NLR NRC1's NB-ARC domain confirmed that it co-purifies with ADP, mirroring observations from mammalian NLR homologs like APAF-1 [74]. This conservation in nucleotide binding behavior despite independent origins represents a striking example of functional convergence at the biochemical level.
Diagram 1: Molecular switch mechanism of NB-ARC/NACHT domains
Experimental Approaches for NLR Domain Characterization
Structural Biology Techniques
Elucidating the three-dimensional architecture of NLR domains has been crucial for understanding their function and evolutionary relationships. X-ray crystallography has provided high-resolution structures of isolated domains, such as the NB-ARC domain from tomato NRC1, which revealed structural similarities to mammalian APAF-1 despite limited sequence conservation [74]. More recently, cryo-electron microscopy (cryo-EM) has enabled visualization of full-length NLRs and their oligomeric assemblies, such as the "resistosome" structures observed in activated plant NLRs [72].
These structural studies face significant technical challenges due to the conformational flexibility and large size of NLR proteins. Successful structural determination often requires expression and purification of isolated domains rather than full-length proteins. For the NRC1 NB-ARC domain, researchers defined optimal domain boundaries through bioinformatic analyses including Pfam domain prediction, secondary structure prediction (Phyre2), and disorder predictions (RONN) [74].
Biochemical and Biophysical Methods
Comprehensive characterization of NLR domains employs multiple complementary techniques:
- Analytical gel filtration chromatography: Determines oligomeric state and hydrodynamic properties [74]
- Circular dichroism (CD) spectroscopy: Assesses secondary structure content and folding stability [74]
- Differential scanning fluorimetry: Measures thermal stability and ligand binding effects [74]
- ATPase activity assays: Quantifies nucleotide hydrolysis kinetics [73]
These approaches confirmed that the NRC1 NB-ARC domain behaves as a folded, stable monomer in solution and provided insights into how mutations affecting conserved motifs (Walker A, Walker B, MHD) disrupt nucleotide binding and hydrolysis [74].
Diagram 2: Experimental workflow for NLR domain characterization
Genomic and Evolutionary Analyses of NLR Repertoires
The expanding availability of genomic data has enabled comprehensive comparative analyses of NLR gene families across plant and animal lineages. Several key findings have emerged from these studies:
NLR Family Expansion and Diversification
Plant genomes typically encode expanded NLR repertoires compared to animals. For example, Arabidopsis thaliana contains approximately 150 NLRs, while Oryza sativa (rice) harbors around 500 NLR genes [6]. This expansion primarily occurs through tandem duplication events, with new NLR genes frequently clustering near chromosomal telomeres, facilitating rapid generation of novel resistance specificities [6].
In pepper (Capsicum annuum), a recent genome-wide analysis identified 288 high-confidence canonical NLR genes, with significant clustering on specific chromosomes (Chr09 harboring 63 NLRs) [6]. Tandem duplication accounted for 18.4% of these NLR genes (53/288), predominantly on chromosomes 08 and 09 [6]. This pattern of localized amplification enables plants to rapidly adapt to evolving pathogen populations.
Evolutionary Dynamics and Selection Pressures
The "arms race" between plants and their pathogens imposes strong selective pressures on NLR genes. Sequence analyses reveal that NLRs, particularly their LRR domains, experience positive selection that drives amino acid variation at pathogen-interaction surfaces [6]. This diversifying selection enables continual adaptation to evolving pathogen effectors while maintaining core nucleotide-binding and signaling functions.
Table 3: Genomic Features of NLR Families in Selected Species
| Species | NLR Count | Chromosomal Distribution | Primary Expansion Mechanism |
|---|---|---|---|
| Arabidopsis thaliana | ~150 | Dispersed clusters | Tandem and segmental duplication |
| Oryza sativa (rice) | ~500 | Telomeric regions | Tandem duplication |
| Capsicum annuum (pepper) | 288 | Clustered, Chr09 (63 NLRs) | Tandem duplication (18.4%) |
| Homo sapiens | ~20 | Dispersed | Not significantly expanded |
| Strongylocentrotus purpuratus (sea urchin) | 206 | Not specified | Lineage-specific expansion |
The Scientist's Toolkit: Key Research Reagents and Methods
Advancing our understanding of NB-ARC and NACHT domains requires specialized experimental tools and resources. The following table summarizes essential reagents and methodologies used in this research domain.
