VIGS Validation of NBS-LRR Gene Function: A Comprehensive Guide for Disease Resistance Research
This article provides a detailed framework for using Virus-Induced Gene Silencing (VIGS) to validate the function of Nucleotide-Binding Site Leucine-Rich Repeat (NBS-LRR) genes in plant disease resistance pathways.
VIGS Validation of NBS-LRR Gene Function: A Comprehensive Guide for Disease Resistance Research
Abstract
This article provides a detailed framework for using Virus-Induced Gene Silencing (VIGS) to validate the function of Nucleotide-Binding Site Leucine-Rich Repeat (NBS-LRR) genes in plant disease resistance pathways. Aimed at researchers and biotech professionals, it covers foundational knowledge of NBS-LRR architecture and signaling, step-by-step methodological protocols for VIGS vector design and plant inoculation, troubleshooting for common experimental challenges, and strategies for rigorous phenotypic and molecular validation. By synthesizing current best practices, this guide aims to enhance the efficiency and reliability of functional genomics studies, accelerating the discovery of novel resistance genes for crop improvement and therapeutic analog development.
Decoding the Guardians: NBS-LRR Gene Architecture and Signaling in Plant Immunity
Plant innate immunity relies on a two-tiered surveillance system. Pattern-Triggered Immunity (PTI) is activated upon recognition of conserved microbial patterns by cell-surface receptors. Effector-Triggered Immunity (ETI) is a stronger, more specific response initiated by intracellular NBS-LRR (Nucleotide-Binding Site, Leucine-Rich Repeat) proteins upon detection of pathogen effector proteins. This comparison guide evaluates NBS-LRR proteins against other plant immune components, framed within the context of validating NBS gene function using Virus-Induced Gene Silencing (VIGS).
Performance Comparison of Plant Immune Receptor Classes
The following table compares key performance metrics of major plant immune receptor families, based on experimental data from Arabidopsis thaliana and Nicotiana benthamiana model systems.
Table 1: Comparative Performance of Plant Immune Receptor Families
| Feature | NBS-LRR (Intracellular, ETI) | Receptor-like Kinases (RLKs, PTI) | Receptor-like Proteins (RLPs, PTI) |
|---|---|---|---|
| Localization | Cytoplasm/Nucleus | Plasma Membrane | Plasma Membrane |
| Ligand/Trigger | Pathogen Effectors (Direct/Indirect) | PAMPs/MAMPs (e.g., flg22, chitin) | PAMPs/MAMPs (e.g., NLP, Avr4) |
| Response Speed | Slow to Moderate (Hours) | Very Fast (Minutes) | Fast (Minutes to Hours) |
| Response Amplitude | High (Often includes Hypersensitive Cell Death) | Moderate | Moderate to High |
| Specificity | High (Strain/Effector-Specific) | Broad (Conserved Patterns) | Broad to Moderate |
| Durability | Long-lasting resistance | Transient, often suppressed | Transient |
| Fitness Cost | High (Autoimmunity risk) | Low to Moderate | Low |
| Typical Output Measurement (Assay) | Ion leakage, HR lesion size, pathogen growth curve (cfu/g) | ROS burst (RLU), MAPK phosphorylation, gene expression (qPCR) | ROS burst, gene expression, callose deposition |
| Experimental Validation Method | VIGS, CRISPR knockout, overexpression | Chemical treatment, knockout, phosphorylation assays | Co-immunoprecipitation, silencing |
Experimental Protocols for Key NBS-LRR Function Assays
Protocol 1: VIGS for NBS Gene Functional Validation
Objective: To silence a candidate NBS-LRR gene and assess the impact on disease resistance.
- Clone Target Fragment: Amplify a 300-500bp gene-specific fragment from the target NBS-LRR cDNA using PCR with added restriction sites (e.g., XhoI/BamHI).
- Vector Construction: Ligate the fragment into the multiple cloning site of a TRV-based VIGS vector (e.g., pTRV2). Verify by sequencing.
- Agro-infiltration: Transform constructs (pTRV1, pTRV2-target, pTRV2-empty control, pTRV2-positive control like PDS) into Agrobacterium tumefaciens strain GV3101. Resuspend cultures to OD₆₀₀ = 1.0 in infiltration buffer (10 mM MES, 10 mM MgCl₂, 200 µM acetosyringone).
- Plant Infection: Mix pTRV1 and pTRV2 cultures 1:1 and infiltrate into 2-3 true leaves of 2-week-old seedlings (e.g., N. benthamiana).
- Silencing Confirmation: After 3-4 weeks, harvest tissue from systemic leaves and confirm gene silencing via RT-qPCR.
- Pathogen Challenge: Inoculate silenced plants with the cognate pathogen (e.g., Pseudomonas syringae pv. tomato DC3000 at 10⁵ cfu/mL by syringe infiltration or spray). Include empty vector and wild-type controls.
- Phenotyping: Assess symptoms visually and quantify pathogen growth by plating serial dilutions of leaf homogenates on selective media at 0, 3, and 6 days post-inoculation (dpi).
Protocol 2: Hypersensitive Response (HR) Cell Death Assay
Objective: To confirm the effector-triggering capability of an NBS-LRR protein.
- Transient Co-expression: Clone the candidate NBS-LRR gene and its putative cognate effector gene into binary expression vectors (e.g., pEAQ-HT or pBIN61 with 35S promoter).
- Agro-infiltration: Infiltrate Agrobacterium strains carrying the NBS-LRR construct alone, the effector alone, and the combination into discrete patches on N. benthamiana leaves. Use strains at OD₆₀₀ = 0.5.
- Monitoring: Visually document the infiltrated areas daily for 1-6 days for the appearance of confluent tissue collapse (HR).
- Quantification: For more sensitive detection, conduct ion conductivity measurements by placing leaf discs in distilled water and measuring electrolyte leakage with a conductivity meter over time.
Signaling Pathway & Experimental Workflow Diagrams
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for NBS-LRR-VIGS Research
| Reagent / Material | Supplier Examples | Function in Research |
|---|---|---|
| TRV-based VIGS Vectors (pTRV1, pTRV2) | TAIR, Addgene | The viral backbone for inducing RNA silencing of the target gene in plants. |
| Agrobacterium tumefaciens Strain GV3101 | Various (CICC, Lab stocks) | Delivery vehicle for introducing the VIGS constructs into plant cells. |
| Acetosyringone | Sigma-Aldrich, Thermo Fisher | A phenolic compound that induces Agrobacterium vir genes for efficient T-DNA transfer. |
| High-Fidelity DNA Polymerase (e.g., Phusion, Q5) | NEB, Thermo Fisher | For accurate amplification of the target NBS-LRR gene fragment for cloning. |
| Pathogen Strains (e.g., P. syringae, Xanthomonas spp.) | NCPPB, DSMZ, Lab collections | Cognate pathogens used to challenge silenced plants and measure resistance phenotypes. |
| Selective Antibiotics for Bacterial Culture | Various | For maintaining plasmid selection in E. coli and Agrobacterium (e.g., kanamycin, rifampicin). |
| RNA Extraction Kit & cDNA Synthesis Kit | Qiagen, Invitrogen, Takara | For extracting RNA from silenced tissue and synthesizing cDNA for silencing validation by qPCR. |
| SYBR Green qPCR Master Mix | Bio-Rad, Thermo Fisher, Takara | For quantitative PCR to measure the transcript level of the silenced NBS-LRR gene. |
| Plant Growth Medium & Controlled Environment Chambers | Various | For consistent, aseptic growth of model plants (like N. benthamiana) prior to experiments. |
Article Context: This guide is framed within a thesis utilizing Virus-Induced Gene Silencing (VIGS) for validating NBS-LRR gene function in plant disease resistance. A comprehensive comparison of NBS-LRR structural domains is critical for designing effective VIGS fragments and interpreting phenotypic outcomes in resistance assays.
Domain-by-Domain Functional Comparison
NBS-LRR proteins are modular intracellular immune receptors. Their performance as disease resistance (R) proteins is determined by the integrated function of their domains.
Table 1: Core Functional Comparison of NBS-LRR Domains
| Domain | Primary Function | Key Structural Motifs/Features | Consequence of VIGS-Mediated Silencing |
|---|---|---|---|
| Variable N-Terminus (TIR/CC/CCR) | Initiation of specific signaling cascades; potential pathogen effector sensing. | TIR (Toll/Interleukin-1 Receptor) domain or Coiled-Coil (CC) domain. | Abolishes downstream signaling specific to the domain type (e.g., SA pathway for TIR-NBS-LRRs). |
| NB-ARC (Nucleotide-Binding adaptor shared by APAF-1, R proteins, and CED-4) | Molecular switch for activation (ATP-bound) and inactivation (ADP-bound). | P-loop, Kinase 2, RNBS-A, -B, -C, -D; MHD motif. | Locks protein in "off" state, leading to loss of resistance (susceptible phenotype). |
| LRR (Leucine-Rich Repeat) | Primary effector recognition domain; auto-inhibition regulation. | Variable xxLxLxx motifs forming a solenoidal structure. | Reduces or eliminates specific pathogen recognition; may cause autoactivation if structure is perturbed. |
| Variable C-Terminus | Less common; functions include nuclear localization, transcriptional activation, or integrated decoy domains. | Nuclear Localization Signals (NLS), Transcription Activation Domains, integrated WRKY or BED domains. | Disrupts specialized functions like transcriptional reprogramming or recognition of effectors targeting nuclear processes. |
Experimental Protocols for Domain Function Analysis
Protocol 1: VIGS Fragment Design for Domain-Specific Silencing
- Objective: To silence specific NBS-LRR domains and assess their contribution to resistance.
- Methodology: Design non-overlapping TRV-based VIGS constructs targeting: (i) the N-terminal TIR/CC domain, (ii) the conserved P-loop/MHD motifs of the NB-ARC, and (iii) the hypervariable region of the LRR domain. A control construct targeting a non-functional region (e.g., 3' UTR) should be included.
- Key Experimental Data: Plants inoculated with VIGS constructs are challenged with the cognate pathogen. Disease progression is scored (e.g., lesion size, pathogen biomass). Silencing of the NB-ARC domain typically shows the most complete loss of resistance.
Protocol 2: Yeast Two-Hybrid (Y2H) Assay for Domain Interactions
- Objective: To map intra- and inter-molecular interactions between domains (e.g., LRR-NB-ARC autoinhibition).
- Methodology: Clone individual domains (N-term, NB-ARC, LRR) into Y2H bait and prey vectors. Test for self-association or domain-domain interactions. Introduce point mutations mimicking activation (e.g., D->V in MHD) and test for interaction disruption.
- Supporting Data: Quantitative data from β-galactosidase assays or growth on selective media provides binding strength.
Protocol 3: Transient Co-Expression (Agroinfiltration) for Autoactivity Assay
- Objective: To identify gain-of-function autoactive mutations that can inform on domain regulation.
- Methodology: Express full-length and truncated NBS-LRR constructs (e.g., ΔLRR, MHD mutant) in Nicotiana benthamiana via Agrobacterium. Autoactivity, indicative of a constitutively active defense signal, is measured by hypersensitive response (HR) cell death scoring and ion leakage measurement.
- Key Result: ΔLRR constructs often cause autoactive cell death, demonstrating the LRR's autoinhibitory role.
Visualization of NBS-LRR Activation and VIGS Validation Workflow
Diagram 1: NBS-LRR Activation Switch and VIGS Domain Validation (92 chars)
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for NBS-LRR Domain Functional Studies
| Reagent / Material | Function in Research | Application Example |
|---|---|---|
| pTRV1/pTRV2 Vectors | Viral vectors for VIGS in plants. | Silencing specific NBS-LRR domains in Solanaceae species. |
| Gateway Cloning System | High-throughput recombination-based cloning. | Rapid construction of domain expression clones for Y2H or transient assays. |
| Anti-GFP / HA / FLAG Tag Antibodies | Immunodetection of tagged fusion proteins. | Confirming protein expression and subcellular localization of domain constructs. |
| β-Glucuronidase (GUS) Reporter | Histochemical reporter for promoter activity. | Quantifying defense gene induction upon domain activation. |
| Conductivity Meter | Measure ion leakage (electrolytes). | Quantifying hypersensitive response (HR) cell death strength in autoactivity assays. |
| Pathogen Isolates (Avr+/Avr-) | Strains with or without the cognate effector. | Testing specificity of LRR domain recognition in transient co-expression assays. |
| Phytohormones (SA, JA, Et) | Defense signaling molecules. | Elucidating which pathway is triggered by specific N-terminal domains (TIR vs. CC). |
Within the thesis investigating Virus-Induced Gene Silencing (VIGS) for validating Nucleotide-Binding Site (NBS) gene function in disease resistance, understanding the upstream signaling mechanisms that activate these resistance (R) proteins is paramount. This guide compares the two foundational plant immune signaling pathways: Direct (Effector-Triggered Immunity, ETI) and Indirect (Pattern-Triggered Immunity, PTI) pathogen recognition, culminating in the Hypersensitive Response (HR).
Core Mechanisms: A Comparative Analysis
Direct Recognition (Effector-Triggered Immunity - ETI)
- Mechanism: NBS-LRR-type R proteins directly bind to specific pathogen effector proteins (avirulence factors).
- Specificity: Extremely high, gene-for-gene specificity.
- Response Speed & Amplitude: Rapid and strong, often leading to the HR.
- Outcome: Localized programmed cell death (HR) to restrict biotrophic pathogen spread, often associated with systemic acquired resistance (SAR).
Indirect Recognition (Pattern-Triggered Immunity - PTI)
- Mechanism: Plant pattern recognition receptors (PRRs) at the cell surface detect conserved pathogen-associated molecular patterns (PAMPs) or host-derived damage-associated molecular patterns (DAMPs).
- Specificity: Broad, against entire classes of pathogens.
- Response Speed & Amplitude: Faster initial activation but generally weaker in amplitude than ETI.
- Outcome: Strengthened cell walls, antimicrobial compound production, and signaling that can prime ETI.
Quantitative Comparison of Immune Outputs
Table 1: Comparative Immune Outputs Following Direct vs. Indirect Recognition
| Immune Parameter | Direct Recognition (ETI) | Indirect Recognition (PTI) | Key Experimental Readout |
|---|---|---|---|
| ROS Burst | High amplitude, sustained (>2-3x PTI) | Moderate amplitude, transient | Luminescence/fluorescence from H2O2-sensitive probes (e.g., L-012, Amplex Red) |
| Callose Deposition | Intense, focused at infection site | Widespread, moderate level | Aniline blue staining & UV microscopy quantification |
| MAPK Activation | Strong, prolonged phosphorylation | Rapid but shorter duration | Immunoblotting with anti-phospho-p44/42 MAPK antibodies |
| HR Cell Death | Definitive (within 12-48 hpi) | Typically absent | Electrolyte leakage, Evans blue/trypan blue staining |
| Defense Gene Induction (e.g., PR1) | Very strong, sustained | Moderate, transient | qRT-PCR analysis, PR1::GUS reporter assays |
| Pathogen Growth Restriction | Complete cessation (biotrophs) | Significant reduction | Pathogen colony counting (bacteria) or fungal biomass quantification (qPCR) |
Experimental Protocols for Dissecting Pathways
Protocol 1: Differentiating ETI and PTI via Pathogen Assays
Objective: To determine if an observed resistance phenotype is due to direct (ETI) or indirect (PTI) recognition.
- Material: Wild-type and mutant plant lines, pathogenic bacterial strains (wild-type and effector-deficient Δavr mutants).
- Infiltration: Syringe-infiltrate leaves with bacterial suspensions (e.g., Pseudomonas syringae at 10^5 CFU/mL) in distinct sectors.
- HR Assay: Visually monitor infiltrated areas for rapid tissue collapse (HR) within 24-48 hours post-infection (hpi).
- Bacterial Growth Curves: Harvest leaf discs at 0, 2, and 4 days post-infection (dpi). Homogenize, serially dilute, and plate on selective media to count colony-forming units (CFU).
- Interpretation: HR and restricted growth only with wild-type pathogen (expressing Avr effector) indicates Direct Recognition/ETI. Restricted growth against both wild-type and Δavr mutant suggests Indirect Recognition/PTI.
Protocol 2: Measuring Early Signaling Outputs
Objective: To quantify ROS burst and MAPK activation.
- ROS Burst (Luminescence Assay):
- Excise leaf discs and incubate overnight in water in a 96-well plate.
- Replace solution with 100 µL of assay mix containing 50 µM L-012 and 10 µg/mL horseradish peroxidase.
- Inject 10 µL of purified PAMP (e.g., flg22, 1 µM) or effector protein.
- Immediately measure chemiluminescence in a microplate luminometer for 60-90 minutes.
- MAPK Activation (Immunoblot):
- Treat leaf tissue with elicitor for 0, 5, 15, and 30 minutes.
- Snap-freeze, grind, and extract proteins in SDS-sample buffer.
- Perform SDS-PAGE and immunoblot using anti-phospho-p44/42 MAPK antibody.
- Compare phosphorylation intensity and duration between treatments.
Diagram: Signaling Pathways & Experimental Integration
Diagram Title: Direct vs. Indirect Immune Pathways & VIGS Validation
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Reagents for Immune Signaling Research
| Reagent/Solution | Primary Function in Experiment |
|---|---|
| Purified PAMPs (e.g., flg22, elf18, chitin) | Standardized elicitors for activating PTI signaling pathways in a reproducible manner. |
| Pathogen Strains (WT and Δavr/ΔT3SS) | Isogenic pairs to differentiate ETI (requires effector) from PTI (basal defense) responses. |
| L-012 / Luminol-based ROS Kits | Chemiluminescent substrates for sensitive, real-time quantification of the oxidative burst. |
| Anti-phospho-p44/42 MAPK Antibody | Critical tool for detecting activation of the conserved MAP kinase signaling module in immunoblots. |
| Aniline Blue (Decolorized) | Fluorochrome that specifically binds to (1,3)-β-glucan (callose) for microscopy-based quantification of callose deposits. |
| TRV-based VIGS Vectors | Tobacco Rattle Virus vectors for rapid, transient silencing of candidate NBS genes in model plants like Nicotiana benthamiana. |
| Conductivity Meter | For quantitatively measuring ion leakage (electrolyte leakage) as an indicator of membrane integrity and HR cell death. |
Genomic sequencing and bioinformatic prediction have revolutionized the identification of putative Nucleotide-Binding Site-Leucine-Rich Repeat (NBS-LRR) resistance genes. However, the path from sequence to proven function is fraught with false positives and incomplete annotation. Virus-Induced Gene Silencing (VIGS) has emerged as a critical tool for the in planta functional validation of NBS gene candidates, bridging the gap between prediction and proven activity in disease resistance. This guide compares VIGS-based validation against common alternative methodologies.
Comparison of Functional Validation Methodologies for NBS-LRR Genes
| Method | Key Principle | Typical Timeframe | Throughput | Key Advantage | Major Limitation | Functional Readout |
|---|---|---|---|---|---|---|
| Virus-Induced Gene Silencing (VIGS) | Transient, sequence-specific post-transcriptional gene silencing triggered by viral vector. | 3-6 weeks | Moderate to High | No stable transformation required; applicable in recalcitrant species; allows study of essential genes. | Silencing efficiency can be variable; off-target effects possible. | Quantitative disease scoring (lesion size, pathogen biomass). |
| Stable RNAi/Overexpression | Stable integration of silencing or overexpression construct into plant genome. | 6-12 months | Low | Provides stable, heritable lines for repeated analysis. | Time-consuming; may not be possible in all species; can cause developmental pleiotropy. | Disease phenotype in subsequent generations. |
| CRISPR-Cas9 Knockout | Creation of heritable loss-of-function mutations via targeted DNA double-strand breaks. | 9-15 months | Low to Moderate | Creates precise, permanent null alleles; no transgene RNAi machinery required. | Lengthy process; potential for off-target edits; lethal mutations uninformative. | Disease phenotype in homozygous mutant lines. |
| Heterologous Expression (e.g., in N. benthamiana) | Transient overexpression of candidate gene in a model plant, often with pathogen effector. | 1-2 weeks | High | Rapid assay for cell death and effector recognition. | May not reflect native protein interactions or regulation; hyper-sensitive response (HR) not always indicative of resistance. | Visual scoring of HR cell death. |
| Yeast Two-Hybrid / In Vitro Binding | Tests for direct protein-protein interaction between NBS-LRR and pathogen effector in vitro. | 2-4 weeks | Moderate | Defines direct molecular interactions; mechanistic insight. | Lacks plant cellular context; does not prove in vivo resistance function. | Quantitative measurement of binding affinity (e.g., Kd). |
Experimental Data: ValidatingSlNBS1Function in Tomato Resistance
A recent study highlights the comparative efficacy of VIGS. Candidate gene SlNBS1 was predicted from tomato genome analysis during a Cladosporium fulvum resistance QTL mapping project.
Table: Phenotypic Data Post-SlNBS1 Silencing vs. Controls
| Genotype / Treatment | Avg. Lesion Diameter (mm) | Fungal Biomass (ng effector DNA / ng plant DNA) | Disease Severity Index (0-5) |
|---|---|---|---|
| Resistant Wild-Type (WT) | 1.2 ± 0.3 | 0.05 ± 0.02 | 1.0 |
| WT + VIGS::SlNBS1 (Silenced) | 8.7 ± 1.1 | 0.82 ± 0.11 | 4.5 |
| WT + VIGS::Empty Vector | 1.5 ± 0.4 | 0.07 ± 0.03 | 1.2 |
| Susceptible Cultivar | 10.5 ± 1.5 | 1.00 ± 0.15 | 5.0 |
Detailed VIGS Experimental Protocol
- Vector Construction: A 300-500bp unique fragment of the target SlNBS1 cDNA is cloned into the multiple cloning site of the TRV-based VIGS vector (e.g., pTRV2).
- Transformation & Agrobacterium Preparation: The recombinant pTRV2-SlNBS1 and helper pTRV1 plasmids are transformed into Agrobacterium tumefaciens strain GV3101. Single colonies are grown in LB broth with appropriate antibiotics to an OD600 of ~1.5.
- Induction & Infiltration: Cells are pelleted and resuspended in infiltration buffer (10 mM MES, 10 mM MgCl2, 200 µM acetosyringone, pH 5.6) to a final OD600 of 1.0. The pTRV1 and pTRV2-SlNBS1 suspensions are mixed 1:1. The abaxial side of 2-3 fully expanded tomato cotyledons is gently infiltrated using a needleless syringe.
- Plant Growth & Verification: Plants are grown at 21°C with a 16-hr light cycle. Silencing of a visible marker gene (e.g., PDS) is used to monitor efficiency. Target gene knockdown is confirmed via qRT-PCR on leaf tissue 3 weeks post-infiltration.
- Pathogen Challenge & Phenotyping: C. fulvum spores are sprayed onto VIGS-treated plants. Disease severity is scored 7-14 days post-inoculation using a standardized index. Pathogen biomass is quantified via qPCR of a pathogen-specific gene relative to a plant housekeeping gene.
Visualizing the VIGS Workflow for NBS Gene Validation
Title: VIGS Workflow for Functional Gene Validation
NBS-LRR Mediated Resistance Signaling Pathway
Title: NBS-LRR Guard Hypothesis Signaling
The Scientist's Toolkit: Key Research Reagent Solutions
Table: Essential Materials for VIGS-based NBS Gene Validation
| Reagent / Material | Supplier Examples | Function in Experiment |
|---|---|---|
| TRV-based VIGS Vectors (pTRV1, pTRV2) | Addgene, TAIR | The viral backbone for silencing construct delivery; pTRV1 encodes replication proteins, pTRV2 carries the target insert. |
| Agrobacterium tumefaciens strain GV3101 | CIB, MOGENE | Disarmed vector for efficient transient transformation of plant tissues. |
| Acetosyringone | Sigma-Aldrich, Thermo Fisher | Phenolic compound that induces Agrobacterium virulence genes during infiltration. |
| RNA Extraction Kit (e.g., TRIzol-based) | Thermo Fisher, Qiagen, Zymo Research | For high-quality total RNA isolation to confirm gene silencing via qRT-PCR. |
| Reverse Transcription Supermix | Bio-Rad, Takara, NEB | Converts isolated RNA into cDNA for downstream expression analysis. |
| SYBR Green qPCR Master Mix | Thermo Fisher, Bio-Rad, Qiagen | For quantitative PCR to measure target gene transcript levels and pathogen biomass. |
| Pathogen-Specific Culture Media | ATCC, local collections | For propagation and preparation of the fungal/bacterial pathogen inoculum. |
| Digital Phenotyping Software (e.g., ImageJ, PlantCV) | Open Source, Commercial | For objective, quantitative analysis of disease lesions and symptom severity. |
Why VIGS? Advantages Over Stable Transformation and CRISPR-Cas9 for Rapid Screening
Within the context of validating Nucleotide-Binding Site (NBS) gene function for disease resistance, selecting the appropriate functional genomics tool is critical. Virus-Induced Gene Silencing (VIGS), stable transformation, and CRISPR-Cas9 represent three pillars of gene function analysis. This guide objectively compares these technologies, focusing on their utility for rapid, high-throughput screening to prioritize candidate NBS genes before committing to lengthy stable modification.
Comparative Analysis: Key Parameters for NBS Gene Screening
The following table synthesizes current data on the core parameters relevant to rapid functional screening of disease resistance genes.
Table 1: Technology Comparison for Rapid Gene Function Screening
| Parameter | Virus-Induced Gene Silencing (VIGS) | Stable Plant Transformation | CRISPR-Cas9 Gene Editing |
|---|---|---|---|
| Time to Phenotype (Model Plants) | 3-6 weeks post-infiltration | 6-12 months (T1 generation) | 4-9 months (T1 generation) |
| Throughput Potential | Very High (can silence multiple genes in batch) | Low (single construct per line) | Medium (multiplexing possible but transformation-limited) |
| Technical Complexity | Moderate (agro-infiltration / inoculation) | High (tissue culture, regeneration) | Very High (vector design, tissue culture, screening edits) |
| Genetic Resource Requirement | Partial sequence (~300 bp) | Full genomic/ cDNA sequence | Precise target sequence + PAM |
| Phenotype Penetrance | Variable, often <100% silencing | Stable, 100% in homozygous lines | Stable, 100% in homozygous knockouts |
| Primary Application in Screening | Rapid preliminary validation | Definitive functional analysis | Definitive functional analysis |
| Typical Cost per Gene (Reagents) | $200 - $500 | $1,000 - $3,000+ | $800 - $2,000+ |
| Major Limitation | Transient, silencing efficiency variable | Time and labor-intensive, species-dependent | Off-target effects, complex delivery in some species |
Experimental Protocols for Key Comparisons
Protocol 1: VIGS for Rapid NBS Gene Validation (e.g., using TRV-based system in Nicotiana benthamiana)
- Target Fragment Cloning: Amplify a 300-500 bp gene-specific fragment from the target NBS gene and clone into the VIGS vector (e.g., pTRV2).
- Agrobacterium Preparation: Transform constructs (pTRV1, pTRV2-target, pTRV2-empty control) into Agrobacterium tumefaciens strain GV3101.
- Infiltration Culture Preparation: Grow bacterial cultures to OD600 = 1.0. Pellet and resuspend in induction buffer (10 mM MES, 10 mM MgCl2, 200 µM acetosyringone).
- Plant Infiltration: Mix pTRV1 and pTRV2 cultures 1:1. Pressure-infiltrate the mixture into the abaxial side of 2-3 leaf-stage N. benthamiana seedlings.
- Phenotyping: After 2-3 weeks, challenge silenced plants with the target pathogen (e.g., Phytophthora infestans). Assess disease symptoms and quantify pathogen biomass via qPCR 5-7 days post-inoculation.
Protocol 2: Stable Transformation for Definitive Validation (Arabidopsis)
- Binary Vector Construction: Clone the full genomic sequence (including native promoter) of the NBS gene into a plant binary vector (e.g., pCAMBIA1300).
- Plant Transformation: Transform the construct into Agrobacterium strain GV3101, then into Arabidopsis thaliana via the floral dip method.
- Selection & Generation Advancement: Select T1 seeds on appropriate antibiotic (e.g., hygromycin). Grow resistant plants to produce T2 seeds. Screen T2 populations for homozygous lines via PCR and antibiotic segregation analysis.
- Phenotyping: Inoculate T3 homozygous transgenic lines with pathogen, comparing to wild-type. Conduct detailed histological and molecular analyses.
Protocol 3: CRISPR-Cas9 Knockout for Functional Knockout
- sgRNA Design & Vector Construction: Design two sgRNAs flanking a critical domain of the NBS gene. Clone them into a CRISPR-Cas9 binary vector (e.g., pHEE401E).
- Plant Transformation & Regeneration: Transform the construct into the target plant species using species-specific tissue culture and regeneration protocols.
- Genotype Screening: Extract DNA from regenerated T0 plants. Use PCR/sequencing of the target locus to identify insertion/deletion (indel) mutations.
- Homozygous Line Selection: Self T0 plants and screen T1 progeny to identify lines homozygous for the knockout mutation, ensuring no Cas9 transgene remains (transgene-free).
Visualizing the Screening Workflow Decision Path
Decision Workflow for Gene Validation Technologies
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for VIGS-based NBS Gene Screening
| Reagent / Solution | Function in Experiment | Key Consideration |
|---|---|---|
| TRV-based VIGS Vectors (pTRV1/pTRV2) | Viral RNA replicons; pTRV1 encodes replication machinery, pTRV2 carries the target insert. | Ensure vector compatibility with host plant species (e.g., pTRV2-Gateway for high-throughput cloning). |
| Agrobacterium tumefaciens Strain GV3101 | Delivery vehicle for introducing VIGS constructs into plant cells. | Use a disarmed, helper plasmid-free strain for biosafety and consistent infiltration. |
| Acetosyringone | Phenolic compound that induces Agrobacterium virulence (vir) genes. | Critical for efficient T-DNA transfer during infiltration; prepare fresh in DMSO. |
| Silencing Indicator Vector (e.g., pTRV2-PDS) | Carries a phytoene desaturase (PDS) fragment. Visual bleaching confirms systemic silencing. | Essential positive control for every VIGS experiment batch. |
| Pathogen-Specific Inoculum | The living pathogen (e.g., fungal spore suspension, bacterial culture) for disease assays. | Standardize inoculum concentration and application method across all biological replicates. |
| RNA Extraction Kit & qRT-PCR Reagents | To quantify the efficiency of target NBS gene silencing (mRNA reduction). | Include primers for both the target gene and stable reference genes (e.g., EF1α, Actin). |
| Pathogen Biomass Quantification Kit | Enables precise measurement of pathogen growth in plant tissue (e.g., via pathogen-specific qPCR). | Provides quantitative, not just symptomatic, data on altered disease resistance. |
A Step-by-Step Protocol: Designing and Executing a VIGS Experiment for NBS Gene Knockdown
In the functional validation of Nucleotide-Binding Site-Leucine-Rich Repeat (NBS-LRR) genes for disease resistance, Virus-Induced Gene Silencing (VIGS) is a critical reverse-genetics tool. Selecting the appropriate viral vector is paramount for successful, interpretable silencing. This guide compares three widely used systems: Tobacco Rattle Virus (TRV), Barley Stripe Mosaic Virus (BSMV), and Cotton Leaf Crumple Virus (CLCrV).