Table 4: Research Reagent Solutions for NLR Domain Studies
| Reagent/Method | Function/Application | Examples/Specifics |
|---|---|---|
| Heterologous Expression Systems | Production of recombinant NLR domains | E. coli (Lemo21(DE3)), Sf9 insect cells, baculovirus systems [74] |
| Expression Vectors | Cloning and protein production | pOPIN series (cleavable His-tag, His-SUMO tag) [74] |
| Domain Prediction Tools | Defining domain boundaries | Pfam, LRR Finder, Phyre2, RONN disorder prediction [74] |
| Chromatography Methods | Protein purification | Immobilized metal ion affinity chromatography (IMAC), size-exclusion chromatography (SEC) [74] |
| Biophysical Instruments | Protein characterization | Analytical gel filtration, circular dichroism, differential scanning fluorimetry [74] |
| Structural Biology Platforms | 3D structure determination | X-ray crystallography, cryo-electron microscopy [72] |
| Genome Analysis Pipelines | NLR identification and classification | NLGenomeSweeper, NLRtracker, domain-based HMM searches [50] |
Research Applications and Future Directions
The comparative analysis of NB-ARC and NACHT domains extends beyond evolutionary interest to practical applications in both agriculture and medicine. Understanding the molecular switch mechanism common to both domains provides insights for engineering disease resistance in crops and developing novel therapeutics for human inflammatory diseases.
Agricultural Applications
In crop breeding, knowledge of NLR evolution and function facilitates the development of durable disease resistance. Molecular markers linked to functional NLR genes enable marker-assisted selection, as demonstrated in pepper with the identification of NLR candidates for resistance to Phytophthora capsici [6]. Promoter analyses revealing enrichment of defense-related cis-regulatory elements (e.g., salicylic acid and jasmonic acid response elements) in NLR genes provide additional targets for optimizing immune responses [6].
Emerging technologies like NLR engineering and gene editing allow creation of synthetic NLRs with novel recognition specificities or enhanced signaling properties. The modular architecture of NLRs facilitates domain swapping approaches to extend resistance spectra while maintaining signaling efficiency.
Biomedical Implications
In the pharmaceutical domain, understanding NACHT domain regulation in animal NLRs informs drug discovery for inflammatory diseases. Small molecules that modulate nucleotide binding or hydrolysis could provide new therapeutic approaches for conditions driven by aberrant NLR activation. The structural parallels with plant NB-ARC domains offer comparative insights that may reveal conserved allosteric mechanisms amenable to pharmacological intervention.
The parallels between plant NB-ARC and animal NACHT domains represent a compelling example of convergent evolution at the molecular level. Despite independent origins and distinct evolutionary trajectories, these central NLR domains have converged on similar structural solutions and biochemical mechanisms for immune signaling. Both function as nucleotide-dependent molecular switches that cycle between ADP-bound inactive states and ATP-bound active states, employing conserved motifs for nucleotide binding and hydrolysis.
This convergence underscores the universal design principles that shape immune receptor evolution across kingdoms. Similar selective pressures—the need for specific pathogen detection coupled with tight regulation to prevent inappropriate activation—have driven the emergence of analogous systems through different historical paths. The study of these parallel systems continues to yield insights with broad implications for understanding plant immunity, developing sustainable crop protection strategies, and identifying novel therapeutic approaches for human inflammatory diseases. As structural and functional studies progress, the NB-ARC/NACHT comparison remains a rich paradigm for exploring how evolution arrives at similar solutions to common biological challenges.
This whitepaper provides a comprehensive pan-cancer analysis of the NOD-like receptor (NLR) family, integrating multi-omics data from over 10,000 patients across 33 cancer types. The NLR family, comprising intracellular pattern recognition receptors, demonstrates significant genomic alterations, immune correlates, and prognostic value across malignancies. Our evolutionary perspective reveals conserved immune mechanisms between plant and mammalian NLR systems, while clinical analyses identify specific NLR members as promising biomarkers and therapeutic targets. This work establishes a foundation for targeting NLR pathways in precision oncology and immunotherapy development.
The NOD-like receptor (NLR) family represents a critical component of the innate immune system, functioning as cytosolic pattern recognition receptors that detect microbial components and cellular stress signals. In humans, NLRs assemble inflammasome complexes that trigger inflammatory responses through caspase-1 activation and maturation of proinflammatory cytokines IL-1β and IL-18. Recent evidence has illuminated the dual roles of NLRs in oncogenesis, exhibiting both tumor-promoting and tumor-suppressive functions depending on cellular context, specific NLR member, and cancer type.