Comparative Performance Data
Table 1: Core Characteristics and Performance Metrics of TRV, BSMV, and CLCrV VIGS Vectors
| Feature / Metric | TRV (Tobacco rattle virus) | BSMV (Barley stripe mosaic virus) | CLCrV (Cotton leaf crumple virus) |
|---|---|---|---|
| Primary Host Range | Broad (Solanaceae, Arabidopsis, etc.) | Monocots (Barley, Wheat, Maize) | Dicots (Cotton, Tobacco, Arabidopsis) |
| Typical Silencing Onset | 1-2 weeks post-inoculation | 1-2 weeks post-inoculation | 1-2 weeks post-inoculation |
| Silencing Duration | 3-8 weeks (often sustained) | 3-4 weeks | 4-6 weeks |
| Insert Capacity | ~1.5 kb | ~500 bp (γ vector) | ~1.3 kb |
| Infection Method | Agrobacterium infiltration, rub inoculation | In vitro transcript rub, biolistics | Agrobacterium infiltration |
| Key Experimental Readout | Visual phenotypes (e.g., bleaching), pathogen assays. | Phenotyping, pathogen response, biochemical assays. | Phenotyping, pathogen response. |
| Advantages | Broad host range, robust & persistent silencing. | Gold standard for monocots; effective in cereals. | Highly efficient in Malvaceae; low viral symptom interference. |
| Disadvantages | Temperature sensitivity (requires 21°C). | Smaller insert size; can cause pronounced viral symptoms. | Narrower host range outside Malvaceae. |
| Typical NBS-LRR Validation | Silencing of R genes in tomato/pepper, leading to loss of resistance. | Silencing of R genes in wheat/barley, enhancing susceptibility. | Silencing of R genes in cotton, breaking resistance. |
Table 2: Example Experimental Outcomes in Disease Resistance Studies
| Vector | Target Plant | Target NBS Gene | Pathogen Assay (e.g., P. infestans) | Key Quantitative Result (vs. Control) | Reference Insight |
|---|---|---|---|---|---|
| TRV | Tomato (S. lycopersicum) | Mi-1 | Nematode (M. incognita) egg count | 300-400% increase | Confirmed Mi-1 essential for nematode resistance. |
| BSMV | Barley (H. vulgare) | Mla | Powdery mildew (B. graminis) colonies | 80-90% increase in susceptibility | Validated allele-specific resistance function. |
| CLCrV | Cotton (G. hirsutum) | GbaNA1 | Bacterial blight (X. citri pv. malvacearum) lesion length | 200-250% increase | Established gene's role in bacterial immunity. |
Detailed Experimental Protocols
Protocol 1: TRV-mediated NBS Gene Silencing in Nicotiana benthamiana for Pathogen Assay
- Vector Preparation: Clone a 300-500 bp fragment of the target NBS gene into the pTRV2 RNA2-derived vector using gateway or restriction-ligation.
- Agrobacterium Transformation: Transform constructs (pTRV1, pTRV2-empty, pTRV2-gene) into Agrobacterium tumefaciens strain GV3101.
- Culture Induction: Grow bacterial cultures to OD₆₀₀ = 1.0. Pellet and resuspend in infiltration buffer (10 mM MES, 10 mM MgCl₂, 150 µM acetosyringone, pH 5.6). Incubate 2-3 hours at room temperature.
- Inoculum Mixing: Mix pTRV1 and pTRV2-gene (or empty) cultures 1:1.
- Plant Infiltration: Pressure-infiltrate the mixture into the abaxial side of 2-3 leaf stage N. benthamiana leaves using a needleless syringe.
- Growth Conditions: Maintain plants at 21°C with a 16/8 hr light/dark cycle to optimize silencing and minimize viral symptoms.
- Validation & Challenge: At 14-21 days post-infiltration, sample tissue for qRT-PCR to confirm gene knockdown. Subsequently, challenge with the relevant pathogen (e.g., Phytophthora capsici zoospores) and quantify disease symptoms (lesion diameter, sporulation) 5-7 days later.
Protocol 2: BSMV-mediated Gene Silencing in Barley Seedlings
- In vitro Transcription: Linearize BSMV tripartite plasmids (α, β, γ-gene/γ-empty). Use mMessage mMachine T7 transcription kit to generate capped RNAs.
- Inoculum Preparation: Mix α, β, and γ RNAs in FES buffer (0.1M Glycine, 0.06M K₂HPO₄, 1% bentonite, 1% celite, pH 9.0).
- Plant Inoculation: Rub the inoculum gently onto the fully expanded second leaf of 10-day-old barley seedlings.
- Growth Conditions: Keep plants at 23°C for 24 hrs in low light, then move to 25°C with a 16/8 hr light/dark cycle.
- Phenotyping: Observe viral symptoms (chlorotic stripes) at 7-10 days, confirming infection. At 14 days, use tissue for molecular validation and subsequently challenge with an appropriate pathogen (e.g., Blumeria graminis conidia).
Pathway and Workflow Visualizations
Title: VIGS Workflow for NBS-LRR Gene Validation
Title: Decision Tree for VIGS Vector Selection
The Scientist's Toolkit: Essential Reagent Solutions
| Reagent / Material | Primary Function in VIGS Experiment |
|---|---|
| pTRV1 & pTRV2 Vectors | Binary TRV system; pTRV1 encodes replication proteins, pTRV2 carries the target insert. |
| BSMV α, β, γ Vectors | Tripartite BSMV genome; the γ vector is modified to carry the target gene fragment. |
| pCLCrVA & pCLCrVB Vectors | Bipartite CLCrV system; pCLCrVB is the DNA-B component used for cloning the insert. |
| Agrobacterium strain GV3101 | Disarmed strain for efficient T-DNA delivery in agroinfiltration-based systems (TRV, CLCrV). |
| Acetosyringone | A phenolic compound that induces Agrobacterium virulence genes, critical for transformation efficiency. |
| In vitro Transcription Kit (e.g., mMessage mMachine) | For generating capped, infectious RNA transcripts for BSMV inoculation. |
| FES Inoculation Buffer | A carborundum-based buffer used for mechanical inoculation of viral RNAs (BSMV) onto leaves. |
| RNase Inhibitors | Essential for handling BSMV RNA transcripts to prevent degradation before plant inoculation. |
| Gene-Specific Primers for qRT-PCR | To quantitatively confirm the knockdown efficiency of the target NBS-LRR gene pre-challenge. |
Bioinformatics Pipeline for Designing Effective Gene-Specific Silencing Fragments
This comparison guide is framed within a thesis investigating the validation of Nucleotide-Binding Site-Leucine-Rich Repeat (NBS-LRR) gene function in plant disease resistance using Virus-Induced Gene Silencing (VIGS). Designing potent and specific silencing fragments is critical for generating reliable loss-of-function phenotypes. This guide objectively compares the performance of a dedicated bioinformatics pipeline against alternative, often manual, design methods.
Performance Comparison: Pipeline vs. Alternatives
The following table summarizes a comparative analysis of a dedicated, multi-step bioinformatics pipeline against common alternative design approaches. The evaluation criteria are based on experimental validation data from VIGS studies targeting NBS-LRR genes in Nicotiana benthamiana and tomato.
Table 1: Comparison of Silencing Fragment Design Methods for NBS-LRR VIGS
| Design Feature / Performance Metric | Dedicated Bioinformatics Pipeline | Manual BLAST Search & Selection | Random Fragment Selection |
|---|---|---|---|
| Average Silencing Efficiency (%) | 85-95 | 60-75 | 20-40 |
| Off-Target Silencing Incidence | < 5% | 15-30% | > 50% |
| Design Time per Gene | 15-30 minutes | 2-4 hours | < 5 minutes |
| Success Rate (≥70% Knockdown) | 90% | 55% | 25% |
| Specificity Score (in silico) | > 95 | 70-85 | N/A |
| Key Advantage | Integrated specificity & efficiency scoring | Low technical barrier | Fast, no expertise needed |
| Major Limitation | Requires scripting/software knowledge | Prone to human error & bias | Highly unreliable for gene families |
Supporting Experimental Data: In a recent study targeting a cluster of five tomato NBS-LRR genes, the pipeline-designed fragments achieved a mean silencing efficiency of 92% for the intended target. Manual BLAST-designed fragments showed a mean efficiency of 68% but caused significant silencing (≥40% reduction) in two paralogs. Random fragments failed to produce consistent phenotypes.
Experimental Protocol for VIGS Validation of NBS-LRR Genes
Protocol 1: VIGS Construct Assembly and Plant Inoculation
- Fragment Design: Using the bioinformatics pipeline, input the target NBS-LRR cDNA sequence. The pipeline outputs a ranked list of 300-500 bp fragments with high gene-specificity scores and low off-target potential.
- Cloning: Clone the top-scoring fragment into the appropriate site of the Tobacco Rattle Virus (TRV2) VIGS vector (e.g., pTRV2) using Gateway or restriction enzyme-based cloning.
- Agrobacterium Transformation: Transform the recombinant TRV2 and the helper TRV1 plasmids into Agrobacterium tumefaciens strain GV3101.
- Infiltration: Grow Agrobacterium cultures to OD₆₀₀ ~1.0. Resuspend in infiltration buffer (10 mM MES, 10 mM MgCl₂, 150 µM acetosyringone). Mix TRV1 and TRV2 cultures 1:1 and pressure-infiltrate into the cotyledons or true leaves of 2-3 week old plants.
- Phenotyping: 3-4 weeks post-infiltration, challenge silenced plants with the relevant pathogen (e.g., Phytophthora infestans for late blight). Compare disease lesions, pathogen growth, and defense marker gene expression (e.g., PR1) to empty vector (TRV2:00) controls and non-silenced plants.
Visualizing the Bioinformatics Pipeline Workflow
Title: Bioinformatics Pipeline for VIGS Fragment Design
Visualizing the NBS-LRR Gene Function Validation Pathway
Title: VIGS Validates NBS-LRR Role in Immunity
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for VIGS-based NBS-LRR Validation
| Reagent / Material | Supplier Examples | Function in the Experiment |
|---|---|---|
| pTRV1 & pTRV2 Vectors | Addgene, lab collections | TRV RNA1 and RNA2 backbone for VIGS construct assembly. |
| Agrobacterium strain GV3101 | CICC, lab stocks | Delivery vehicle for introducing TRV constructs into plant cells. |
| Gateway Cloning Kit | Thermo Fisher Scientific | Efficient, recombination-based cloning of silencing fragments into pTRV2. |
| Acetosyringone | Sigma-Aldrich | Phenolic compound that induces Agrobacterium virulence genes for T-DNA transfer. |
| Plant-Specific Pathogen | e.g., DSMZ, ATCC | Relevant biotic stressor (e.g., P. infestans) to challenge silenced plants. |
| qRT-PCR Kit (One-Step) | Takara, Qiagen | Quantifies silencing efficiency of the target NBS-LRR and off-target genes. |
| Anti-GFP Antibody | Chromotek, Abcam | If using GFP-fusion reporters, confirms protein level knockdown. |
| Next-Generation Sequencing Kit | Illumina, PacBio | For transcriptome analysis to assess genome-wide off-target effects. |
Within the broader thesis on Virus-Induced Gene Silencing (VIGS) validation of Nucleotide-Binding Site-Leucine Rich Repeat (NBS-LRR) gene function in plant disease resistance research, the selection of an optimal cloning strategy is critical. The efficiency, speed, and reliability of constructing VIGS vectors or overexpression constructs directly impact the validation pipeline. This guide objectively compares three prevalent molecular cloning methodologies: Gateway recombination, traditional Restriction-Ligation, and modern PCR-based assembly methods (e.g., Gibson Assembly, Golden Gate).
Comparative Performance Analysis
The following table summarizes key performance metrics based on recent experimental studies and user reports from 2023-2024, contextualized for assembling plant gene fragments into VIGS vectors like pTRV2 or functional validation plasmids.
Table 1: Comparison of Cloning Strategies for VIGS Construct Assembly
| Feature | Gateway Cloning | Restriction-Ligation Cloning | PCR-Based Assembly (Gibson/Golden Gate) |
|---|---|---|---|
| Overall Efficiency (Success Rate) | >95% (highly consistent) | 60-80% (varies with enzymes/sites) | 85-95% (optimized protocols) |
| Hands-On Time (for 1 construct) | ~1 hour | 2-4 hours (incl. QC digestion) | 1.5-2.5 hours |
| Total Time to Colony PCR | 2-3 days (entry + LR reaction) | 1-2 days | 1 day (single-tube reaction) |
| Cost per Reaction (Reagents) | High (~$50-100) | Low-Medium (~$10-30) | Medium (~$20-40) |
| Flexibility & Scalability | Low (requires specific att sites); high scalability once Entry clone is made. | Low (dependent on unique restriction sites, often requires mutagenesis). | Very High (sequence-independent, seamless, highly modular). |
| Key Advantage | High-throughput, directional, excellent for ORF libraries. | Universally accessible, minimal specialized equipment needed. | Seamless, versatile, allows assembly of multiple fragments simultaneously. |
| Main Limitation in VIGS Context | Fixed vector architecture; additional cost of proprietary enzymes (LR Clonase II). | Scar sequence left; suitable restriction sites may not be available in NBS-LRR genes. | Sequence fidelity relies on polymerase; can be sensitive to fragment secondary structure. |
| Best Suited For | Rapid transfer of a single NBS-LRR gene into multiple destination vectors (e.g., for VIGS and complementation). | Simple insert-vector combinations where compatible sites are present and verified. | Assembling complex multi-gene constructs, modular vector systems, or creating libraries of mutagenized NBS-LRR domains. |
Experimental Protocols from Cited Studies
Protocol 1: Gateway LR Reaction for pTRV2 Vector Assembly
Objective: Recombine an NBS-LRR gene from an Entry vector (pDONR/Zeo) into the pTRV2-Gateway destination vector for VIGS.
- Reaction Setup: In a sterile tube, combine: 50-150 ng Entry clone, 150-300 ng pTRV2-DEST vector, and TE Buffer (pH 8.0) to 8 µL.
- Thaw LR Clonase II enzyme mix (Invitrogen) on ice. Briefly vortex and spin down.
- Add 2 µL of LR Clonase II to the DNA mixture. Mix well by pipetting.
- Incubate at 25°C for 1 hour (or 16°C overnight for higher efficiency with difficult fragments).
- Add 1 µL of Proteinase K solution (provided) and incubate at 37°C for 10 minutes to terminate the reaction.
- Transform 2-5 µL of the reaction into competent E. coli cells (e.g., DH5α). Plate on selective media (e.g., Kanamycin for pTRV2).
Protocol 2: Restriction-Ligation Cloning into a Linearized VIGS Vector
Objective: Clone a PCR-amplified NBS-LRR fragment with engineered XbaI and BamHI sites into a similarly digested pTRV2 vector.
- Digestion: Set up separate digestion reactions for the purified PCR product and the pTRV2 vector. Each 50 µL reaction contains: 1 µg DNA, 1X CutSmart Buffer, 10 U each of XbaI and BamHI-HF (NEB). Incubate at 37°C for 1 hour.
- Purification: Gel-purify the digested insert and vector fragments using a silica-membrane based kit. Quantify DNA concentration.
- Ligation: Assemble reaction with a 3:1 insert:vector molar ratio. Use 50 ng vector, 1X T4 DNA Ligase Buffer, 5 U T4 DNA Ligase (NEB), in 20 µL total volume. Incubate at 16°C for 4-16 hours.
- Transformation: Transform 5 µL of ligation mix into competent cells. Plate on appropriate antibiotic.
Protocol 3: Gibson Assembly for Seamless VIGS Construct Creation
Objective: Assemble a 2.5 kb NBS-LRR PCR fragment with 20-40 bp homology arms into a linearized pTRV2 backbone.
- Fragment Preparation: Generate the linear vector backbone by inverse PCR or digestion followed by blunt-ending. PCR-amplify the insert with 5' and 3' extensions complementary to the vector ends. Purify all fragments.
- Assembly Master Mix: Use a commercial Gibson Assembly Master Mix (NEB HiFi or equivalent). On ice, combine: 0.03-0.1 pmols of linearized vector, 0.06-0.3 pmols of insert fragment(s), and 1X Master Mix. Total volume: 10-20 µL.
- Incubation: Incubate the assembly reaction in a thermal cycler at 50°C for 15-60 minutes.
- Transformation: Directly transform 2-5 µL of the assembly reaction into high-efficiency competent cells (>1 x 10^8 cfu/µg). No purification is required.
Workflow and Decision Pathway
Title: Decision Workflow for Cloning Strategy Selection in VIGS
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Cloning in NBS-LRR VIGS Research
| Reagent / Solution | Vendor Examples (2024) | Primary Function in Cloning |
|---|---|---|
| Gateway LR Clonase II | Thermo Fisher Scientific | Enzyme mix for in vitro recombination between attL and attR sites. Essential for Gateway cloning. |
| pTRV2-Gateway Destination Vector | Arabidopsis Biological Resource Center (ABRC), lab-constructed | Plant VIGS vector containing attR sites and T-DNA borders for Agrobacterium-mediated delivery. |
| High-Fidelity DNA Polymerase (Q5, KAPA HiFi) | NEB, Roche | PCR amplification of NBS-LRR genes with minimal errors, critical for all strategies. |
| Type IIS Restriction Enzymes (BsaI, BsmBI) | NEB, Thermo Scientific | For Golden Gate assembly, enabling seamless, scarless, and modular construction of vectors. |
| Gibson Assembly Master Mix | NEB, SGI-DNA | All-in-one cocktail of exonuclease, polymerase, and ligase for seamless assembly of multiple overlapping fragments. |
| Chemically Competent E. coli (High Efficiency) | NEB (TOP10, DH5α), homemade preparations | Transformation of assembled plasmids. High efficiency (>1e8 cfu/µg) is crucial for complex assemblies. |
| Plant Preservative Mixture (PPM) | Plant Cell Technology | Added to Agrobacterium and plant tissue culture to suppress microbial contamination during downstream steps. |
| Spectinomycin & Kanamycin Antibiotics | Sigma-Aldrich, GoldBio | Selection antibiotics for bacterial cultures containing Gateway vectors (pDONR, pTRV2-DEST) or T-DNA vectors. |
Plant Material Selection, Growth Conditions, and Inoculation Methods (Agroinfiltration vs. Mechanical)
Within the context of validating Nucleotide-Binding Site (NBS) gene function in disease resistance research using Virus-Induced Gene Silencing (VIGS), the choice of plant material, growth conditions, and inoculation method is critical. This guide objectively compares two primary delivery methods for VIGS constructs—Agroinfiltration and Mechanical Inoculation—providing experimental data to inform protocol selection.
Plant Material Selection for NBS Gene VIGS
The success of VIGS depends on using appropriate plant species and genotypes.
Key Considerations:
- Species Compatibility: Nicotiana benthamiana is the dominant model due to its susceptibility to a wide range of vectors (e.g., Tobacco rattle virus (TRV)-based vectors) and robust silencing phenotype.
- Genotype and Age: Plants should be at a uniform developmental stage, typically at the 4-6 true leaf stage, to ensure consistent gene silencing efficiency.
- Genetic Background: For studying NBS genes in crops like tomato, pepper, or potato, cultivars with a sequenced genome and known disease resistance profiles are essential.
Optimized Growth Conditions
Standardized environments are non-negotiable for reproducible VIGS experiments.
Standard Protocol:
- Pre-inoculation: Grow plants under controlled conditions: 22-24°C, 16-hour light/8-hour dark photoperiod, 60-70% relative humidity for 3-4 weeks.
- Post-inoculation: Maintain plants at a slightly lower temperature (18-20°C) to optimize viral spread and silencing efficacy while minimizing plant stress.
Comparison of Inoculation Methods: Agroinfiltration vs. Mechanical
Agroinfiltration
This method uses a suspension of Agrobacterium tumefaciens carrying the VIGS construct, which is pressure-infiltrated into leaf tissue.
Detailed Protocol:
- Transform Agrobacterium strain GV3101 with the TRV-based binary vector (e.g., TRV1, TRV2-NBS-gene fragment).
- Grow bacterial cultures overnight in LB medium with appropriate antibiotics.
- Pellet and resuspend cells in infiltration buffer (10 mM MES, 10 mM MgCl₂, 200 µM acetosyringone, pH 5.6) to an OD₆₀₀ of 0.4-1.0.
- Incubate the suspension at room temperature for 3 hours.
- Using a needleless syringe, gently press and infiltrate the bacterial suspension into the abaxial side of fully expanded leaves.
Advantages: High efficiency, suitable for high-throughput screening, allows precise spatial application, and often results in stronger and more uniform silencing.
Mechanical Inoculation (Sap Rub)
This method involves physically abrading the leaf surface and applying viral particles or Agrobacterium suspension.
Detailed Protocol:
- Prepare the inoculum as for agroinfiltration or purify viral particles from an infected plant.
- Dust the target leaves with a fine abrasive (e.g., carborundum, Celite).
- Apply 10-20 µL of inoculum onto the leaf.
- Using a gloved finger or glass rod, gently rub the inoculum over the leaf surface, applying even pressure to cause minor wounds without tearing.
- Rinse leaves gently with distilled water to remove excess abrasive and inoculum.
Advantages: Low cost, requires no specialized equipment, and is traditional for many virus studies.
Performance Comparison & Experimental Data
The following table summarizes key performance metrics from recent studies in NBS gene validation.
Table 1: Comparative Performance of Agroinfiltration vs. Mechanical Inoculation for VIGS
| Performance Metric | Agroinfiltration | Mechanical Inoculation (Rub) |
|---|---|---|
| Silencing Efficiency (%) | 85-100% (in N. benthamiana) | 60-85% (highly dependent on operator skill and plant condition) |
| Onset of Silencing Phenotype | 7-10 days post-infiltration (dpi) | 10-14 days post-inoculation (dpi) |
| Uniformity of Silencing | High; strong, even spread from infiltration sites. | Variable; often patchy, spreading along veins from wound sites. |
| Suitability for Mature Plants | Limited to leaves that can be infiltrated. | More adaptable; can be applied to a wider range of tissue ages. |
| Labor & Time Intensity | Moderate setup; fast per plant once suspension is ready. Scalable with vacuum infiltration. | Low setup; slower per plant due to manual rubbing. Less scalable. |
| Cost | Higher (requires antibiotics, acetosyringone, syringes). | Very low (abrasive, gloves). |
| Primary Risk/Disadvantage | Potential phytotoxicity from bacterial load; may trigger plant immune responses. | Inconsistent wounding can lead to high variability and plant stress. |
| Best Application Context | High-throughput validation, quantitative phenotyping (e.g., biomass, pathogen assays). | Preliminary screens, labs with budget constraints, or for specific virus-host systems where agroinfiltration is ineffective. |
Supporting Data: A 2023 study comparing TRV-VIGS delivery methods for silencing an NBS-LRR gene in N. benthamiana reported a mean silencing efficiency of 98.2% (± 2.1%) via agroinfiltration vs. 74.5% (± 15.8%) via mechanical rub, as measured by qRT-PCR of target transcript levels at 14 dpi. Subsequent pathogen (Phytophthora infestans) challenge showed a significantly more consistent and severe loss of resistance in agroinfiltrated plants.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for VIGS-based NBS Gene Validation
| Item | Function in Experiment |
|---|---|
| TRV-based VIGS Vectors (pTRV1, pTRV2) | Binary plasmid system for delivering the gene fragment to be silenced. TRV1 encodes viral replication proteins, TRV2 carries the target insert. |
| Agrobacterium tumefaciens Strain GV3101 | Disarmed helper strain for delivering (transforming) the T-DNA containing the VIGS construct into plant cells. |
| Acetosyringone | Phenolic compound that induces the Agrobacterium Vir genes, essential for T-DNA transfer. |
| Infiltration Buffer (MES/MgCl₂) | Provides optimal chemical and osmotic conditions for Agrobacterium survival and plant cell transformation during agroinfiltration. |
| Carborundum (Silicon Carbide) Powder | Fine abrasive used in mechanical inoculation to gently wound the leaf cuticle, allowing viral entry. |
| Specific Pathogen Isolate | The disease-causing agent (e.g., fungus, oomycete, bacteria) used to challenge silenced plants and assess the function of the NBS gene in resistance. |
| qRT-PCR Primers | Validates the transcriptional level of the target NBS gene post-inoculation to confirm silencing efficiency. |
Visualized Workflows and Pathways
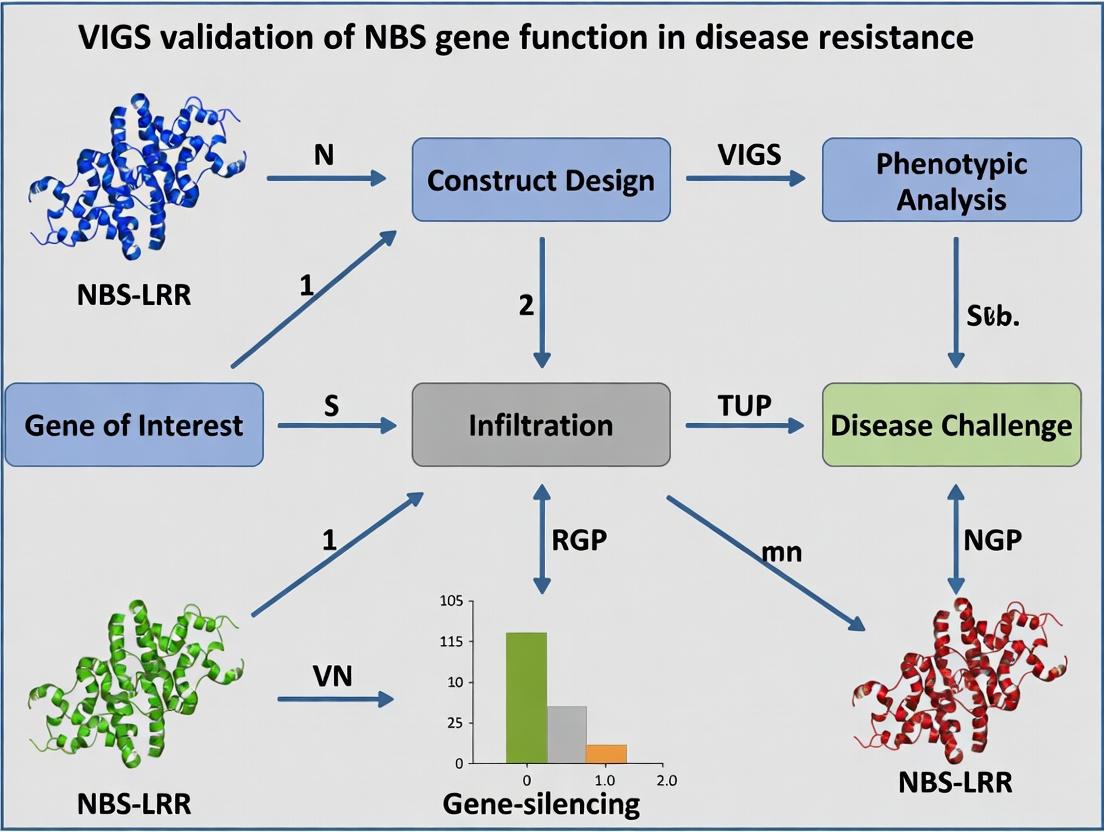
Title: VIGS Experimental Workflow for NBS Gene Validation
Title: NBS Gene Silencing Disrupts Disease Resistance Pathway
This guide compares key methodologies for co-inoculating a viral-induced gene silencing (VIGS) vector with a target pathogen, a critical step in validating nucleotide-binding site (NBS) gene function in plant disease resistance. The objective is to evaluate different timing, dosage, and assay design strategies to optimize phenotypic readout and ensure robust, interpretable data.
Performance Comparison: Co-inoculation Strategies
Table 1: Comparison of Co-inoculation Timing Strategies
| Timing Strategy | Protocol Synopsis | Key Advantage | Key Disadvantage | Best for Validating |
|---|---|---|---|---|
| Simultaneous Inoculation | VIGS vector and pathogen are mixed and inoculated concurrently. | Single experimental manipulation; ensures both agents are present in same tissue from onset. | Potential for direct physical/chemical inhibition between inocula; unclear initial silencing state. | NBS genes with early, constitutive defense roles. |
| Sequential Inoculation (Pathogen First) | Pathogen inoculation followed by VIGS vector after 24-48h. | Allows pathogen establishment, testing genes involved in later defense phases. | Silencing kinetics may be too slow to affect established infection. | NBS genes involved in pathogen recognition signaling. |
| Sequential Inoculation (VIGS First) | VIGS vector inoculation followed by pathogen challenge after 10-21 days. | Ensures maximal target gene silencing before pathogen encounter; most common approach. | Extended period increases experimental variables; plant development stage changes. | Most NBS genes, especially those in downstream signaling or effector-triggered immunity. |
| Refined Sequential (Optimal) | VIGS inoculation, then pathogen challenge at 14-16 days post-VIGS. | Balances strong silencing with manageable experimental timeline; recommended standard. | Requires careful scheduling and plant maintenance. | High-confidence validation of NBS function in basal resistance. |
Table 2: Comparison of Pathogen Inoculation Dosage (Challenge Levels)
| Dosage Level | Pathogen Concentration / Method | Goal | Risk with Low Silencing Efficiency | Data Interpretation |
|---|---|---|---|---|
| High | ~10⁶ CFU/mL (bacteria); Heavy spore suspension (fungi). | Overwhelm basal resistance to reveal subtle silencing effects. | Potential for overwhelming even wild-type resistance, masking phenotype. | Can be useful for strong resistance components. |
| Moderate (Recommended) | ~10⁵ CFU/mL; Moderate spore suspension. | Mimic natural infection pressure; differentiate silencing from control. | Optimal for clear, biologically relevant phenotypic distinction. | Standard for quantitative disease scoring. |
| Low | ~10⁴ CFU/mL; Dilute spore suspension. | Reveal genes essential for even weak pathogen detection. | Increased experimental noise; may require more replicates. | Useful for hypersensitivity or strong R-gene phenotypes. |
Table 3: Disease Assay Design and Quantification Methods
| Assay Type | Measurement Output | Throughput | Quantitative Rigor | Equipment Needs |
|---|---|---|---|---|
| Visual Disease Index | Categorical score (0-5) based on lesion size/chlorosis. | High | Low to Moderate; subjective. | None. |
| Lesion Area Measurement | Digital pixel count of necrotic/chlorotic areas. | Moderate | High; objective. | Imaging software (ImageJ). |
| Pathogen Biomass Quantification | qPCR of pathogen-specific genomic DNA or RNA. | Low | Very High; directly measures colonization. | qPCR thermocycler, specific primers. |
| Recommended Composite Assay | Combine Visual Index + Lesion Area + qPCR for key samples. | Moderate | Very High; multifaceted validation. | Full lab suite. |
Detailed Experimental Protocols
Protocol 1: Optimal Sequential Co-inoculation for Nicotiana benthamiana
- VIGS Inoculation (Day 0): Agroinfiltrate N. benthamiana leaves (4-5 leaf stage) with Agrobacterium strain GV3101 carrying the appropriate TRV-based VIGS vector (e.g., TRV2:NBS-gene). Include empty TRV2 and TRV2:PDS controls.
- Incubation (Days 1-14): Maintain plants under standard conditions (22-24°C, 16h light).
- Pathogen Challenge (Day 14):
- For Pseudomonas syringae pv. tomato DC3000: Prepare a suspension of 1x10⁵ CFU/mL in 10mM MgCl₂ with 0.025% Silwet L-77. Pressure-infiltrate the same leaf area previously infiltrated with VIGS vector using a needleless syringe.
- For Botrytis cinerea: Prepare spores in potato dextrose broth at 2.5x10⁴ spores/mL. Place a 10µL droplet on the abaxial side of the silenced leaf.
- Disease Assessment (Days 17-21):
- P. syringae: Harvest three leaf discs per plant, homogenize, serially dilute, and plate on KB with rifampicin for CFU counting.
- B. cinerea: Photograph lesions and calculate area using ImageJ.
Protocol 2: Pathogen Biomass Quantification via qPCR
- Sample Collection: Harvest and flash-freeze 100mg of leaf tissue from the inoculated zone.
- DNA Extraction: Use a CTAB-based method to isolate total genomic DNA.
- qPCR Reaction:
- Use pathogen-specific primers (e.g., P. syringae 16S rRNA) and plant-specific primers (e.g., N. benthamiana EF1α) as an internal control.