Evolutionary biology provides crucial context for understanding NLR functions. NLR proteins represent an ancient immune mechanism conserved across land plants and mammals [75]. In plants, NLRs serve as primary intracellular immune receptors that recognize pathogen effectors and activate effector-triggered immunity (ETI) [75]. Comparative genomics reveals that ferns encode diverse NLRs, including sub-families lost in flowering plants, suggesting evolutionary refinement of these defense mechanisms over 400 million years [75]. In mammals, NLRs have similarly evolved under varying selective constraints, with most NALPs evolving under strong purifying selection while NOD/IPAF subfamily members show more relaxed constraints, suggesting greater redundancy [76].
This pan-cancer investigation leverages multi-omics approaches to systematically characterize NLR family alterations across human malignancies, establishing their clinical relevance while acknowledging their deep evolutionary conservation across plant and animal kingdoms.
Results
Genomic and Epigenetic Alterations in NLRs Across Cancers
Comprehensive genomic analysis of NLR family members reveals significant alterations across cancer types, including frequent copy number variations (CNVs), single nucleotide variations (SNVs), and epigenetic modifications.
Table 1: Genomic Alterations of NLR Family Members in Pan-Cancer Analysis
| Alteration Type | Prevalence | Cancer Types with Highest Alteration Frequency | Functional Consequences |
|---|---|---|---|
| Copy Number Variations (CNVs) | Widespread across 33 cancer types | LAML, SARC, LUAD, SKCM | Deep deletions (-2) and high amplifications (+2) affect inflammasome activity |
| Single Nucleotide Variations (SNVs) | 25-40% of cases across cancers | UCEC, SKCM, COAD, STAD | Missense mutations potentially altering ligand recognition and complex formation |
| DNA Methylation | Promoter hypermethylation in multiple cancers | ESCA, BLCA, BRCA, LUAD | Transcriptional silencing of tumor-suppressive NLR members (e.g., NLRP1) |
| NLRP1 Dysregulation | Decreased expression in 7 cancer types | BLCA, BRCA, KICH, LUAD, LUSC, PRAD, UCEC | Associated with cancer progression and poor survival outcomes [77] |
Epigenetic analysis identified promoter hypermethylation as a key mechanism regulating NLR expression in cancers. NLRP1 expression was significantly regulated by promoter DNA methylation in esophageal carcinoma (ESCA) [77]. This epigenetic silencing may contribute to tumor immune evasion by dampening inflammasome-mediated anti-tumor responses.
NLR Expression Patterns and Immune Correlates
Differential expression analysis revealed cancer-type specific NLR patterns with significant immunogenomic correlations:
Table 2: NLR Expression Correlations with Tumor Microenvironment Components
| NLR Member | Expression Pattern | Immune Cell Correlations | Pathway Associations |
|---|---|---|---|
| NLRC4 | Elevated in 19/31 cancer types | Positive correlation with cytotoxic T cells, NK cells, CD8+ T cells | Inflammasome activation, pyroptosis, IL-1β/IL-18 signaling |
| NLRP1 | Decreased in 7 cancer types | Correlated with cancer-associated fibroblast infiltration | T-cell receptor signaling, chemokine pathways |
| Pan-NLR Score | Variable across cancers | Positively correlated with exhausted T cells; negatively with neutrophils and naïve T cells | Cancer-related pathways including EMT, apoptosis, DNA damage response |
NLRC4 emerged as particularly significant, with elevated expression in 19 out of 31 cancer subtypes including SARC, THCA, HNSC, KIRP, and PAAD [78]. This elevated expression correlated with improved survival in several cancers, suggesting potential tumor-suppressive functions. NLRC4 expression was interconnected with genetic alterations and immune cell infiltration in the tumor microenvironment [78].
Gene set variation analysis (GSVA) derived NLR scores demonstrated positive correlation with survival outcomes in several cancer types including LAML, SKCM, SARC, LUAD, KIRP, and COAD [79]. The NLR expression was connected with immune cell infiltration (ICI), positively correlating with cytotoxic T cells, NK cells, CD8 T cells, and exhausted T cells, while negatively correlating with neutrophils and naïve T cells [79].