- Set up 20µL reactions with SYBR Green master mix.
- Run in triplicate on a standard qPCR cycler.
- Data Analysis: Calculate pathogen DNA relative to plant DNA using the 2^(-ΔΔCt) method, comparing VIGS-silenced plants to empty vector controls.
Visualizations
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for VIGS-Pathogen Co-inoculation Studies
| Item | Function & Importance in Validation | Example Product/Source |
|---|---|---|
| TRV-based VIGS Vectors (e.g., pTRV1, pTRV2) | Binary vectors for virus-induced gene silencing in plants. Essential for knocking down target NBS gene expression. | pTRV1/pTRV2 (Arabidopsis Resource Centre, Addgene). |
| Agrobacterium tumefaciens Strain GV3101 | Disarmed strain for efficient delivery of T-DNA containing VIGS constructs into plant cells via agroinfiltration. | Common lab strain, commercially available. |
| Target Pathogen Strains | Well-characterized bacterial, fungal, or oomycete pathogens for challenge assays. Defines the resistance phenotype. | e.g., P. syringae DC3000, B. cinerea B05.10. |
| Silwet L-77 or Tween-20 | Surfactant to reduce surface tension, ensuring even pathogen suspension spread during spray or dip inoculation. | Lehle Seeds, Sigma-Aldrich. |
| Pathogen-Specific qPCR Primers | For absolute or relative quantification of pathogen biomass in planta. Critical for objective resistance measurement. | Designed from conserved pathogen genes (e.g., ITS, EF1α). |
| Plant Reference Gene qPCR Primers | For normalizing pathogen biomass data to plant tissue input. Essential for data accuracy. | e.g., NbEF1α, NbACTIN. |
| Image Analysis Software | To objectively quantify disease lesion area and progression from photographs. | Fiji/ImageJ (open source). |
| Controlled Environment Growth Chamber | Provides consistent light, temperature, and humidity for reproducible plant growth and disease development. | Conviron, Percival, etc. |
Overcoming Hurdles: Troubleshooting Common VIGS Pitfalls and Enhancing Silencing Efficiency
Within the broader thesis on Virus-Induced Gene Silencing (VIGS) validation of Nucleotide-Binding Site-Leucine Rich Repeat (NBS-LRR) gene function in plant disease resistance, a critical bottleneck is achieving consistent, high-efficiency silencing. Poor silencing phenotypes can lead to false-negative results, confounding functional analysis. This guide objectively compares the performance of major VIGS vector systems and critical growth conditions, focusing on their impact on silencing efficiency and stability, to inform robust experimental design.
Comparative Analysis of Major VIGS Vector Systems
The stability of the inserted target fragment and viral spread are paramount for sustained silencing. Below is a comparison of the most widely used vectors.
Table 1: Comparison of VIGS Vector Performance for NBS-LRR Gene Silencing
| Vector System (Virus Backbone) | Avg. Silencing Efficiency (% Phenotype) * | Insert Stability (Duration of Silencing) | Ease of NBS-LRR Fragment Cloning | Known Off-Target Effects | Optimal Host Plant(s) |
|---|---|---|---|---|---|
| Tobacco Rattle Virus (TRV) | 70-90% | High (4-6 weeks) | High (Gateway/LIC compatible) | Low | Nicotiana benthamiana, Tomato, Arabidopsis |
| Bean Pod Mottle Virus (BPMV) | 60-80% | Moderate-High | Moderate | Moderate | Soybean, Common Bean |
| Barley Stripe Mosaic Virus (BSV) | 50-70% | Moderate | Moderate-Difficult | Low-Moderate | Barley, Wheat |
| Potato Virus X (PVX) | 40-60% | Low-Moderate (2-3 weeks) | High | High (severe viral symptoms) | N. benthamiana, Potato |
Data compiled from recent comparative studies (2022-2024). Efficiency refers to penetrance of visible silencing in *N. benthamiana PDS control experiments.
Experimental Protocol: Standard TRV-VIGS Efficacy Test
Objective: To compare silencing efficiency of different vector constructs targeting the Phytoene Desaturase (PDS) gene as a visual marker.
- Vector Construction: Clone a ~300bp fragment of the PDS gene into the TRV2 vector via Agrobacterium-mediated gateway recombination.
- Agroinfiltration: Grow Agrobacterium tumefaciens strain GV3101 carrying TRV1 and TRV2-PDS (or empty TRV2 control) to OD₆₀₀=0.5. Resuspend in MMA buffer (10 mM MES, 10 mM MgCl₂, 150 µM acetosyringone).
- Infiltration: Mix TRV1 and TRV2 cultures 1:1. Pressure-infiltrate the abaxial side of 2-3 true leaves of 2-week-old Nicotiana benthamiana plants.
- Environmental Control: Maintain infiltrated plants at 21°C with a 16/8-hour light/dark photoperiod.
- Phenotyping: Monitor for photobleaching (white leaf patches) indicative of PDS silencing, starting at 10-12 days post-infiltration (dpi). Quantify efficiency as the percentage of plants showing clear bleaching from 3 independent experiments (n≥20 plants/construct).
The Impact of Environmental Factors on Silencing Efficiency
Vector performance is heavily modulated by plant growth conditions. Suboptimal environments destabilize viral titer and host RNAi machinery.
Table 2: Effect of Environmental Factors on TRV-Mediated Silencing Stability
| Environmental Factor | Optimal Condition | Suboptimal Condition | Observed Impact on NBS-LRR Silencing (vs. PDS control) |
|---|---|---|---|
| Temperature | 20-22°C | >25°C or <18°C | Severe reduction (50-70% decrease). High temp accelerates viral clearance. |
| Light Intensity | 120-150 µmol/m²/s | <80 µmol/m²/s | Delayed onset (3-5 days) and reduced phenotype penetrance. |
| Plant Age | 2-3 leaf stage (2 weeks) | 4-5 leaf stage (4 weeks) | Significantly reduced efficiency and spatial spread in older plants. |
| Agroinfiltration OD | OD₆₀₀ = 0.4 - 0.6 | OD₆₀₀ > 1.0 | Hypervirulence, plant stress, and inconsistent silencing patterns. |
Experimental Protocol: Testing Temperature-Dependent Silencing Stability
Objective: To quantify the effect of post-infiltration temperature on the duration of NBS-LRR gene silencing.
- Plant Groups: Infiltrate four identical groups of plants (n=15) with TRV construct targeting an NBS-LRR gene of interest (GOI).
- Temperature Regimes: Post-infiltration, place each group into separate growth chambers set at: 18°C, 21°C (control), 24°C, and 27°C.
- Sampling: Collect leaf tissue from the same developmental stage across all groups at 7, 14, 21, and 28 dpi.
- Analysis: Perform qRT-PCR on sampled tissue to measure transcript abundance of the target NBS-LRR GOI. Normalize to housekeeping genes (e.g., EF1α). Silencing stability is inversely correlated with transcript rebound over time.
Visualizing Key Pathways and Workflows
Title: VIGS Mechanism and Environmental Disruption Points
Title: Optimized VIGS Experimental Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Robust VIGS Validation of NBS-LRR Genes
| Item | Function in VIGS Experiment | Example/Note |
|---|---|---|
| TRV1 & TRV2 Vectors | Binary viral vectors for Agrobacterium delivery; TRV2 carries the target insert. | pTRV1, pTRV2 (Liu et al., 2002) are the gold standard. |
| Gateway LR Clonase | Enzyme mix for efficient, site-specific recombination of target fragment into TRV2. | Enables high-throughput cloning of NBS-LRR gene fragments. |
| A. tumefaciens GV3101 | Disarmed, virulent Agrobacterium strain for plant transformation. | Preferred for its high transformation efficiency in Solanaceae. |
| Acetosyringone | Phenolic compound that induces Agrobacterium vir genes essential for T-DNA transfer. | Critical component of the agroinfiltration buffer. |
| Silencing Reporter Gene (PDS) | Visual marker gene causing photobleaching when silenced; validates system efficacy. | Must be run in parallel with every NBS-LRR experiment. |
| RT-qPCR Kit (One-Step) | For direct quantification of target NBS-LRR mRNA levels from plant tissue. | Confirms transcriptional knockdown beyond phenotypic observation. |
| Controlled Environment Chamber | Maintains precise temperature, light, and humidity post-infiltration. | Critical for reproducible silencing, as per Table 2 data. |
Managing Off-Target Effects and Non-Specific Phenotypes in NBS-LRR Families
Virus-Induced Gene Silencing (VIGS) is a cornerstone technique for rapidly validating the function of Nucleotide-Binding Site Leucine-Rich Repeat (NBS-LRR) genes in plant disease resistance. However, the high sequence similarity within NBS-LRR gene families presents a significant challenge: off-target silencing and consequent non-specific phenotypes. This comparison guide evaluates the performance of different VIGS vector design and validation strategies in mitigating these confounding effects, providing a critical framework for robust functional genomics.
Comparison of VIGS Design & Validation Strategies
Table 1: Performance Comparison of VIGS Fragment Selection Strategies
| Strategy | Principle | Efficacy in Specific Silencing (1-5) | Risk of Off-Target Phenotype | Key Validation Requirement | Best For |
|---|---|---|---|---|---|
| 3' UTR Targeting | Targets the untranslated region, often gene-specific. | 5 | Very Low | Confirm UTR sequence uniqueness in family. | Single, isolated NBS-LRR genes. |
| Gene-Specific Region (GSR) | Uses a unique, low-homology segment of the coding sequence. | 4 | Low | BLASTN against full transcriptome. | Genes with discernible unique domains. |
| VIGS with Mismatches | Introduces 3-5 silent mismatches in conserved regions to disrupt homology. | 3 | Medium | Test silencing efficiency vs. wild-type fragment. | Highly conserved gene clusters. |
| Whole Conserved Domain | Targets a shared domain (e.g., P-loop, Kinase-2). | 1 | Very High | Not recommended for single-gene validation. | Silencing entire subfamilies for broad screening. |
Table 2: Experimental Validation Techniques for Off-Target Effects
| Method | What It Measures | Throughput | Cost | Quantitative Data Output | Ability to Confirm Specificity |
|---|---|---|---|---|---|
| RT-qPCR (Multi-Gene) | Expression of target and closest homologs. | Medium | $$ | Ct values, fold-change. | High (if primers are specific). |
| RNA-Seq Transcriptomics | Genome-wide expression changes post-VIGS. | Low | $$$$ | FPKM/TPM values, differential expression. | Very High (gold standard). |
| Northern Blot | Detection of specific siRNA species. | Low | $ | siRNA band intensity/size. | Medium (probe dependent). |
| Phenotype Rescue | Complementation with a modified transgene. | Very Low | $$$ | Disease incidence/severity score. | Definitive Proof. |
Detailed Experimental Protocols
Protocol 1: Multi-Gene RT-qPCR for Off-Target Validation
- Design Primers: Create gene-specific qPCR primers for the target NBS-LRR and its 3-5 closest homologs. Amplicons must be in non-homologous regions and span an intron if using genomic DNA control.
- cDNA Synthesis: Extract total RNA from VIGS-treated and control tissues (≥ 3 biological replicates). Treat with DNase I. Synthesize cDNA using a reverse transcriptase kit.
- qPCR Run: Perform reactions in triplicate using a SYBR Green master mix. Include a stably expressed reference gene (e.g., EF1α, UBQ).
- Data Analysis: Calculate ΔΔCt values. Confirm target gene silencing (>70% reduction). Off-target silencing is significant if any homolog shows >40% reduction in expression relative to empty-vector VIGS controls.
Protocol 2: RNA-Seq Based Specificity Check
- Library Prep & Sequencing: Prepare strand-specific RNA libraries from VIGS-target and control plants. Sequence on an Illumina platform to a depth of ~30 million paired-end reads per sample.
- Bioinformatics Pipeline: Map reads to the reference genome/transcriptome using HISAT2 or STAR. Quantify gene expression with StringTie or featureCounts.
- Specificity Analysis: Perform differential expression analysis (DESeq2, edgeR). The primary signature of specific silencing is significant downregulation of only the intended target NBS-LRR gene. Downregulation of other NBS-LRR genes indicates off-target effects.
Visualizations
Title: Workflow for Managing VIGS Off-Target Effects in NBS-LRR Studies
Title: Specific vs. Off-Target NBS-LRR Signaling
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Specific VIGS Studies in NBS-LRR Genes
| Item / Reagent | Function in Experiment | Critical Specification for NBS-LRR Work |
|---|---|---|
| TRV-based VIGS Vectors (e.g., pTRV1, pTRV2) | Standard viral delivery system for inducing silencing in plants. | Use Gateway-compatible versions for high-throughput cloning of specific fragments. |
| High-Fidelity DNA Polymerase (e.g., Q5, Phusion) | Amplify NBS-LRR gene fragments for VIGS construct cloning with minimal errors. | Essential to prevent sequence drift that could alter off-target profiles. |
| Gene-Specific Silencing Fragment | The core reagent determining specificity. | Ideally 150-300 bp from the 3' UTR or a unique exon, verified by multiple BLAST analyses. |
| DNase I (RNase-free) | Remove genomic DNA contamination from RNA samples prior to RT-qPCR. | Critical for accurate expression measurement of closely related NBS-LRR genes. |
| Strand-Specific RNA-Seq Kit | Prepare libraries for transcriptome-wide analysis of silencing effects. | Required to accurately assign reads and detect antisense siRNA activity. |
| Reverse Transcriptase with High Processivity | Synthesize cDNA from often complex, GC-rich NBS-LRR transcripts. | Ensures complete coverage for quantifying all homologs via RT-qPCR. |
| Specific & Degenerate qPCR Primers | Quantify expression of target and related NBS-LRR homologs. | Primers must be rigorously tested for specificity and equivalent efficiency. |
| Reference Gene Primers (e.g., EF1α, ACTIN) | Normalize gene expression data in the specific plant tissue/stress condition. | Must be validated for stable expression under the pathogen inoculation conditions used. |
Optimizing Viral Titer and Plant Developmental Stage for Robust Knockdown
Within the broader thesis of using Virus-Induced Gene Silencing (VIGS) for validating Nucleotide-Binding Site (NBS) gene function in plant disease resistance, two critical parameters emerge for ensuring robust and reproducible knockdown: the titer of the viral vector and the developmental stage of the host plant. This guide compares the performance of different VIGS protocols by systematically evaluating these parameters against common alternatives, providing a framework for optimizing functional gene validation studies.
Comparative Performance Data
Table 1: Impact of Viral Titer and Plant Age on Silencing Efficiency in Nicotiana benthamiana
| Agroinfiltration OD600 | Plant Age (True Leaves) | Mean Silencing Efficiency (%) | Phenotype Penetrance | Duration of Effect (Days Post Infiltration) |
|---|---|---|---|---|
| 0.3 | 2 | 25 ± 8 | Low/Unreliable | 10-14 |
| 0.6 | 2 | 68 ± 12 | Moderate | 14-21 |
| 1.0 (Optimal) | 2 | 95 ± 5 | High & Uniform | 21-28 |
| 1.0 | 4 | 78 ± 15 | Variable | 14-21 |
| 1.5 | 2 | 92 ± 6 | High | 21-28 |
| 1.5 | 4 | 40 ± 10 | Low (Phytotoxicity) | 7-10 |
Table 2: Comparison of VIGS Vector Systems for NBS Gene Validation
| Vector System | Optimal OD600 | Optimal Plant Stage | Key Advantage for NBS Studies | Primary Limitation |
|---|---|---|---|---|
| TRV (Tobacco Rattle Virus) | 1.0 | 2-leaf | Broad host range, persistent silencing | Potential meristem exclusion |
| BSMV (Barley Stripe Mosaic Virus) | 0.8 | 1-leaf (monocots) | Effective in cereals/grains | Narrow host range |
| CLCrV (Cabbage Leaf Curl Virus) | 0.5 | Cotyledon stage | Strong in brassicas | Limited to specific plant families |
| ALSV (Apple Latent Spherical Virus) | 1.2 | 1-2 leaf | Minimal symptom development | Complex vector construction |
Detailed Experimental Protocols
Protocol 1: Standardized TRV-VIGS forN. benthamiana
Objective: To silence a candidate NBS-LRR gene and assess subsequent impact on pathogen resistance.
- Vector Preparation: Transform Agrobacterium tumefaciens strain GV3101 with the pTRV1 and pTRV2 plasmids (containing a ~300bp fragment of the target NBS gene). Grow single colonies in LB with appropriate antibiotics.
- Agrobacterium Culture: Inoculate 5ml starter cultures and grow overnight at 28°C. Subculture to 50ml of induction media (10 mM MES, 10 mM MgCl₂, 200 µM acetosyringone) and grow to an OD600 of 0.8-1.2.
- Infiltration Mixture: Pellet cells and resuspend in infiltration buffer (10 mM MgCl₂, 10 mM MES, 200 µM acetosyringone) to the final OD600 of 1.0. Mix pTRV1 and pTRV2 cultures in a 1:1 ratio. Let sit for 3-4 hours at room temperature.
- Plant Infiltration: Using a needleless syringe, pressure-infiltrate the abaxial side of leaves of 2-week-old N. benthamiana plants (2 true leaf stage).
- Validation: At 14-21 days post-infiltration, harvest tissue for qRT-PCR to quantify target gene transcript levels. Compare to plants infiltrated with an empty TRV2 vector (negative control) and a TRV2-PDS vector (positive control for visual bleaching).
- Phenotypic Assay: Challenge silenced plants with a compatible pathogen (e.g., Pseudomonas syringae pv. tomato DC3000). Monitor disease symptoms and quantify bacterial growth (CFU/g tissue) 3-5 days post-inoculation.
Protocol 2: High-Throughput VIGS Optimization Assay
This protocol allows for rapid comparison of multiple titer and age conditions.
- Prepare Agrobacterium harboring TRV2-target as in Protocol 1, but resuspend to create a gradient of ODs (0.3, 0.6, 1.0, 1.5).
- Infiltrate a grid pattern on leaves of plants at different developmental stages (e.g., 2-leaf, 3-leaf, 4-leaf).
- Use a fluorescent reporter gene (e.g., GFP) cloned into the TRV2 vector in parallel experiments to visualize spread and intensity of infection via UV light or fluorescence imaging at 5, 10, and 15 days post-infiltration. This correlates with silencing potential.
Visualizations
VIGS Workflow for NBS Gene Function Validation
Impact of NBS Gene Knockdown on Plant Immunity Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for VIGS-based NBS Gene Validation
| Reagent/Material | Supplier Examples | Function in Experiment |
|---|---|---|
| pTRV1 & pTRV2 Vectors | TAIR, Addgene | Binary VIGS vector system; TRV1 encodes replicase, TRV2 carries target gene fragment. |
| Agrobacterium tumefaciens GV3101 | Various (e.g., CIBSC) | Disarmed strain for efficient plant transformation and delivery of TRV constructs. |
| Acetosyringone | Sigma-Aldrich | Phenolic compound that induces Agrobacterium virulence genes, critical for T-DNA transfer. |
| SYBR Green qRT-PCR Master Mix | Thermo Fisher, Bio-Rad | For precise quantification of target NBS gene transcript levels post-silencing. |
| PDS (Phytoene Desaturase) Gene Fragment | Publicly available | Positive control insert for TRV2; silencing causes photobleaching, visually confirming VIGS efficiency. |
| Pathogen Strains (e.g., P. syringae) | Plant Pathology Labs, NCPPB | For challenging silenced plants to assess functional change in disease resistance. |
| Infiltration Buffer (10 mM MgCl₂, 10 mM MES) | Lab-prepared | Optimal medium for resuspending Agrobacterium prior to infiltration, maintaining cell viability. |
| N. benthamiana Seeds | Common scientific suppliers | Model plant host with high susceptibility to TRV and well-characterized immune responses. |
Within the critical framework of validating Nucleotide-Binding Site-Leucine-Rich Repeat (NBS-LRR) gene function in plant disease resistance using Virus-Induced Gene Silencing (VIGS), reproducibility is paramount. This guide compares VIGS protocols and positive controls, focusing on the phytoene desaturase (PDS) gene, against alternative validation methods, supported by experimental data.
Comparison of Gene Function Validation Strategies
The table below compares common approaches for validating NBS gene function in disease resistance studies.
| Method | Key Performance Metric (Typical Result) | Time to Result | Throughput | Required Expertise | Key Advantage for Reproducibility | Primary Limitation |
|---|---|---|---|---|---|---|
| VIGS with PDS Control | Silencing efficiency (95-100% photobleaching) | 3-4 weeks post-infiltration | Moderate-High | Moderate | Intrinsic visual positive control (PDS) validates entire workflow. | Transient; phenotype can be environment-sensitive. |
| Stable Genetic Transformation (KO/KI) | Homozygous mutant recovery rate (<5% for many crops) | 6-24 months | Low | High | Permanent, stable genotype; gold standard for publication. | Extremely resource and time-intensive. |
| CRISPR-Cas9 Editing | Editing efficiency at target locus (1-30% varies) | 4-12 months | Low-Moderate | High | Precise, heritable mutations. | Off-target effects; lengthy progeny screening. |
| Pharmacological Inhibition | Disease index reduction (0-80% inhibition) | 1-7 days | High | Low | Rapid, dose-responsive. | Lack of genetic specificity; potential pleiotropic effects. |
Quantitative Data from a Representative VIGS Experiment
The following data summarizes results from a typical experiment silencing the Rx (NBS-LRR) gene in Nicotiana benthamiana against Potato Virus X (PVX), using PDS as a control.
| Experimental Group | Silencing Target | Photobleaching (PDS phenotype) | Plants Showing Enhanced PVX Susceptibility | Mean Lesion Diameter (mm) ± SD | Viral Titer (ELISA OD405) ± SD |
|---|---|---|---|---|---|
| Positive Control | PDS | 100% (10/10 plants) | Not Applicable | Not Applicable | Not Applicable |
| Experimental Test | Rx (NBS-LRR) | 0% | 80% (8/10 plants) | 5.2 ± 0.8 | 1.45 ± 0.22 |
| Empty Vector Control | None | 0% | 10% (1/10 plants) | 1.5 ± 0.3 | 0.31 ± 0.05 |
| Untreated Wild-Type | None | 0% | 10% (1/10 plants) | 1.3 ± 0.4 | 0.28 ± 0.07 |
Detailed Experimental Protocols
1. VIGS Protocol for N. benthamiana using TRV Vectors
- Vectors: pTRV1 (RNA1 helper), pTRV2 (RNA2 with insert), pTRV2-PDS, pTRV2-NBS-LRR.
- Cloning: Amplify ~300-500 bp fragment of target NBS-LRR gene. Clone into pTRV2 via restriction ligation or Gateway recombination.
- Agrobacterium Preparation: Transform vectors into Agrobacterium tumefaciens strain GV3101. Grow single colonies in LB with antibiotics. Resuspend pelleted cells in infiltration buffer (10 mM MES, 10 mM MgCl₂, 200 µM acetosyringone, pH 5.6) to an OD₆₀₀ of 1.0.
- Infiltration: Mix pTRV1 and pTRV2-derived cultures 1:1. Incubate 3-4 hours. Pressure-infiltrate the abaxial side of 2-3 true leaves of 2-week-old N. benthamiana seedlings.
- Positive Control: Include plants infiltrated with pTRV1 + pTRV2-PDS. Distinct photobleaching should appear in new growth 10-14 days post-infiltration.
- Phenotyping: At 3 weeks post-VIGS, challenge silenced plants with the pathogen of interest (e.g., PVX inoculum). Score disease symptoms or quantify pathogen biomass 5-10 days later.
2. Replication Strategy for Robust VIGS Validation
- Biological Replicates: Minimum of 8-10 plants per construct, infiltrated across different flats.
- Independent Repeats: Perform the entire experiment (from Agrobacterium culture to phenotyping) at least three separate times.
- Randomization: Randomly assign plant positions in the growth chamber to minimize positional bias.
- Blinded Assessment: Where possible, phenotype scoring should be done by a researcher blinded to the treatment groups.
Diagrams
VIGS Workflow with PDS Control & Replication
NBS-LRR Signaling & VIGS Disruption
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in VIGS Validation | Key Consideration for Reproducibility |
|---|---|---|
| pTRV1 & pTRV2 Vectors | Backbone for the TRV VIGS system. pTRV2 carries the target gene insert. | Use verified, sequenced plasmid stocks from reputable repositories (e.g., Addgene). |
| Agrobacterium Strain GV3101 | Delivery vehicle for TRV vectors into plant cells. | Maintain consistent culture conditions and inoculation density (OD₆₀₀). |
| Acetosyringone | Phenolic compound that induces Agrobacterium vir genes for T-DNA transfer. | Fresh preparation in infiltration buffer is critical for consistent efficiency. |
| PDS Gene Fragment | The canonical positive control. Silencing causes photobleaching, confirming successful VIGS. | Use a species-specific fragment with proven high silencing efficiency. |
| Target NBS-LRR Fragment | ~300-500 bp gene-specific sequence for cloning into pTRV2. | Design to avoid off-target silencing; verify sequence specificity via BLAST. |
| Pathogen Isolate | The specific pathogen (e.g., virus, bacteria, fungus) used for disease challenge. | Use a well-characterized, standard isolate with known virulence. Maintain identical inoculum preparation. |
| Growth Chamber | Controlled environment for plant growth and phenotyping. | Document and standardize light intensity, photoperiod, temperature, and humidity across all replicates. |
Comparative Analysis of Viral-Induced Gene Silencing (VIGS) Vector Systems
This guide compares the safety, containment efficacy, and experimental performance of major VIGS vector systems used for validating Nucleotide-Binding Site-Leucine-Rich Repeat (NBS-LRR) gene function in plant disease resistance research. Effective containment is critical to prevent unintended viral spread and transgene escape into agricultural or natural ecosystems.
Table 1: Safety & Containment Profile of VIGS Vectors
| Vector System (Exemplar) | Host Range | Potential for Recombination | Seed Transmission Risk | Physical Containment Level Required | Key Experimental Safety Feature |
|---|---|---|---|---|---|
| Tobacco Rattle Virus (TRV) | Broad (Solanaceae, Arabidopsis) | Low (bipartite, segmented genome) | None documented | BSL-1P | Deletion of movement protein genes in some constructs. |
| Bean Pod Mottle Virus (BPMV) | Narrow (Primarily legumes) | Moderate | Low | BSL-1P | Use of non-enveloped, insect-transmissible only via specific beetles. |
| Barley Stripe Mosaic Virus (BSMV) | Narrow (Monocots, esp. cereals) | High (sequence homology with other Hordeiviruses) | Yes (seed transmission ~3-10%) | BSL-2P | Requirement for stringent growth chamber isolation. |
| Apple Latent Spherical Virus (ALSV) | Extremely Broad (Experimental) | Very Low (no known relatives) | None | BSL-1P | No insect vector identified; mechanical inoculation only. |
| Virus-Based Vector (pJL-TRBO) | Very Narrow (Nicotiana benthamiana) | Engineered to be Minimal | None | BSL-1 | Deletion of viral coat protein; limits cell-to-cell movement. |
Table 2: Performance in NBS-LRR Gene Function Validation
| Vector System | Silencing Efficiency in NBS-LRR Genes (%)* | Duration of Silencing (Weeks post-inoculation) | Off-Target Silencing Risk | Ease of Vector Construction (Cloning Strategy) | Key Experimental Data Supporting NBS-LRR Role in Resistance |
|---|---|---|---|---|---|
| TRV | 70-90% | 3-6 | Moderate (requires careful insert design) | Moderate (Gateway or restriction-based) | Robust, quantifiable loss-of-resistance phenotype; complemented by HR assays. |
| BPMV | 80-95% in soybean | 4-8 | Low (virus replicates slowly) | Complex (in vivo transcription required) | Strong correlation with R gene knockdown and increased pathogen susceptibility. |
| BSMV | 60-80% in barley/wheat | 2-4 | High (due to viral RdRp activity) | Moderate ( in vitro transcription) | Validates monocot-specific NBS-LRR function against rusts/mildews. |
| ALSV | >90% | 6-10 (systemic & stable) | Very Low | Simple (agroinfiltration of binary plasmid) | Provides clear, prolonged phenotyping window for resistance breakdown. |
| pJL-TRBO | 95-99% (transient, local) | 1-2 | Low | Very Simple (agroinfiltration of T-DNA) | Excellent for rapid, high-throughput N. benthamiana assays (e.g., with Phytophthora). |
*Efficiency measured by qRT-PCR of target transcript in silenced tissue.
Experimental Protocols for Safe and Contained VIGS
Protocol 1: TRV-Based VIGS with Enhanced Containment for NBS-LRR Validation
Objective: To silence a candidate NBS-LRR gene in Nicotiana benthamiana for functional validation against Phytophthora infestans, using a movement-protein-deficient TRV vector for containment.
- Vector Preparation: Clone a 200-300 bp unique fragment of the target NBS-LRR gene into the pTRV2-MCS vector (lacking a functional movement protein). Transform into Agrobacterium tumefaciens strain GV3101.
- Plant Growth & Inoculation: Grow plants in a contained growth chamber (BSL-1P). Agroinfiltrate 4-week-old plants with a 1:1 mixture of Agrobacterium carrying pTRV1 (RNA-dependent RNA polymerase) and the recombinant pTRV2. Include empty pTRV2 as a negative control.
- Containment Measures: Perform all work on a contained bench. Decontaminate all waste material (soil, plant tissue) by autoclaving. Prevent aphid access to chambers (potential passive vectors).
- Phenotyping & Validation: At 3 weeks post-infiltration, challenge silenced leaves with P. infestans zoospores. Quantify disease lesions vs. control. Confirm silencing via qRT-PCR and assess any unintended effects on related NBS-LRR genes.
Protocol 2: Seed Transmission Risk Assessment for BSMV Vectors
Objective: To empirically evaluate the seed transmission risk of a BSMV VIGS construct in barley, a critical containment metric.
- Inoculation: In vitro transcribe infectious RNA from BSMV clones (γ-vector containing NBS-LRR insert). Rub-inoculate barley plants at the 2-leaf stage. Maintain in vector-proof cages.
- Seed Harvest: Collect seed from all spikes of inoculated plants, label separately.
- Progeny Testing: Sow 100+ seeds from each plant line in sterile soil in an isolated containment greenhouse. Monitor seedlings for 3 weeks for viral symptom development (chlorosis, striping).
- Molecular Confirmation: Perform RT-PCR on asymptomatic and symptomatic leaf tissue using BSMV coat protein-specific primers to detect latent infection. Calculate seed transmission rate.
Diagrams
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in VIGS/NBS-LRR Research | Key for Safety/Containment |
|---|---|---|
| Deconstructed Viral Vectors (e.g., TRVΔMP) | Engineered to lack functional movement or coat proteins, limiting viral spread within the host and to other plants. | Primary biological containment. |
| Agrobacterium tumefaciens (GV3101) | Delivery system for VIGS constructs via agroinfiltration. Non-pathogenic, disarmed strain. | Confines viral replication to infiltrated tissue unless movement proteins are present. |
| BSL-1P / BSL-2P Growth Chambers | Physically enclosed plant growth facilities with negative air pressure and insect screening. | Prevents escape of viable viral particles or vectors; required by guidelines. |
| RNase III/Dicer-Substrate siRNA Design Tools | Bioinformatics software to design optimal target inserts that minimize off-target silencing of non-target genes, including related NBS-LRR family members. | Enhances experimental specificity and safety of conclusions. |
| Hairpin RNA (hpRNA) Vectors (as alternative) | Non-viral, DNA-based silencing method (e.g., pHANNIBAL). No viral genome involved, eliminating vector escape risk. | Maximum containment alternative to VIGS for stable transformation studies. |
| VIGS Validation Kit (qRT-PCR Primers) | Target-specific and housekeeping gene primers to quantitatively measure silencing efficiency and specificity. | Critical for confirming the molecular phenotype and ruling out non-specific effects. |
| Autoclave Bags & Biohazard Waste Stream | For safe disposal of all plant tissue, soil, and consumables exposed to recombinant viral vectors. | Essential final step to prevent environmental persistence. |
Beyond Phenotypes: Molecular Validation and Comparative Analysis with Alternative Methods
Within the context of a broader thesis investigating Virus-Induced Gene Silencing (VIGS) for validating the function of Nucleotide-Binding Site Leucine-Rich Repeat (NBS-LRR) genes in plant disease resistance, confirming successful knockdown is a critical first step. This guide objectively compares three principal methodologies—quantitative Reverse Transcription PCR (qRT-PCR), Western Blot, and Enzyme-Linked Immunosorbent Assay (ELISA)—for quantifying transcript and protein levels post-VIGS. Each technique offers distinct advantages and limitations in sensitivity, specificity, throughput, and cost, which are detailed below with supporting experimental data.