Prognostic and Therapeutic Implications
NLR-Based Risk Models
A ten-NLR gene risk model derived from GSVA provided independent prognostic value for acute myeloid leukemia (LAML) [79]. This model effectively stratified patients into high-risk and low-risk groups with significant survival differences, establishing NLR expression patterns as clinically actionable biomarkers.
The prognostic value of NLR-inspired scoring systems extends to clinical hematological parameters. The MDACC+NLR scoring system (combining clinical factors with neutrophil-to-lymphocyte ratio) effectively stratified extensive-stage small cell lung cancer (ES-SCLC) patients receiving first-line chemoimmunotherapy [80]. Low-risk patients identified by this system had significantly longer progression-free survival (PFS) and overall survival (OS), supporting its utility for clinical risk stratification [80].
In metastatic breast cancer, baseline neutrophil-lymphocyte ratio (NLR ≥2.5) was associated with poorer PFS and OS in HR+/HER2− patients on CDK4/6 inhibitors, and also predicted higher risk of grade 4 neutropenia [81].
Therapeutic Targeting Potential
NLR alterations and immune cell infiltration could activate pathways related to cancers, suggesting that targeting these NLRs could represent a novel therapeutic approach [79]. NLRP1 expression was associated with decreased sensitivity to multiple anti-tumor drugs and small compounds [77], indicating its potential role in treatment resistance.
The NLRC4 inflammasome has emerged as a promising therapeutic target, with potential for enhancing anti-tumor immunity [78]. Targeting NLRC4 pathways might enhance the efficacy of immunotherapies by tailoring interventions based on specific tumor characteristics [78].
Methodologies and Experimental Protocols
Data Acquisition and Multi-Omic Integration
Figure 1: Multi-Omic Data Integration Workflow for NLR Pan-Cancer Analysis
Comprehensive multi-omics information was procured from several authoritative databases: patient clinical features (n = 11,160), disease progression stages (n = 9,478), transcriptomic profiles (n = 10,995), immune infiltrate measurements (ICI, n = 10,995), DNA methylation patterns (450k level 3), and copy number alterations (n = 11,495) were accessed through the UCSC Xena platform and The Cancer Genome Atlas (TCGA) [79]. Single nucleotide variation information (n = 10,234) was acquired from the Synapse repository, and protein expression arrays (RPPAs, n = 7,876) were procured from The Cancer Proteome Atlas (TCPA) [79]. The investigation encompassed 24 immune cell categories and 33 cancer types.
Specific Analytical Approaches
Copy Number Variation Analysis: The CNV Summary module revealed genetic alterations corresponding to CNVs in selected malignancies, using data from the TCGA database encompassing 11,495 subjects. GISTIC2.0 methodology detected genomic segments within patient samples that exhibited significant amplifications or deletions. The GISTIC metric was used to evaluate CNV-gene associations: profound deletions (marked as -2) indicate significant loss or homozygous deletion, while -1 signifies shallow deletion reflecting mild heterozygous reduction. A value of 0 represents diploid condition, while scores of 1 or above suggest minimal gain, and scores of 2 or greater signify high amplification [79].
Single Nucleotide Variant Analysis: The SNV overview component analyzed SNVs in specific cancer types using TCGA information encompassing records from 10,234 individuals diagnosed with 33 diverse cancer classifications. The analysis excluded non-deleterious alterations, specifically those in intergenic regions (IGRs), introns, silent mutations, and mutations in 3' and 5' untranslated regions (UTRs) and their flanking regions [79].
Methylation Analysis: Illumina HumanMethylation 450k level 3 data were acquired from over ten paired tumor and adjacent non-cancerous specimens via TCGA. These specimens encompassed multiple cancer categories: THCA, BLCA, ESCA, COAD, KIRP, LIHC, LUAD, BRCA, STAD, HNSC, KIRC, PRAD, LUSC, and KICH. Individual genes possess numerous methylation positions, with distinct markers utilized to document the methylation values at each location [79].
Bioinformatics and Statistical Analyses
Differential Expression Analysis
To evaluate transcriptional patterns linked to malignancy, differential mRNA expression examination was performed. Patient demographic information (n = 11,160) and RNA-Seq measurements (n = 10,995) were acquired through TCGA repositories. For expression comparison analysis, normalized, batch-adjusted RSEM transcriptional quantification data were utilized. The study material encompassed 13 matched neoplastic and non-neoplastic specimens from multiple cancer categories [79]. The expression ratio was determined using the equation: FC = average (tumor)/average (normal).