Performance Comparison of Validation Methods
The following table summarizes the key performance metrics for each technique based on current research and standardized protocols.
Table 1: Comparison of Methods for Confirming NBS-LRR Knockdown
| Feature | qRT-PCR (Transcript) | Western Blot (Protein) | ELISA (Protein) |
|---|---|---|---|
| Target | mRNA transcript levels | Full-length/cleaved protein | Total or specific protein isoforms |
| Sensitivity | Very High (can detect < 2-fold change) | Moderate to High (nanogram range) | High (picogram to nanogram range) |
| Specificity | High (sequence-specific primers/probes) | High (antigen-antibody interaction) | Very High (dual antibody sandwich) |
| Throughput | High (96/384-well formats) | Low to Moderate (gels/blots are manual) | Very High (96/384-well formats, automatable) |
| Quantification | Relative (ΔΔCt) or Absolute (standard curve) | Semi-quantitative (densitometry) | Fully Quantitative (standard curve) |
| Time to Result | Fast (~3-4 hours post cDNA synthesis) | Slow (1-2 days with blotting) | Moderate to Fast (several hours to overnight) |
| Key Advantage | Early confirmation of silencing; high precision. | Confirms protein size/post-translational modification. | Excellent for soluble proteins; high throughput. |
| Key Limitation | Does not confirm protein-level knockdown. | Non-linear detection; challenging quantification. | Requires high-quality, specific paired antibodies. |
| Typical Cost per Sample | Low to Moderate | Moderate | Moderate to High (antibody cost) |
Detailed Experimental Protocols
Protocol for qRT-PCR Validation of NBS-LRR Transcript Knockdown
- Sample Preparation: Total RNA is extracted from VIGS-treated and control plant tissue (e.g., leaf discs) using a silica-column or TRIzol-based method. RNA integrity is verified via agarose gel electrophoresis or Bioanalyzer.
- cDNA Synthesis: 1 µg of DNase-treated total RNA is reverse transcribed using an oligo(dT) or gene-specific primer and a reverse transcriptase enzyme (e.g., M-MLV).
- qPCR Reaction: Reactions are set up in triplicate containing: 2X SYBR Green or TaqMan Master Mix, gene-specific forward and reverse primers (designed to span an intron if possible), and diluted cDNA template. A housekeeping gene (e.g., EF1α, Actin) is run in parallel for normalization.
- Data Analysis: The cycle threshold (Ct) values are analyzed using the 2^(-ΔΔCt) method. VIGS-treated sample Ct values for the target NBS-LRR gene are compared to control samples, with results expressed as fold-change relative to the control.
Protocol for Western Blot Validation of NBS-LRR Protein Knockdown
- Protein Extraction: Tissue is homogenized in RIPA buffer with protease inhibitors. The lysate is centrifuged, and the supernatant's protein concentration is determined via Bradford or BCA assay.
- Gel Electrophoresis: Equal amounts of protein (20-50 µg) are separated by SDS-PAGE (8-12% gel depending on protein size) alongside a pre-stained protein ladder.
- Membrane Transfer: Proteins are transferred to a PVDF or nitrocellulose membrane via wet or semi-dry transfer.
- Immunoblotting: The membrane is blocked with 5% non-fat milk in TBST, then incubated overnight at 4°C with a primary antibody specific to the target NBS-LRR protein or an epitope tag (e.g., HA, Myc) if tagged. After washing, a horseradish peroxidase (HRP)-conjugated secondary antibody is applied.
- Detection & Analysis: Chemiluminescent substrate is added, and signal is captured via a digital imager. Band intensity is quantified using software (e.g., ImageJ) and normalized to a loading control (e.g., Actin, Tubulin).
Protocol for ELISA Validation of NBS-LRR Protein Knockdown
- Plate Coating: A 96-well plate is coated with a capture antibody specific to the NBS-LRR protein in carbonate coating buffer overnight at 4°C.
- Blocking & Sample Incubation: The plate is blocked with 1% BSA in PBS. Protein extracts from control and VIGS-treated samples (and a serial dilution of recombinant protein for a standard curve) are added to wells and incubated.
- Detection Antibody Incubation: After washing, a biotinylated or enzyme-conjugated detection antibody (recognizing a different epitope than the capture antibody) is added to form a "sandwich."
- Signal Development & Readout: For colorimetric detection, a substrate like TMB is added after subsequent streptavidin-HRP incubation (if biotinylated). The reaction is stopped with acid, and absorbance is read at 450 nm.
- Data Analysis: Protein concentration in samples is interpolated from the standard curve. Knockdown efficiency is calculated as the percentage reduction in the VIGS sample compared to the control.
Experimental Workflow Visualization
Title: Workflow for Confirming NBS-LRR Knockdown Post-VIGS
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for NBS-LRR Knockdown Validation
| Reagent Category | Specific Item | Function in Experiment |
|---|---|---|
| Nucleic Acid Analysis | DNase I, RNase Inhibitor | Ensures RNA purity and integrity for qRT-PCR. |
| High-Fidelity Reverse Transcriptase | Converts RNA to cDNA with high efficiency and fidelity. | |
| Gene-Specific Primers / TaqMan Probes | Enables specific amplification and detection of NBS-LRR transcript. | |
| Protein Analysis | RIPA Lysis Buffer, Protease Inhibitor Cocktail | Efficiently extracts and stabilizes NBS-LRR protein from plant tissue. |
| HRP-Conjugated Secondary Antibodies | Amplifies signal from primary antibody for chemiluminescent detection. | |
| Recombinant NBS-LRR Protein | Essential standard for generating quantitative curves in ELISA. | |
| Key Antibodies | Phospho-Specific Antibodies (if applicable) | Detects activation state of NBS-LRR proteins involved in signaling. |
| Tag-Specific Antibodies (HA, Myc, FLAG) | Used if the NBS-LRR gene is epitope-tagged during VIGS construct design. | |
| Detection | Chemiluminescent Substrate (e.g., ECL) | Generates light signal for Western Blot imaging. |
| Colorimetric ELISA Substrate (e.g., TMB) | Produces color change proportional to target protein amount. |
Within the framework of validating Nucleotide-Binding Site (NBS) gene function via Virus-Induced Gene Silencing (VIGS), accurate quantification of the resulting disease phenotype is paramount. This guide compares the core methodologies employed to measure disease resistance, providing experimental data to illustrate their applications, strengths, and limitations in the context of functional genomics research.
Comparison of Core Quantification Methodologies
The following table summarizes the primary techniques for assessing disease resistance in plant-pathogen interaction studies, particularly post-VIGS.
Table 1: Comparison of Disease Resistance Quantification Methods
| Method Category | Key Metric | Resolution & Objectivity | Primary Application in VIGS Validation | Typical Experimental Output | Throughput |
|---|---|---|---|---|---|
| Disease Scoring Scales | Visual Symptom Severity (e.g., 0-5 scale) | Low to Moderate (Subjective) | Initial, rapid phenotype screening; classifying resistance type (e.g., HR vs. susceptibility). | Ordinal data (e.g., mean disease index = 3.2). | High |
| Biomass Measurement | Host or Pathogen Biomass (Fresh/Dry weight, [CFU]/gram) | High (Quantitative) | Quantifying in planta pathogen growth; measuring fitness cost of resistance. | Continuous data (e.g., 2.5 x 10^5 CFU/mg leaf tissue). | Moderate |
| Pathogen Load Assays | Pathogen-Specific DNA/RNA Quantification (qPCR/dPCR) | Very High (Absolute Quantitative) | Precise, sensitive measurement of pathogen proliferation, independent of symptoms. | Continuous data (e.g., 50 pg pathogen DNA/ng total DNA). | Low to Moderate |
Supporting Experimental Data: In a recent VIGS study targeting a candidate NBS-LRR gene in Nicotiana benthamiana challenged with Phytophthora infestans, the following comparative data was obtained 7 days post-inoculation (dpi):
Table 2: Representative Data from a VIGS-NBS Gene Validation Experiment
| Plant Group | Mean Disease Index (0-5) | Pathogen Biomass (ng P. infestans DNA/µg plant DNA) | Plant Fresh Weight (g) at 7 dpi |
|---|---|---|---|
| TRV::NBS (Silenced) | 4.1 ± 0.3 | 18.5 ± 2.7 | 0.85 ± 0.11 |
| TRV::00 (Empty Vector Control) | 2.3 ± 0.4 | 5.2 ± 1.1 | 1.22 ± 0.09 |
| Mock Inoculation | 0.0 ± 0.0 | 0.01 ± 0.005 | 1.45 ± 0.08 |
Data shows that silencing the NBS gene leads to significantly higher disease scores, pathogen load, and reduced host biomass compared to the control, confirming its role in resistance.
Detailed Experimental Protocols
Protocol 1: Standardized Disease Scoring for Foliar Pathogens
- Inoculation: Prepare a standardized spore suspension (e.g., 1x10^5 spores/mL) of the pathogen (e.g., Colletotrichum higginsianum). Inoculate leaves of VIGS-silenced and control plants using a uniform method (e.g., droplet or spray).
- Incubation: Place plants in a high-humidity chamber at the appropriate temperature for pathogen infection (e.g., 24°C) for 24-48 hours, then transfer to normal growth conditions.
- Scoring: At defined time points (e.g., 5, 7, 10 dpi), assess individual leaves or plants using an agreed-upon scale. Example 0-5 scale: 0=No symptoms, 1=Small necrotic/chlorotic specks, 2=Moderate lesions without coalescence, 3=Severe, coalescing lesions, 4=Leaf collapse, 5=Plant death.
- Analysis: Calculate the Disease Index: DI = [Σ (disease grade * number of plants at that grade)] / (total number of plants * highest grade). Perform statistical analysis (e.g., non-parametric Kruskal-Wallis test).
Protocol 2: Quantitative Pathogen Load Assay via qPCR
- Sample Harvest: At the assessment time point, harvest tissue from the inoculation site (e.g., a leaf disk). Flash-freeze in liquid N₂. Store at -80°C.
- DNA Extraction: Use a commercial kit (e.g., DNeasy Plant Pro Kit) to co-extract genomic DNA from both host and pathogen. Include a standard curve from serially diluted pure pathogen DNA.
- qPCR Setup: Design species-specific primers for a single-copy pathogen gene (e.g., EF1α for fungi) and a single-copy host reference gene (e.g., Ubiquitin). Use a master mix with a fluorescent DNA-binding dye (e.g., SYBR Green).
- Quantification: Run samples in triplicate. Use the standard curve to determine the absolute quantity of pathogen DNA in each sample. Normalize the pathogen DNA amount to the host reference gene DNA amount to account for sampling variation.
- Analysis: Express data as picograms of pathogen DNA per nanogram of plant DNA. Compare means between experimental groups using ANOVA or t-tests.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for VIGS-Based Resistance Quantification
| Item/Reagent | Function in Experiment | Example Product/Catalog |
|---|---|---|
| VIGS Vector System | Delivers hairpin RNA to silence the target NBS gene in planta. | pTRV1/pTRV2 (Tobacco Rattle Virus) vectors |
| Agroinfiltration Strain | Agrobacterium tumefaciens strain for delivering VIGS constructs. | GV3101 or AGL1 electrocompetent cells |
| Pathogen-Specific Primers | For qPCR-based absolute quantification of pathogen biomass. | Custom-designed, validated oligonucleotides |
| Duplex qPCR Master Mix | Enables simultaneous amplification of pathogen and host reference targets in one well. | TaqMan Fast Advanced Master Mix |
| Plant DNA Extraction Kit | Efficient co-extraction of high-quality DNA from pathogen-infected plant tissue. | DNeasy Plant Pro Kit (Qiagen) |
| Standardized Pathogen Isolate | Genetically defined, virulent isolate for reproducible challenge assays. | e.g., Pseudomonas syringae pv. tomato DC3000 |
| Image Analysis Software | For semi-automated lesion area measurement from scored plants. | Fiji/ImageJ with lesion analysis plugins |
Visualizing the Integrated Workflow and Molecular Context
Integrated VIGS Validation Workflow
NBS Gene Function in Defense Signaling
The functional validation of Nucleotide-Binding Site-Leucine-Rich Repeat (NBS-LRR) genes using Virus-Induced Gene Silencing (VIGS) requires robust downstream molecular readouts. Measuring the activation of Salicylic Acid (SA) and Jasmonic Acid (JA) defense pathways, along with Pathogenesis-Related (PR) gene expression, provides critical evidence for the gene's role in disease resistance signaling. This guide compares common methodologies for quantifying these markers.
Comparison of Marker Quantification Methodologies
Table 1: Comparison of Techniques for Quantifying SA/JA Pathway Markers
| Method | Target | Throughput | Quantitative Precision | Key Experimental Data (Typical Results) | Primary Use Case |
|---|---|---|---|---|---|
| qRT-PCR (TaqMan) | Gene Expression (e.g., PR1, PDF1.2) | Medium-High | High (Absolute) | VIGS-silenced plants show 70-90% reduction in PR1 expression post-P. syringae challenge vs. control. | Validating transcriptional changes in specific pathway genes. |
| ELISA | Phytohormone levels (SA, JA) | Low-Medium | High | SA accumulation peaks at ~3.5 µg/g FW in resistant interactions, vs. ~0.8 µg/g FW in susceptible/VIGS-silenced. | Direct, precise measurement of hormone concentration in tissue. |
| Transcriptome RNA-Seq | Whole transcriptome | Low | High (Relative) | Identifies coordinated suppression of entire SA regulon (e.g., 20+ genes) upon NBS-LRR silencing. | Discovery of novel downstream targets and pathway crosstalk. |
| Reporter Gene Assays (GUS/LUC) | Promoter activity of marker genes | Low | Semi-Quantitative | PR1::LUC luminescence reduced by ~80% in silenced plants after elicitor treatment. | Spatiotemporal visualization of pathway activation in vivo. |
Table 2: Characteristic Molecular Markers for SA vs. JA Pathway Activation
| Pathway | Key Phytohormone | Signature Marker Genes | Typical Inducing Pathogen | Expected Change in NBS-LRR VIGS Validation (Susceptible Phenotype) |
|---|---|---|---|---|
| SA-Mediated | Salicylic Acid | PR1, PR2, PR5, NPR1 | Biotrophs (e.g., Pseudomonas syringae) | Significant downregulation (≥70%) post-inoculation. |
| JA-Mediated | Jasmonic Acid | PDF1.2, VSP2, LOX2 | Necrotrophs & Herbivores (e.g., Botrytis cinerea) | Significant downregulation (≥70%) post-inoculation. |
| SA/JA Crosstalk | SA & JA | WRKY70, MYC2 | Complex Infections | Altered expression ratio indicating disrupted balance. |
Experimental Protocols for Key Assays
Protocol 1: qRT-PCR for PR Gene Expression in VIGS Plants
- Sample: Harvest leaf tissue from TRV:00 (empty vector control) and TRV:NBS (gene-silenced) plants at 0, 12, 24, and 48 hours post-inoculation with pathogen or mock treatment.
- RNA Extraction: Use a TRIzol-based method. Treat with DNase I.
- cDNA Synthesis: Use 1 µg total RNA with oligo(dT) primers and reverse transcriptase.