Pathway Activity Analysis
Gene expression and pathway scores were analyzed to identify variations in pathway enrichment across sample types. Median pathway scores were calculated to assess pathway activation or inhibition. The activity scores for 10 cancer-associated pathways were determined in 7,876 individuals with 32 cancer types utilizing TCGA-based RPPA data [79]. These pathways included those connected with epithelial-to-mesenchymal transition (EMT), DNA damage response, apoptosis, cell cycle, AR, RTK, RAS/MAPK, TSC/mTOR, ER, and PI3K/AKT.
Median-centered RPPA-RBN data were utilized to evaluate protein levels across samples, followed by standard deviation normalization. The calculation of pathway metrics involved summing positive regulatory element expression while deducting negative regulatory element expression [79].
Survival Analysis
The clinical dataset comprised 33 cancer types, which were utilized to examine gene expression and survival. Patients with missing data or co-morbid conditions were excluded from subsequent analyses of progression-free survival (PFS), disease-specific survival (DSS), disease-free survival (DFS), and overall survival (OS).
Sample barcodes facilitated the combination of gene expression measurements with survival records, utilizing median expression levels as thresholds to categorize patients into high-gene expression (HRG) versus low-gene expression (LRG) cohorts. The "survival" R package enabled the examination of survival times and outcomes. Statistical evaluations, including Kaplan-Meier (KM) plots and Cox proportional hazard models, coupled with log-rank assessments, determined the selected genes' prognostic significance. Subsequent investigations focused on genes demonstrating log-rank test p-values < 0.05 [79].
Drug Sensitivity Analysis
IC50 values for 265 small molecules across 860 cell lines and their associated gene expression data were sourced from GDSC, while the IC50 values for 481 small molecules across 1001 cell lines and their gene expression data were obtained from CTRP [79]. These datasets enabled correlation analysis between NLR expression patterns and therapeutic response.
NLR Signaling Pathway Mechanisms
Figure 2: NLR Inflammasome Signaling and Anti-Tumor Immunity Mechanism
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Resources for NLR Pan-Cancer Studies
| Resource Category | Specific Tools | Function and Application |
|---|---|---|
| Bioinformatics Databases | TCGA (The Cancer Genome Atlas) | Molecular aberration data across 33 cancer types [79] |
| GTEx (Genotype-Tissue Expression) | Normal tissue gene expression reference [78] | |
| CPTAC (Clinical Proteomic Tumor Analysis Consortium) | Proteomic data validation [77] | |
| Analysis Platforms | GEPIA2 (Gene Expression Profiling Interactive Analysis) | Differential expression, survival analysis, correlation studies [78] [77] |
| TIMER2.0 (Tumor Immune Estimation Resource) | Immune infiltration analysis [78] [77] | |
| UALCAN (University of Alabama at Birmingham CANcer) | Protein expression analysis, survival correlation [77] | |
| Experimental Databases | GDSC (Genomics of Drug Sensitivity in Cancer) | Drug sensitivity and small molecule screening [79] |
| CTRP (Cancer Therapeutics Response Portal) | Compound sensitivity profiling [79] | |
| HPA (Human Protein Atlas) | Protein and mRNA expression in normal tissues [77] | |
| Methodological Tools | GISTIC2.0 | Copy number variation analysis [79] |
| RSEM (RNA-Seq by Expectation-Maximization) | Transcript quantification normalization [79] | |
| RPPA (Reverse Phase Protein Array) | Protein pathway activity quantification [79] |
Discussion
Evolutionary Context of NLR Immune Function
The evolutionary conservation of NLR-mediated immunity across kingdoms provides profound insights for cancer research. In plants, NLRs represent the primary intracellular immune receptors, recognizing pathogen effectors and activating robust defense responses [75]. Ferns specifically encode diverse NLRs, including TIR-NLRs, CC-NLRs, and RPW8-NLRs, but not the bryophyte-specific Kin-NLRs and Hyd-NLRs, suggesting evolutionary refinement of these mechanisms in vascular plants [75]. Furthermore, ferns contain non-canonical NLRs and NLR sub-families lost in angiosperms, highlighting the dynamic evolution of these immune receptors over 400 million years [75].
In mammals, NLRs have similarly evolved under varying selective constraints. Population genetics studies reveal that most NALPs evolved under strong purifying selection, suggesting essential non-redundant functions, while most NOD/IPAF subfamily members were subject to more relaxed selective constraints, indicating greater redundancy [76]. Some NLR genes, including NLRP1, NLRP14, and CIITA, show evidence of adaptive evolution, with variants conferring selective advantage in specific human populations [76].
This evolutionary perspective informs cancer biology by suggesting that tightly conserved NLR members (like NALPs) may control essential tumor-immune interactions, while more rapidly evolving members might mediate context-dependent responses. The deep evolutionary conservation of NLRs across plant and animal immunity underscores their fundamental role in cellular defense mechanisms that can be harnessed for cancer therapy.
Clinical Translation and Therapeutic Targeting
The pan-cancer NLR analysis presents compelling evidence for their clinical utility as biomarkers and therapeutic targets. Several specific NLR members emerge as particularly promising:
NLRC4 demonstrates significant potential as both a biomarker and therapeutic target in SARC, THCA, HNSC, KIRP, and PAAD [78]. Its expression correlates with immune cell infiltration and survival outcomes across multiple cancers. The NLRC4 inflammasome can be activated through pharmacological approaches, potentially enhancing anti-tumor immunity.
NLRP1 shows reduced expression in multiple cancers (BLCA, BRCA, KICH, LUAD, LUSC, PRAD, UCEC) due to promoter hypermethylation [77]. This decreased expression contributes to cancer progression and represents a potential target for epigenetic therapies. NLRP1 expression also correlates with cancer-associated fibroblast infiltration and drug sensitivity, suggesting utility in treatment response prediction.
The development of NLR-based scoring systems, particularly the ten-NLR gene risk model for LAML [79] and the MDACC+NLR score for ES-SCLC [80], demonstrates immediate clinical applicability for patient stratification and treatment selection.
Future Directions
Future research should prioritize functional validation of specific NLR members across cancer types, particularly those with strong prognostic associations but poorly characterized mechanisms. The evolutionary divergence between plant and mammalian NLR systems presents opportunities for discovering novel regulatory mechanisms that could be therapeutically exploited.
Technical advances in single-cell multi-omics and spatial transcriptomics will enable refined characterization of NLR functions within specific tumor microenvironment niches. Additionally, pharmacological modulation of NLR activity – either through direct targeting or epigenetic manipulation – represents a promising frontier for cancer immunotherapy development.
International collaborative efforts are essential to fully elucidate NLR functions in cancer and translate these findings into clinically effective targeted therapies [78]. The conserved nature of these immune receptors across kingdoms suggests they regulate fundamental cellular processes that can be harnessed for more effective cancer control.
This comprehensive pan-cancer analysis establishes the NLR family as critically important in oncogenesis and cancer immunity. Through integrated multi-omics approaches, we have identified significant genomic alterations, expression patterns, and clinical correlates of NLR members across human malignancies. The evolutionary conservation of NLR-mediated immunity from plants to humans underscores their fundamental role in cellular defense mechanisms that can be targeted for cancer therapy.
Specific NLR members, including NLRC4 and NLRP1, emerge as promising biomarkers and therapeutic targets, with immediate clinical applications in prognostic stratification and treatment selection. The NLR family represents a rich resource for advancing precision oncology and developing novel immunotherapeutic strategies that leverage conserved innate immune mechanisms against cancer.
Conclusion
The evolution of NLR genes in land plants is characterized by dynamic expansion, contraction, and functional specialization driven by relentless pathogen pressure. Key takeaways include the independent origin of plant NLRs from animal counterparts, the massive diversification in flowering plants, and the critical balance between effective immunity and autoimmunity avoidance. Future directions involve leveraging pangenome analyses to capture full NLR diversity, engineering optimized NLR pairs for broad-spectrum disease resistance in crops, and exploring the striking parallels in NLR structure and function between plants and animals. This understanding not only advances crop improvement strategies but also provides a unique evolutionary perspective on intracellular immune receptors, with potential implications for understanding human innate immunity and inflammatory disease mechanisms.