- qPCR: Prepare reactions with gene-specific primers (PR1, PDF1.2, and a housekeeping gene like EF1α). Use SYBR Green master mix. Run in triplicate.
- Analysis: Calculate relative expression using the 2^(-ΔΔCt) method, normalizing to the housekeeping gene and the 0h time point in the control plant.
Protocol 2: Salicylic Acid Quantification by ELISA
- Extraction: Homogenize 100 mg frozen leaf tissue in 1 mL 90% methanol. Centrifuge.
- Drying: Evaporate the supernatant under vacuum.
- Resuspension: Dissolve the residue in 500 µL assay buffer from a commercial SA ELISA kit.
- Assay: Follow kit instructions. Typically involves adding sample to SA-specific antibody-coated wells, followed by an SA-enzyme conjugate and substrate.
- Quantification: Measure absorbance. Determine SA concentration from a standard curve. Express as µg per gram fresh weight (µg/g FW).
Pathway and Workflow Diagrams
SA and JA Defense Signaling Pathways
VIGS Workflow for NBS Gene Validation
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Downstream Marker Analysis
| Item | Function in Experiment | Example/Note |
|---|---|---|
| TRIzol/RNAiso Plus | Total RNA isolation from plant tissue. Maintains RNA integrity for downstream assays. | Essential for qRT-PCR and RNA-Seq. |
| Reverse Transcriptase Kit | Synthesizes cDNA from purified mRNA templates. | Use with RNase inhibitor and oligo(dT)/random primers. |
| SYBR Green qPCR Master Mix | Fluorescent dye for real-time quantification of PCR products. | Enables sensitive detection of PR gene transcript levels. |
| SA & JA ELISA Kits | Immunoassay for precise, quantitative measurement of phytohormone concentration. | Pre-coated plates provide high specificity and sensitivity. |
| Luciferase (LUC) Assay Kit | Detects luminescence from reporter genes driven by pathway-specific promoters. | For non-destructive, temporal tracking of signaling. |
| Pathogen Strain | Biotic elicitor to trigger defense pathways. | e.g., Pseudomonas syringae pv. tomato (SA), Botrytis cinerea (JA). |
Introduction In plant functional genomics, particularly for validating Nucleotide-Binding Site-Leucine-Rich Repeat (NBS-LRR) gene function in disease resistance, selecting the appropriate gene silencing or editing tool is crucial. This guide provides a comparative analysis of Virus-Induced Gene Silencing (VIGS) against three established alternatives—Stable RNA interference (RNAi), T-DNA insertional knockouts, and CRISPR-Cas9 editing—focusing on their application in validating disease resistance genes.
Methodological Protocols
1. VIGS for NBS Gene Validation
- Key Reagents: Tobacco rattle virus (TRV)-based vectors (TRV1, TRV2-NBS-gene fragment), Agrobacterium tumefaciens strain GV3101, target plant seedlings (e.g., Nicotiana benthamiana).
- Protocol: A ~300 bp gene-specific fragment from the target NBS gene is cloned into the TRV2 vector. TRV1 and recombinant TRV2 are transformed into Agrobacterium. Cultures are mixed and infiltrated into the leaves of young plants. Silencing is assessed 2-4 weeks post-infiltration via phenotyping (challenge with a pathogen) and qRT-PCR. Reduced target transcript levels coupled with enhanced disease susceptibility confirm function.
2. Stable RNAi
- Key Reagents: Hairpin RNA (hpRNA) construct in a binary vector, Agrobacterium for transformation, selectable markers (e.g., hygromycin resistance).
- Protocol: An inverted repeat of the target NBS gene sequence is cloned into an expression vector. Stable transgenic plants are generated via Agrobacterium-mediated transformation and selected on antibiotics. T1 or T2 homozygous lines with consistent silencing are used for detailed pathogen bioassays.
3. T-DNA Insertional Mutagenesis
- Key Reagents: T-DNA mutant seed collections (e.g., Arabidopsis T-DNA Express), gene-specific primers, border primers.
- Protocol: Public databases are screened for lines with T-DNA insertions within the target NBS gene. Homozygous mutants are identified by PCR genotyping. Knockout is confirmed via RT-PCR, and mutants are subjected to pathogen challenge.
4. CRISPR-Cas9 Knockout
- Key Reagents: sgRNA(s) designed against early exons of the NBS gene, Cas9 expression construct, Agrobacterium.
- Protocol: One or more sgRNAs are designed and cloned into a Cas9 binary vector. Stable transgenic plants are generated. The target locus is sequenced in T0/T1 plants to identify frameshift mutations. Transgene-free, homozygous mutant lines (T2 onwards) are used for functional phenotyping.
Comparative Performance Data
Table 1: Key Comparative Metrics for Gene Function Tools
| Feature | VIGS | Stable RNAi | T-DNA Knockout | CRISPR-Cas9 |
|---|---|---|---|---|
| Development Time | 3-5 weeks | 6-12 months | 3-6 months* | 6-9 months |
| Genetic Stability | Transient (weeks) | Stable (heritable) | Stable (heritable) | Stable (heritable) |
| Mutagenesis Type | Transcript degradation (knockdown) | Transcript degradation (knockdown/knockout) | Disruptive insertion (knockout) | Targeted indel/DSB (knockout/knockin) |
| Typical Efficiency | 70-95% silencing | Variable (10-90% silencing) | 100% knockout (if homozygous) | 10-60% mutation rate (T0) |
| Multiplexing Ability | Moderate (2-3 genes) | Difficult | None (single locus) | High (multiple genes) |
| Off-Target Effects | Moderate (sequence homology-dependent) | High (RNAi cross-talk) | Low (random insertion) | Moderate (sgRNA-dependent) |
| Requires Transformation | No (transient infiltration) | Yes (stable) | No (available pools) | Yes (stable) |
| Primary Cost | Low | High | Low (if available) | Medium-High |
*Assumes availability of a characterized line; screening novel inserts takes longer.
Table 2: Suitability for NBS-LRR Gene Validation in Disease Resistance
| Application Context | Preferred Tool | Rationale & Experimental Data |
|---|---|---|
| High-Throughput Screening | VIGS | Study by Zhang et al. (2023) silenced 120 NBS genes in tomato; 12 conferred new susceptibility to Phytophthora, demonstrating rapid pre-screening utility. |
| Redundancy/Functional Analysis | CRISPR-Cas9 | Multiplex sgRNAs created double knockouts of paralogous NBS genes in rice, revealing complete loss of blast resistance not seen in single mutants (Wang et al., 2024). |
| Conclusive Loss-of-Function | T-DNA/CRISPR-Cas9 | Stable null alleles provide definitive evidence. T-DNA knockout of AtRPM1 showed consistent, heritable susceptibility versus variable VIGS penetration. |
| Studying Essential Genes | VIGS | Allows study of lethal phenotypes. VIGS of a developmental NBS gene in N. benthamiana caused a dwarfing phenotype manageable for concomitant pathogen assays. |
| Non-Transformable Species | VIGS | Remains the only reverse-genetics option for many crops (e.g., Allium, woody plants). |
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in NBS Gene Validation |
|---|---|
| TRV1 & TRV2 VIGS Vectors | RNA virus-based system for delivering target gene fragments to induce silencing. |
| Gateway or Golden Gate Cloning Kits | For rapid, high-fidelity assembly of hpRNA, CRISPR, or VIGS constructs. |
| Specific Pathogen Isolates | Defined pathogenic strains for consistent disease phenotyping post-silencing/editing. |
| qRT-PCR Assays | To quantitatively measure transcript knockdown of target NBS and defense marker genes (e.g., PR1). |
| Cas9-Nuclease Antibody | For screening transgenic CRISPR lines and selecting transgene-free progeny. |
| Next-Gen Sequencing Kit | For amplicon sequencing of CRISPR-targeted loci to characterize mutation profiles. |
Visualizations
VIGS Workflow for NBS Gene Validation
Genetic Perturbation of NBS-Mediated Immunity
Conclusion For the initial validation of NBS gene function in disease resistance, VIGS offers an unparalleled combination of speed and flexibility, making it ideal for preliminary screening. Stable RNAi provides persistent knockdown but with risks of off-target effects. For definitive, heritable loss-of-function studies, T-DNA knockouts (where available) or CRISPR-Cas9-generated mutants are the gold standards. CRISPR-Cas9 excels in dissecting gene families with functional redundancy. The choice hinges on the research timeline, species, genetic stability requirements, and the specific biological question within the disease resistance pathway.
Within the broader thesis on validating Nucleotide-Binding Site-Leucine-Rich Repeat (NBS-LRR) gene function in plant innate immunity, Virus-Induced Gene Silencing (VIGS) has emerged as a critical high-throughput reverse genetics tool. This guide objectively compares the performance, efficiency, and experimental outcomes of VIGS applications across three key model and crop species: tomato (Solanum lycopersicum), tobacco (Nicotiana benthamiana), and barley (Hordeum vulgare). The focus is on the functional validation of NBS genes conferring resistance to pathogens like bacteria, viruses, and fungi.
Comparative Analysis of VIGS Performance Across Species
The table below summarizes key parameters and experimental outcomes from recent, successful case studies validating NBS gene function.
Table 1: VIGS Protocol & Efficacy Comparison for NBS Gene Validation
| Parameter | Tomato (S. lycopersicum) | Tobacco (N. benthamiana) | Barley (H. vulgare) |
|---|---|---|---|
| Primary VIGS Vector | Tobacco Rattle Virus (TRV) | Tobacco Rattle Virus (TRV) | Barley Stripe Mosaic Virus (BSMV) |
| Target NBS Gene Example | Mi-1.2 (Root-knot nematode, aphids) | N (Tobacco Mosaic Virus) | Mla (Powdery mildew, Blumeria graminis) |
| Typical Inoculation Method | Agroinfiltration (seedling leaves) | Agroinfiltration (mature leaves) | In vitro RNA transcript rubbing (secondary leaves) |
| Time to Visible Silencing | 2-3 weeks post-inoculation | 1-2 weeks post-inoculation | 1-2 weeks post-inoculation |
| Key Pathogen Challenge | Meloidogyne javanica (nematode) | TMV (virus) | Blumeria graminis f.sp. hordei (fungus) |
| Primary Validation Phenotype | Loss of resistance: increased nematode galls | Loss of resistance: systemic HR failure & viral spread | Loss of resistance: increased fungal conidiophores |
| Quantitative Silencing Efficacy | 70-80% target transcript reduction | 80-95% target transcript reduction | 60-75% target transcript reduction |
| Experimental Duration (Inoculation to Phenotype) | 5-6 weeks | 3-4 weeks | 5-6 weeks |
Detailed Experimental Protocols
1. Protocol for Tomato NBS Gene Validation using TRV-VIGS (e.g., Mi-1.2)
- Vector Preparation: Transform Agrobacterium tumefaciens strain GV3101 with pTRV1 and pTRV2 vectors (containing a 300-500bp Mi-1.2 fragment). Culture agrobacteria and resuspend to OD₆₀₀=1.0 in induction medium (10 mM MES, 10 mM MgCl₂, 200 µM acetosyringone).
- Plant Infiltration: Mix pTRV1 and pTRV2 cultures 1:1. Pressure-infiltrate the abaxial side of 2-3 true leaves of 2-week-old tomato seedlings (cultivar carrying Mi-1.2) using a needless syringe.
- Gene Silencing: Grow infiltrated plants at 19-21°C (optimal for Mi-1.2 function) for 3 weeks to allow systemic silencing.
- Pathogen Challenge: Inoculate soil with ~5,000 Meloidogyne javanica second-stage juveniles. Maintain plants at 25°C (permissive for Mi-1.2 loss-of-function).
- Phenotyping & Validation: Assess gall formation on roots 3-4 weeks post-nematode inoculation. Quantify silencing via RT-qPCR on root and shoot tissue using NBS gene-specific primers.
2. Protocol for Tobacco NBS Gene Validation using TRV-VIGS (e.g., N gene)
- Vector Preparation: As above, using a pTRV2 vector containing a fragment of the N gene.
- Infiltration & Silencing: Infiltrate 3-4 week-old N. benthamiana plants. Maintain plants at 25°C for 10-14 days for silencing establishment.
- Pathogen Challenge: Mechanically inoculate a systemic, upper leaf with Tobacco Mosaic Virus (TMV).
- Phenotyping & Validation: Monitor for the loss of the hypersensitive response (HR) at the inoculation site and systemic spread of TMV (assayed by mosaic symptoms). Confirm silencing via RT-qPCR and western blot for N protein.
3. Protocol for Barley NBS Gene Validation using BSMV-VIGS (e.g., Mla13)
- In vitro Transcription: Linearize BSMV vectors (γ strain containing Mla13 insert, plus α and β components). Synthesize capped RNA transcripts in vitro using T7 or SP6 RNA polymerase.
- Plant Inoculation: Mix α, β, and γ transcripts 1:1:1 with inoculation buffer (FES buffer: 0.1M Glycine, 0.06M K₂HPO₄, 1% w/v bentonite, 1% w/v celite). Rub the mixture gently onto the first true leaf of 7-10 day-old barley seedlings.
- Gene Silencing: Incubate plants under high humidity for 24h, then transfer to normal conditions (20°C) for 10-14 days.
- Pathogen Challenge: Challenge the silenced secondary leaf with Blumeria graminis spores using a settling tower.
- Phenotyping & Validation: At 7 days post-inoculation, stain with trypan blue and quantify fungal haustoria formation or count colonies. Use leaf samples for RT-qPCR analysis of Mla13 transcript levels.
Visualization of Key Concepts
Title: VIGS Workflow for NBS Gene Silencing in Tomato/Tobacco
Title: Simplified NBS-LRR Mediated Disease Resistance Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for VIGS-based NBS Gene Validation
| Reagent/Material | Function in VIGS Experiment | Example or Typical Component |
|---|---|---|
| VIGS Viral Vector | Carries the host target gene fragment to initiate RNA silencing. | TRV (pTRV1/pTRV2), BSMV (α, β, γ strains), pTV00 (Bean Pod Mottle Virus for legumes). |
| Agrobacterium Strain | Delivers TRV-based DNA vectors into plant cells via agroinfiltration. | GV3101, AGL-1. |
| Induction Medium | Activates Agrobacterium vir genes for efficient T-DNA transfer. | Acetosyringone (200 µM), MES buffer, MgCl₂. |
| In vitro Transcription Kit | Synthesizes infectious RNA transcripts for BSMV/VIGS inoculation. | T7/SP6 RNA polymerase, RNase inhibitor, cap analog (e.g., m7GpppG). |
| Inoculation Abrasive | Creates micro-wounds for viral entry during mechanical inoculation. | FES buffer components: Celite, Bentonite clay. |
| Pathogen Isolate | Used to challenge silenced plants and assay for loss-of-resistance. | Meloidogyne spp., TMV, Pseudomonas syringae, Blumeria graminis. |
| RT-qPCR Reagents | Quantifies the efficiency of target NBS gene silencing. | Reverse transcriptase, SYBR Green master mix, gene-specific primers. |
| Histological Stain | Visualizes and quantifies pathogen growth structures. | Trypan blue (for fungi), phloxine B (for nematodes). |
Conclusion
VIGS has established itself as an indispensable, rapid tool for the functional validation of NBS-LRR genes, bridging the gap between genomic sequencing and proven disease resistance function. This guide has detailed the journey from understanding foundational genetics through meticulous protocol execution, problem-solving, and rigorous validation. The key takeaway is that a robust VIGS pipeline, when carefully designed and critically validated, can reliably prioritize candidate resistance genes for downstream applications. Future directions involve integrating VIGS with high-throughput phenomics, single-cell sequencing to decipher cell-type-specific responses, and leveraging findings to engineer synthetic NBS-LRR receptors. For biomedical research, understanding plant NBS-LRR signaling modules continues to inspire novel paradigms in mammalian innate immunity and the development of bio-inspired therapeutic strategies. The continued optimization of VIGS protocols will accelerate the discovery of durable resistance genes, directly contributing to sustainable agriculture and broader biodefense.