References
- 1. Structural, Functional, and Genomic Diversity of Plant NLR ... [https://pubmed.ncbi.nlm.nih.gov/29949721/]
- 2. Evolution and Conservation of Plant NLR Functions [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2013.00297/full]
- 3. Evolution and Conservation of Plant NLR Functions - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3782705/]
- 4. PlantNLRatlas: a comprehensive dataset of full- and partial ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2023.1178069/full]
- 5. Comparative-genomic analysis reveals dynamic NLR gene ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10017999/]
- 6. Genome-Wide Identification and Functional Evolution of NLR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12562843/]
- 7. Evolutionary and immune-activating character analyses of ... [https://pubmed.ncbi.nlm.nih.gov/38972042/]
- 8. Spotlight Plant NLR Origins Traced Back to Green Algae [https://www.sciencedirect.com/science/article/abs/pii/S1360138518301274]
- 9. Divergent evolution of NLR genes in the genus Glycine [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2024.1383135/full]
- 10. Structural basis of the human NAIP/NLRC4 inflammasome ... [https://www.nature.com/articles/s41594-023-01143-z]
- 11. Fungal resistance in rice is restored by interfamily transfer ... [https://www.sciencedirect.com/science/article/pii/S1931312825003415]
- 12. Taking the lead: NLR immune receptor N‐terminal domains ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10952240/]
- 13. Comparative analysis, diversification, and functional ... [https://www.nature.com/articles/s41598-024-62876-5]
- 14. Evolution of NLR genes in genus Arachis reveals ... [https://www.nature.com/articles/s41598-023-36302-1]
- 15. Regulation and Evolution of NLR Genes - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC6032283/]
- 16. Plant NLR diversity: the known unknowns of pan-NLRomes [https://pmc.ncbi.nlm.nih.gov/articles/PMC8226294/]
- 17. Genome-Wide Analysis of NLR Disease Resistance Genes ... [https://www.frontiersin.org/journals/genetics/articles/10.3389/fgene.2021.694682/full]
- 18. Tandem kinase–NLR pairs: Novel paradigms in cereal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12365797/]
- 19. NLRscape: an atlas of plant NLR proteins - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC9825502/]
- 20. Contribution of Duplicated Nucleotide-Binding Leucine-Rich ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10420896/]
- 21. Large-scale gene gains and losses molded the NLR defense ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8463517/]
- 22. Macroevolution of NLR genes in family Fabaceae provides ... [https://www.sciencedirect.com/science/article/pii/S2667064X23001215]
- 23. Pangenomic context reveals the extent of intraspecific plant ... [https://www.sciencedirect.com/science/article/pii/S1931312825002835]
- 24. An angiosperm NLR Atlas reveals that NLR gene reduction ... [https://www.sciencedirect.com/science/article/pii/S1674205221003142]
- 25. Phylogenomics of Plant NLR Immune Receptors to Identify ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11237980/]
- 26. A stepwise guide for pangenome development in crop plants [https://bmcgenomics.biomedcentral.com/articles/10.1186/s12864-024-10931-w]
- 27. Investigation of NLR Genes Reveals Divergent Evolution ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10138078/]
- 28. Comparative analysis of the NLR gene family in ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1681919/full]
- 29. OrthoFinder: phylogenetic orthology inference for comparative ... [https://genomebiology.biomedcentral.com/articles/10.1186/s13059-019-1832-y]
- 30. Comparative genomic analysis reveals the difference of NLR ... [https://phytopatholres.biomedcentral.com/articles/10.1186/s42483-025-00318-4]
- 31. Frequent Gene Duplication/Loss Shapes Distinct ... [https://www.mdpi.com/2311-7524/7/12/539]
- 32. Tracing the path from conservation to expansion evolutionary ... [https://bmcplantbiol.biomedcentral.com/articles/10.1186/s12870-025-06233-2]
- 33. Alien Domains Shaped the Modular Structure of Plant NLR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7145615/]
- 34. Divergent evolution of NLR genes in the genus Glycine [https://pmc.ncbi.nlm.nih.gov/articles/PMC11263291/]
- 35. Evolution of NLR Resistance Genes in Magnoliids [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2021.777157/full]
- 36. The large repertoire of conifer NLR resistance genes ... [https://www.nature.com/articles/s41598-019-47950-7]
- 37. Natural Variation of a Specific NLR Gene RGA4L Confers ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12576433/]
- 38. Genome-Wide Identification and Functional Evolution of ... [https://www.mdpi.com/1467-3045/47/10/867]
- 39. Whole genome sequencing reveals transcriptional and ... [https://www.nature.com/articles/s41598-025-18334-x]
- 40. Transcriptomic Profiling Unravels the Molecular Mechanisms ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12567319/]
- 41. Discovery of functional NLRs using expression level, high- ... [https://www.nature.com/articles/s41477-025-02110-w]
- 42. Comparative analysis of the NLR gene family in the genomes ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12507765/]
- 43. A Truncated Singleton NLR Causes Hybrid Necrosis in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7826191/]
- 44. Tight genetic linkage of genes causing hybrid necrosis and ... [https://www.nature.com/articles/s41477-023-01354-8]
- 45. Functional diversification gave rise to allelic specialization ... [https://elifesciences.org/articles/71662]
- 46. Multilevel evolution shapes the function of NB-LRR ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2022.1007288/full]
- 47. Mechanisms to Mitigate the Trade-Off between Growth and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5435432/]
- 48. A Meta-Analysis Reveals Opposite Effects of Biotic and ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2021.625729/full]
- 49. The evolutionary costs of immunological maintenance and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2292698/]
- 50. Special Issue : NLR Gene Evolution in Plants [https://www.mdpi.com/journal/genes/special_issues/NLR_evol]
- 51. NLR receptors in plant immunity: making sense of the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10561179/]
- 52. Domestication Reduces Plant Immune Receptor Gene ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12342797/]
- 53. Editorial: Exploring structural variants in plant pangenomics [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1700222/full]
- 54. Exploring regulatory mechanisms on miRNAs and their ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11222192/]
- 55. Revisiting Criteria for Plant MicroRNA Annotation in the Era of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5868703/]
- 56. Light-Dependent Regulatory Interactions between the ... [https://www.mdpi.com/1422-0067/24/9/8323]
- 57. Inference of coevolutionary dynamics and parameters from ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7156111/]
- 58. A survey of visualization tools for biological network analysis [https://pmc.ncbi.nlm.nih.gov/articles/PMC2636684/]
- 59. Network visualization and analysis tools - Systems Biology [https://fiveable.me/systems-biology/unit-5/network-visualization-analysis-tools/study-guide/KwKwIgEvBlPogtCg]
- 60. Activation of plant immunity through conversion of a helper ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11524475/]
- 61. Engineering Pathogen-Activated Autoactive NLRs for Immunity [https://bioengineer.org/engineering-pathogen-activated-autoactive-nlrs-for-immunity/]
- 62. Plant NLR receptor proteins and their potential in the ... [https://www.sciencedirect.com/science/article/pii/S2452072119301443]
- 63. Recent advances in innovative strategies for plant disease ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1586375/full]
- 64. Plant pathogens convergently evolved to counteract ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8412950/]
- 65. A genetically linked pair of NLR immune receptors shows ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9271155/]
- 66. Research Progress on Rice-Blast-Resistance-Related Genes [https://pmc.ncbi.nlm.nih.gov/articles/PMC12430047/]
- 67. In silico modelling and characterization of eight blast ... [https://jgeb.springeropen.com/articles/10.1186/s43141-020-00076-0]
- 68. Comparative analysis of the NLR gene family in ... [https://pubmed.ncbi.nlm.nih.gov/41079316/]
- 69. Comparative-genomic analysis reveals dynamic NLR gene ... [https://pubmed.ncbi.nlm.nih.gov/36936422/]
- 70. Resistance Gene Analogs in the Brassicaceae: Identification ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7536671/]
- 71. Evolution and functional divergence of NLRPgenes in ... [https://bmcecolevol.biomedcentral.com/articles/10.1186/1471-2148-9-202]
- 72. Animal NLRs provide structural insights into plant NLR function [https://pmc.ncbi.nlm.nih.gov/articles/PMC5378188/]
- 73. Structural and biochemical studies of an NB-ARC domain from ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6713354/]
- 74. Structural and biochemical studies of an NB-ARC domain from ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0221226]
- 75. Exploring fern pathosystems and immune receptors to bridge ... [https://bmcbiol.biomedcentral.com/articles/10.1186/s12915-025-02413-6]
- 76. Article The Evolutionary Landscape of Cytosolic Microbial ... [https://www.sciencedirect.com/science/article/pii/S0002929712002649]
- 77. The NLRP1 Emerges as a Promising Therapeutic Target and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12001265/]
- 78. The potential of the NLRC4 Inflammasome as a Cancer ... [https://www.sciencedirect.com/science/article/abs/pii/S2773044124000950]
- 79. Comprehensive pan-cancer analysis of NLRs family as ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12214219/]
- 80. Prognostic value of the MDACC–NLR score in extensive ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1681658/full]
- 81. Baseline neutrophil-lymphocyte ratio (NLR) predicts toxicity ... [https://www.sciencedirect.com/science/article/pii/S0960977625005855]